Wearable optical patches for non-invasive sweat sensing
Abstract
As a non-invasive and information-rich diagnostic biofluid, sweat offers a promising alternative to conventional blood analysis, circumventing issues of invasiveness, patient discomfort, and complex laboratory processing. Wearable optical patches for non-invasive sweat sensing are emerging as a transformative platform in personalized and preventive healthcare, enabling real-time, continuous monitoring of metabolic and electrolyte biomarkers. This review comprehensively summarizes recent advances in wearable flexible optical sweat sensors, focusing on the following primary optical mechanisms: colorimetry, surface-enhanced Raman scattering, fluorescence, electrochemiluminescence and other optical approaches. First, we discuss the integration of these sensing modalities with soft, stretchable substrates, such as hydrogels, textiles, paper, and polymer films, and highlight key innovations in sweat collection, fabrication techniques, adhesion, and encapsulation that enable robust wearable operation. Second, we examine performance metrics, current challenges, and future perspectives for translating these technologies from laboratory prototypes to clinical and commercial applications. Finally, the challenges and future perspectives of the next-generation wearable optical platforms for continuous sweat analysis and personalized health monitoring are discussed.
Keywords
INTRODUCTION
Wearable devices equipped with chemical, biological, and physical sensors and actuators are increasingly indispensable in people’s lives due to their wide applications, including individual health tracking and monitoring[1,2]. The ultimate aim is to create sensors that seamlessly integrate into everyday wearable forms such as wristbands, adhesive patches, garments or even epidermal tattoos, enabling continuous, real-time monitoring of various physiological indicators[3-6]. By tracking a range of signals from the body’s physiological processes, skin adhesive wearable devices provide critical and multi-parameter information of an individual’s health status[7-9]. Conventional blood analysis is limited by its invasiveness, discomfort, and lengthy processing time, and is also constrained by its reliance on well-trained staff and expensive, large-scale, and professional equipment[9]. Conversely, the analysis of other non-invasive body fluids, such as sweat, saliva[10,11], exhaled breath condensate[12,13], and urine can be collected painlessly. Among them, sweat analysis offers a distinct method that takes advantage of its non-invasiveness, comfort, and the capacity of continuous health monitoring[3,14,15]. Its non-invasive nature also allows for the seamless integration into wearable devices for daily health tracking, which is important for the early warning of diseases, evaluation of therapeutic efficiency, and long-term individual health monitoring[16,17]. Therefore, sweat sensing provides a promising method to realize real-time and non-invasive health management.
The remarkable potential of sweat as a diagnostic biofluid is attributed to its rich and physiologically relevant composition. Human eccrine sweat, actively produced by highly vascularized sweat glands from interstitial fluid and blood plasma, contains abundant biomarkers, ranging from metabolites (e.g., glucose, lactate[18], urea, uric acid, ascorbic acid) and electrolytes (e.g., Na+, K+, Cl-, Ca2+) to neuromarkers, hormones (e.g., cortisol, neuropeptide Y) and even exogenous substances such as drugs[19-21]. These biomarkers correspond to distinct physiological states: sweat Cl- concentration is used for cystic fibrosis diagnosis[3,16,22]; sweat glucose correlates to blood glucose levels for potential diabetes management[3,16,23]; lactate serves as an indicator for anaerobic metabolism and fatigue[3,18,24]; and cortisol is a key marker for stress[3,16,25,26]. Evidence from sweat patch studies suggests that the concentrations of certain biomarkers (e.g., hormones and proteins) in sweat can correlate directly with their blood levels, enabling the non-invasive monitoring of critical physiological parameters typically measured in blood[19,27,28]. The existing clinical applications for sweat analysis including sweat chloride testing for cystic fibrosis analysis[29], and the promising uses cover monitoring sweat volume and electrolyte loss for dehydration diagnosis, measuring cortisol for stress evaluation, and analyzing drug contents to guide medical dosage[19,29-32]. Collectively, sweat analysis presents a compelling avenue for non-invasive and continuous health management, offering significant value for personalized healthcare[33-35].
Building on this foundational understanding of the diagnostic value of sweat, the technology of sensing platforms has progressed rapidly. Despite its relatively short history, sweat sensing has witnessed numerous innovative demonstrations for fundamental research, commercial applications, and clinical translation[19]. The most widely used sweat analysis method is electrochemical detection, which detects potentials or currents at functionalized electrodes to reflect biomarker concentrations in sweat[3,36-38]. However, this technology-driven design, particularly in electrochemical sensing platforms, often fails to adequately address real-world challenges imposed by human motion and the biological environment[19]. Consequently, a fundamental challenge of electrochemical systems lies in their motion artifacts and electronic noise, which largely affect signal integrity in sweat analysis[39]. Optical sensing, in contrast, offers a promising alternative for wearable sweat systems, beneficial for their cost-effectiveness and operational simplicity[40-42]. A principal advantage of optical analysis is its elimination of electronics in the sensor patch, avoiding the challenges of electronic noise and intricate wiring associated with electrochemical approaches[39]. For example, colorimetric sweat sensors operate via chemochromic mechanisms, where chromogenic molecules directly interact with target analytes to alter their colors, avoiding intricate wiring or batteries[43,44]. These optical sensors are built by flexible and stretchable soft materials such as polydimethylsiloxane (PDMS), hydrogels, and biodegradable cellulose paper, which exhibit superb biocompatibility, mechanical properties, and transparency, ensuring that colorimetric readings are unaffected and wearing comfort[43].
For effective long-term on-body monitoring, wearability and comfort at the skin-device interface are important[19]. Such wearable devices must be robust enough to prevent leakage or contamination, and furthermore, they can maintain non-invasiveness and ensure safety of the wearer[19]. The integration between the optical sensing materials and the flexible substrates should be powerful, enabling the sensing chemistry to be embedded within soft, stretchable, and skin-conformal materials for comfortable long-term wear and sustained performance. For instance, hydrogel, a type of polymer-based material crosslinked by a 3-dimensional (3D) network, shows advantages in sweat sensor substrates because of its good transparency, hydrophilicity, and water retention ability, which help sustain sensor performance during wearing[43]. Moreover, the integration of advanced fluid-handling technologies (e.g., microfluidics, superwettable interfaces) into optical sweat sensors enhances their performance. These innovations can improve sweat sampling efficiency, reduce backflow contamination and sweat evaporation, and increase detection accuracy[43]. Given that numerous works have been developed in this field in recent years and their great impact, several reviews have focused on the advanced sweat sensing technologies. For instance, Bariya et al. focused on the fundamental principles and system integration challenges of general wearable sweat sensors[3], but they lacked in-depth discussion of advanced optical sensing mechanisms. Min et al. delivered a thorough review of skin-interfaced sweat sensing technologies, including optical approaches; however, it lacked a dedicated and comparative analysis of the specific characteristics, detection performance, and practical applicability of different optical modalities for wearable sweat metabolite detection[45]. Wang et al. systematically summarized epidermal wearable optical sensors, but they did not sufficiently emphasize the flexibility aspect[43]. Heng et al. emphasized the system integration of flexible sweat sensors, while it lacked a focus on optical detection methods for metabolites[28]. To date, a comprehensive review that provides systematic comparison and specific details of wearable flexible optical sensors for sweat metabolites, encompassing advanced mechanisms, material innovations, microfluidic integration, and future application challenges, is still absent and urgently needed to bridge this critical gap and guide future developments in this emerging field.
This review aims to address this need by offering a structured, comparative perspective on the principal optical sensing modalities used in wearable sweat analysis [Figure 1]. Building on decades of innovation in sweat sensing that have demonstrated its applicability to commercial markets, clinical translation, and physiological monitoring[19]. The fundamental optical sensing mechanisms [e.g., colorimetry, surface-enhanced Raman scattering (SERS), fluorescence, and electrochemiluminescence (ECL)], as well as emerging approaches such as optical fiber-based sensing will be explored. A balanced assessment is provided of their underlying operating principles, performance characteristics including sensitivity, specificity, and multiplexing capability, and the practical considerations associated with their integration into flexible, skin-conformable devices. We discuss in detail the flexible materials and fabrication strategies that enable seamless integration with the skin, along with the incorporation of microfluidic systems for efficient sweat collection and transport, thereby offering a comprehensive, system-level perspective. By systematically comparing various optical modalities and elucidating their respective strengths and limitations, this review serves as a clear and informed guide for researchers and engineers seeking to select and optimize optical technologies for sweat sensing. Finally, we outline the current challenges and future directions for translating this promising technology from laboratory research into commercial and clinical practice. We emphasize that successful advancement in this field will require continued collaboration between engineers and physiologists to overcome existing technical barriers and facilitate effective clinical translation.
Figure 1. Schematic of a layered wearable microfluidic optical sweat sensing platform. The structure comprises (from bottom to top): a skin-conformal adhesive layer with sweat collection apertures; a flexible substrate (e.g., PDMS, hydrogel, textile, or paper) integrating a microfluidic network for sweat transport; an optical sensing zone (e.g., colorimetry, SERS, fluorescence, and ECL); and an encapsulation layer. Arrows indicate the path of optical signal transmission and the flow direction for analytical data output. Created in BioRender. Chu Wang. (2026) https://BioRender.com/dtltj3c, Adobe Illustrator, and Maxon Cinema 4D R21. PDMS: Polydimethylsiloxane; TPU: thermoplastic polyurethane; SERS: surface-enhanced Raman spectroscopy; ECL: electrochemiluminescence.
FLEXIBLE OPTICAL SENSING PLATFORMS
Flexible optical sensing platforms represent a pivotal advancement in wearable sweat analysis. By integrating flexible materials and optical transduction mechanisms, they enable non-invasive, real-time monitoring of metabolites. These platforms combine flexible substrates with optical detection techniques to achieve conformal skin contact, mechanical robustness, and high sensing performance. This section delves into the key components of such systems, starting with the flexible substrates that form their foundation, followed by critical aspects of platform fabrication techniques, sweat collection, adhesion, and encapsulation that ensure reliable operation under real-world conditions.
Flexible substrates
Flexible substrates are essential for wearable optical sweat sensors[46], providing mechanical compliance with skin deformations, long-term wearing comfort, and compatibility with optical sensing modalities. Various materials have been explored, including stretchable polymers, hydrogels, textiles, paper, and ultrathin or structured films [Figure 2]. These materials offer distinct advantages in flexibility, breathability, biodegradability, optical transparency, and integration with microfluidics and optical components.
Figure 2. Representative flexible substrates and their material characteristics for wearable optical sweat sensors. The illustrated substrates include PDMS, hydrogels, textiles, paper, and TPU. Created in BioRender. Chu Wang. (2026) https://BioRender.com/w58hd9a, Adobe Illustrator, and Maxon Cinema 4D R21. PDMS: Polydimethylsiloxane; TPU: thermoplastic polyurethane.
Polydimethylsiloxane
PDMS is widely used for fabricating various microfluidic chips owing to its flexibility, adhesion, and elasticity[43,47,48]. Its advantages include dimensional stability in water, biocompatibility, low modulus, elasticity, and compatibility with simple molding and bonding processes[49]. Fundamentally, PDMS is formed by cross-linking a silicon-based polymer, typically via mixing a base and a curing agent (e.g., 10:1 ratio), resulting in a hydrophobic, non-swollen elastomeric matrix with a continuous, non-porous structure[48]. For instance, a colorimetric microfluidic device can be fabricated by bonding two soft PDMS layers (e.g., a 200 μm microchannel layer and a 100 μm capping layer) via oxygen plasma treatment, maintaining flexibility and skin conformability[50]. Its hydrophobic nature and dimensional stability make it ideal for constructing defined, leakage-free microchannels[50], and its low elastic modulus (~ 1-3 MPa) ensures mechanical compatibility with skin[51]. Furthermore, PDMS-based microfluidic systems can be integrated with other flexible substrates such as textiles to merge wearability (the breathability of the textile) with mechanical stability (the inherent flexibility of PDMS)[51]. However, the hydrophobicity can impede passive sweat uptake into channels, often necessitating surface treatments[51]. Moreover, its non-porous structure offers no inherent fluid wicking capability, relying entirely on engineered microfluidics for sweat transport. Challenges in mass production and susceptibility to surface contamination also limit its use in low-cost, reusable applications.
Hydrogels
Hydrogels are 3D polymer networks with excellent biocompatibility, mechanical tunability, hydrophilicity, and water retention ability, making them inherently suitable for sweat sensing[43,52,53]. Unlike PDMS, hydrogels are formed by crosslinking hydrophilic polymer chains [e.g., polyvinyl alcohol (PVA), alginate, gelatin] in water, resulting in a soft, water-swollen, and often porous matrix[43]. They can be synthesized via various methods, including chemical crosslinking (e.g., free-radical polymerization), physical interactions (e.g., freeze-thaw cycles), and even rapid, initiator-free processes such as ultrasound cavitation[54]. For instance, a flexible SERS sensing platform was developed using a photo-crosslinked sulfonated cellulose-based hydrogel, demonstrating a facile preparation process[55]. The hydrogel network can absorb small molecules and sweat via hydrophilic groups, facilitating analyte diffusion and effective collection. Hydrogels reduce the sweat secretion resistance at the skin interface, and their hydrophilicity lowers the Laplace pressure barrier, allowing the hydrogel to serve as a sweat reservoir that wicks sweat away from skin to prevent local saturation[45,56,57]. This inherent porosity and hydrophilicity, a direct consequence of their aqueous formation, enable hydrogels to function as both the substrate and a passive pump for sweat, eliminating the need for complex microfluidic structures in some designs[45]. With a simple touch, such sensors can directly extract sweat and monitor biomarkers without additional stimulation[45]. A self-healing, flexible, colorimetric hydrogel-based sweat sensor has been designed by incorporating colorimetric reagents into a sucrose/PVA hydrogel for analyzing sweat metabolites such as glucose, pH value, Ca2+, and Cl-[43]. Similarly, a hydrogel fluorescence patch loaded with upconversion nanoparticles (UCNPs) enables rapid urea detection with minimal environmental and skin interference[43]. The porous construction of the hydrogel provides better permeability and lower impedance than nonporous materials, reducing the sample collecting time and sensing delay[45,58,59]. Specifically, the tunable porosity achieved through templates such as sucrose can significantly enhance permeability and reduce sensing lag[45]. Their tissue-like mechanical properties (e.g., tunable softness and high stretchability) and superior biocompatibility promote comfortable, long-term epidermal integration[59]. Hydrogels stand out as substrates due to their unique combination of bio‑friendliness, inherent water affinity, and tunable porosity, which together enable gentle skin interfacing and efficient sweat handling without external pumps or complex fluidics. Nevertheless, the high water content that enables their function also makes them prone to dehydration in air, potentially limiting operational lifetime. Their mechanical strength, while tunable, is generally lower than that of elastomers such as PDMS, and signal crosstalk in dense arrays can be an issue[54].
Textiles and fibers
Textiles, as the most common materials in daily wear, offer unique advantages for sweat sensors[60]. Their multilayer and reticulated nature allows them to withstand relatively large and long-term deformation[61-63]. Moreover, inter-fiber pores enhance sensor breathability, improving wearing comfort. Commonly used textile substrates are nylon, cotton, woven/knitted fabrics, and polyethylene terephthalate (PET), providing abrasion resistance and mechanical support for the fabricated sensing platforms. Sensors fabricated by fibers are shown to have superb sensing ability below 200% tensile deformation, with some displaying excellent bending and torsional performance[64]. For example, a carbon textile derived from silk fabric serves as a highly conductive, structure-maintained, nitrogen-doped electrode for detecting six biomarkers in sweat[60]. Commercial cotton textiles can be functionalized into SERS-active substrates by immobilizing gold nanoparticles. These substrates exhibit outstanding flexibility withstanding 1,000 folding cycles (bending radius: 2 mm) without significant SERS activity loss, and can be integrated with PDMS microfluidic channels for sweat transport[65]. Janus fabrics, fabricated by modifying one side of a polyester fabric with superhydrophilic SiO2 nanoparticles and the other with a superhydrophobic fluoropolymer, enable unidirectional sweat transport and can be coupled with microstructured optical fibers for SERS signal transmission[50]. Similarly, fluorescent Janus textiles coated on one side of cotton fabric with a fluorescent composite (e.g., Rhodamine B-doped PDMS) and on the other with a hydrophobic polyurethane (PU) layer allow real-time lactate monitoring and can be bent to a radius of 1 mm without fluorescence loss[66].
Paper-based substrates
Filter paper, a degradable cellulose natural material, offers an appropriate substrate for sweat sensors[43]. Paper-based substrates are low-cost, biodegradable, and highly porous, facilitating sweat absorption and transport[67,68]. For instance, a piece of Whatman Grade 1 filter paper can be embedded into a PDMS microfluidic device as a pre-loaded colorimetric reagent reaction zone, where its high porosity promotes efficient sweat absorption[50]. Plasmonic paper substrates can be fabricated by adsorbing silver nanoparticles (Ag NPs) onto filter paper fibers. The resulting flexible substrate can be folded and bent without cracking and retains its plasmonic properties even after multiple bending cycles[67,69]. Furthermore, cellulose-containing fluorescent sensors are prepared by linking fluorescent probes on paper substrates and integrating a cotton thread for sweat collection, making them compatible with portable imaging modules for in situ data capture[43].
Other polymer films and structured substrates
Besides PDMS, other polymer films such as polyimide (PI) and thermoplastic polyurethane (TPU) are also employed. PI films (e.g., Kapton) serve as flexible substrates for laser-induced graphene (LIG) sensors, which exhibit excellent flexibility (bending radius down to 5 mm) without significant conductivity loss and can be sewn into textile substrates to enhance wearability[70,71]. TPU possesses significant mechanical characteristics and good biosafety and can be processed into highly flexible and elastic microfibrous meshes via melt electrowriting[60]. The TPU microfibrous mesh shows ultra-high elasticity and flexibility, can be repeatedly folded and rubbed with nearly complete recovery, and its large pore size and high porosity provide unparalleled breathability[60]. Furthermore, electrospun Au/TPU nanofibrous mats exhibit a nanostructured porous architecture that conforms well to complex skin geometry and promotes sweat uptake via capillary action, offering a simple, low-cost fabrication route for SERS-active substrates[70].
Structural designs, including ultrathin and wrinkled architectures[72], can promote the flexibility of inherently rigid or soft materials. In ultrathin structures, bending strains reduce linearly with thickness, allowing nearly any material to become flexible and adapt to skin curvatures when made sufficiently thin[60]. Wrinkles, as natural 3D stretchable structures, can store pre-strain to absorb energy during stretching. This mechanism boosts stretchability and shape adaptability while providing an expansive surface area to facilitate electrochemical reactions with sweat[60]. For instance, a stretchable, wrinkled nanohybrid fiber featuring reduced graphene oxide (rGO)-covered gold nanowrinkles has been fabricated for sweat glucose detection[60].
Sweat collection
Effective and reliable sweat collection serves as a cornerstone for accurate metabolite sensing in wearable optical platforms, directly influencing data integrity. A key advancement in this field is the introduction of epidermal microfluidic systems that enable simultaneous sweat collection and in-flow analysis[39]. These systems address persistent challenges such as low sweat volumes, irregular sweating without stimulation, and potential biodegradation or evaporation[3], which have limited earlier methods. By leveraging efficient, miniaturized microfluidics specifically designed for passive sweat collection[39], such devices offer an integrated solution. They not only reduce evaporation and skin contamination[39] but also isolate sweat from the skin surface, thereby preventing continuous leaching of chemicals into the sweat[3]. Strategically designed microchannels direct older sweat away from the sensing zones while allowing freshly secreted sweat to flow in, which facilitates real-time analysis. For optical sensing specifically, these microfluidic channels act as dedicated conduits, delivering fresh sweat to sensing zones containing colorimetric or fluorescent reagents. This enables sequential analysis and volume measurement while protecting the sensing chemistry from the external environment, directly enhancing optical readout reliability and accuracy[73,74].
Beyond fundamental capture, sophisticated design enhances temporal resolution and functionality. For chronological sampling and dynamic analysis, passive capillary bursting valves (CBVs) with precisely engineered bursting pressure gradients are integrated into microfluidic networks to guide sweat in sequentially filling a series of chambers, spatially separating samples over time[71]. This enables time-sequenced capture and analysis, crucial for tracking metabolic dynamics. For instance, a patch with a 0.5 µL microchamber volume operating at 0.25 µL/min can fill each chamber within about 2 min, yielding a well-defined temporal profile[71]. Another critical aspect for continuous operation is ensuring that newly produced sweat promptly interacts with the sensor and is subsequently transported away to allow a constant reaction process. This can be achieved by designing channels within the adhesive layer to divert analyzed sweat away from the sensor surface[75]. Moreover, when natural passive sweat production is insufficient, localized sweat stimulation via transcutaneous iontophoretic delivery of agonists such as acetylcholine or pilocarpine provides an active means to generate adequate sample volume for subsequent detection and analysis[75]. Collectively, these strategies such as passive collection, timed valving, flow management, and active stimulation ensure the acquisition of high-integrity sweat samples for reliable optical sensing.
Fabrication techniques
Beyond the design of sweat collection and fluidic management systems, the practical realization of wearable optical sweat sensors hinges on advanced fabrication techniques that translate conceptual designs into functional, reliable devices. These techniques must accommodate a wide range of flexible substrates from elastomers and hydrogels to textiles and paper, while enabling the precise integration of microfluidic networks, optical sensing elements, and sometimes electronic components. The choice of fabrication method directly impacts sensor performance, scalability, and cost. The diverse manufacturing approaches for various optical sensors were summarized in Table 1.
Analytes in sweat and sensing performance of different optical sensors
| Detection Methods | Fabrication methods | Analytes | LOD | Detection range | Response time | Potential applications | Refs. |
| Colorimetry | Soft lithography/Laser ablation | Cl- | 0.1 mM | 10-100 mM | - | Sports science/Hydration monitoring | [76] |
| Paper-based fabrication | Lactate | 0.06 mM | 0-1 mM | - | Sports endurance/Fatigue analysis | [77] | |
| Soft lithography (Microfluidics) | Glucose | 3 μM | 25-100 μM | - | Health monitoring/Diabetes management | [90] | |
| Soft lithography (Microfluidics) | pH | 0.1 pH units | 4.5-6.5 | - | Skin health / Acid-base balance | [90] | |
| Soft lithography/Plasma bonding | Creatinine | - | 31.3-1,000 μM | < 1 min | Kidney function/Daily health | [50] | |
| Textile weaving/Nanoparticle coating | Urea | 3.05 mM | 3-250 mM | - | Kidney function/Skin-friendly monitoring | [91] | |
| Soft lithography/Molding | Vitamin C | - | 1-37 μM | < 5 min | Nutrition management | [89] | |
| Soft lithography / Molding | Calcium | - | 0.2-11 mM | < 5 min | Electrolyte balance / Nutrition | [89] | |
| Soft lithography/Molding | Zinc | - | 5-17 μM | < 5 min | Trace element monitoring | [89] | |
| Soft lithography/Molding | Iron | - | 0.4-20 μM | < 5 min | Trace element monitoring | [89] | |
| SERS | Hydrogel photo-crosslinking | Urea | 63.1 µM | 0.1-1,000 mM | ≤ 15 s | Metabolic monitoring | [55] |
| Dip coating on chromatography paper | Uric Acid | 1 µM | 20-100 µM | < 5 min | Point-of-care (Gout monitoring) | [67] | |
| Electrospinning | pH | 0.14 pH units | pH 4-8 | - | Wearable skin pH monitoring | [70] | |
| Textile integration/Optical fiber coupling | Sodium lactate | 0.1 mM | 0.1-100 mM | ~ 1 s | Real-time exercise monitoring | [66] | |
| Solid-state dewetting/Microfluidics | Lactate | 1 mM | 0-20 mM | - | Exercise physiology/Dietary monitoring | [71] | |
| Solid-state dewetting/Microfluidics | Tyrosine | 50 μM | 0-200 μM | - | Metabolic profiling | [71] | |
| Immunoassay assembly/Magnetic separation | Cortisol | 7 ng/mL | 12.5-400 ng/mL | - | Stress evaluation / Mental health | [78] | |
| Template-assisted electrodeposition | Glucose | 0.168 µM | 0.1-1,000 µM | - | Diabetes management | [124] | |
| Silver nanoflake deposition | Creatinine | - | 20-40 ng | - | Kidney health monitoring | [125] | |
| Fluorescence | Laser cutting (Paper microfluidics) | Glucose | 7 µM | 10-250 µM | - | Personalized perspiration analysis | [79] |
| Laser cutting (Paper microfluidics) | Lactate | 0.4 mM | 1.0-12.5 mM | - | Anaerobic metabolism tracking | [79] | |
| Laser cutting (Paper microfluidics) | Cl- | 5 mM | 10-100 mM | - | Cystic fibrosis screening/Hydration | [79] | |
| Laser cutting (Paper microfluidics) | pH | - | pH 4.1-6.6 | - | Skin health monitoring | [79] | |
| Soft lithography (Microfluidics) | Zn2+ | - | 1-30 µM | - | Exercise monitoring | [102] | |
| Soft lithography (Microfluidics) | Na+ | - | 5-100 mM | - | Electrolyte loss/Dehydration | [102] | |
| Film casting/Nanohybrid assembly | Glucose | - | - | - | Diabetic hyperglycemia monitoring | [104] | |
| Soft lithography (Microfluidics) | Total Amino Acids | 3.68 µM | 0-60 mM | < 20 min | Nutrient loss during exercise | [103] | |
| Soft lithography (Microfluidics) | Alanine | 0.49 µM | 0-8 mM | < 20 min | Nutrient loss during exercise | [103] | |
| Soft lithography (Microfluidics) | Lysine | 0.13 µM | 0-3 mM | < 20 min | Nutrient loss during exercise | [103] | |
| ECL | Hydrogel polymerization/Templating | 11 neurotransmitters (e.g., His, ATP, Glu) | 7.5 µM | 0.1-1.0 mM | - | Stress/Neurological health | [108] |
| Nanowire growth/Drop-casting | Glucose | 1.2 nM | 20 nM-12 mM | - | Non-invasive glucose monitoring | [112] | |
| Hydrogel encapsulation | Lactic acid | 2.52 µM | 5-30 µM | - | Muscle fatigue analysis | [109] | |
| Enzyme immobilization on CNTs | Lactate | 8.9 pM | 8.9 pM-8.9 μM | - | High-performance athlete monitoring | [110] | |
| Electrochemical polymerization (MIP) | Urea | 15.0 µM | 45 µM-19.5 mM | - | Renal function/Metabolic sensing | [106] | |
| Hydrogel self-assembly | Cl- | - | 1-800 µM | - | Motion and sweat analysis | [111] |
Several key fabrication paradigms have been developed. Soft lithography is a cornerstone technique for creating intricate microfluidic structures in elastomers such as PDMS. It involves casting and curing PDMS prepolymer against lithographically prepared silicon molds to produce solid elastomers with relief features, which are then bonded to form sealed microfluidic channels and reservoirs[50]. For rapid prototyping and potentially high-throughput manufacturing, laser cutting and die cutting of thin polymer films have emerged as viable alternatives. These techniques can process multilayer stacks of materials in a roll-to-roll manner, enabling the fast and cost-effective production of complex microchannel and assay well networks[76]. When working with fibrous materials, electrospinning is employed to create flexible, porous substrates such as thermoplastic TPU nanofibrous mats. This process involves ejecting a polymer solution under a high-voltage electric field to deposit fine fibers on a collector, which can subsequently be functionalized (e.g., with SERS-active gold coatings) for sensing applications[70]. For substrates such as paper, laser cutting based on pre-designed patterns allows for the precise shaping of microfluidic channels[67], while dip coating is used to uniformly modify these porous matrices with functional nanomaterials, such as adsorbing gold nanorods (Au NRs) onto chromatography paper for SERS sensing[67]. Furthermore, techniques such as hydrogel photo-crosslinking under ultraviolet (UV) light enable the formation of tough, adhesive, and sensor-encapsulating hydrogel matrices from precursor solutions[55], and solid-state dewetting of metal films on flexible supports facilitates the creation of large-area, uniform plasmonic nanostructures for SERS[71]. The integration of biochemical recognition elements often relies on enzyme immobilization or immunoassay assembly protocols on these fabricated platforms[77,78]. For more complex integration, additive manufacturing techniques such as 3D printing are utilized to produce custom components such as smartphone-based imaging modules housing light sources and filters[79]. These diverse fabrication strategies collectively underpin the development of the multi-analyte detection platforms summarized in Table 1, enabling the transition from laboratory concepts to wearable devices.
Adhesion and encapsulation
The successful translation of these fabricated components into a robust wearable device requires effective strategies for skin adhesion and system encapsulation. Robust skin adhesion and effective system encapsulation are paramount for ensuring stable operation, reliable data acquisition, and user comfort in wearable optical sweat sensors[80]. The choice of form factor and adhesion strategy is often application-dependent; for medical uses, patch-style formats that discreetly adhere to the skin are preferable[3]. A common design for such epidermal devices involves a multi-layer structure, where a functional layer for sensing and fluidics is integrated with a dermal contact layer for skin adhesion, frequently utilizing a medical adhesive with a sweat collection port to ensure stable conformal contact[71]. Wearable flexible microfluidic platforms use low-modulus elastomeric (e.g., PDMS) and biocompatible materials and hypoallergenic silicone adhesives to fabricate a watertight and strong interface for sweat collection[9]. The general approach to achieving a strong packing around the skin sweat capture region, essential for accurate sampling, is using a robust seal to prevent leakage and contamination[45,81]. A key issue is ensuring secure yet comfortable adhesion, particularly for users with sensitive skin. Hydrogel-based adhesives, offering biocompatibility and breathability, provide a promising solution by enabling firm yet gentle adhesion that adapts to skin movements[15]. Furthermore, a robust skin adhesion can elevate signal reliability, but excessive adhesion makes removal difficult and causes skin irritation. Recently, some researchers reported dry adhesive electrodes, aiming to realize a reversible and robust adhesion on skin for sweat collection[46].
Beyond skin-device attachment, effective encapsulation is critical for maintaining integrity of the internal sensing environment and managing mechanical stresses. A significant challenge in flexible sensors is the interfacial instability, where strain and/or stress concentration occurs at soft-hard interfaces, resulting in a main cause of failure via detachment/delamination[82]. Generally, the principles in solving soft-hard instability are avoiding abrupt hardness/softness differences and improving interfacial adhesion[82]. For systems that incorporate less stretchable functional components, a widespread design technique is to combine them in “islands” electrically and mechanically interconnected by stretchable conductors, thereby achieving overall system stretchability while protecting critical elements[75]. The encapsulation of microfluidic and optical sensing components themselves is often realized through precise multilayer assembly and lamination. This can involve fabricating structures by stacking and laser-cutting layers of medical adhesives, with the assembly completed by aligning and attaching polymer films (e.g., PET) using additional adhesive layers to form sealed, functional devices[71,81]. To achieve long-term and continuous sensing, a well-sealed detection chamber has been implemented by combining flexible plastic substrates (e.g., PET) with soft microfluidics (e.g., PDMS). The chamber effectively harvests sweat, reducing sample and contamination and evaporation[45]. Even with the mitigating effects of highly multiplexed sensors and nuanced designs, developing robust encapsulation materials and packaging to protect against corrosion, moisture, and other noise factors remains important for reliable optical sensing, particularly for ultralow concentration species[9]. The resulting mechanical robustness and flexibility of the fully encapsulated device ensure conformal dermal contact and enable it to securely preserve sweat under twisting, detachment, compression, and other physical stresses, thereby validating the overall reliability of the integrated system for real-world use[71].
OPTICAL SENSORS FOR DETECTION OF SWEAT ANALYTES
A diverse range of flexible, conformable, and stretchable sensors have been fabricated for the noninvasive analysis of sweat analyte variations. With the development of sweat analysis technologies, optical sensing has emerged as an attractive alternative. Unlike electrochemical sensing, which requires complicated circuitry, precise electronics, and external power sources, optical systems offer a simpler and more compact approach. The analysis of sweat and the sensing performance of the various optical sensors are shown in Table 1. The strategy is based on the conversion of sweat chemical signals into optical signals via chromogenic, fluorescent, or other responsive molecules, allowing for direct visual interpretation or readout with portable equipment. The following sections outline four major optical methods: colorimetry, SERS, fluorescence, and ECL [Figure 3].
Figure 3. Schematic illustration of optical sensing mechanisms for wearable sweat analysis. (A) Colorimetric detection based on chromophore–biomarker interaction, where the resulting color change is analyzed via optical readout and digital image processing (e.g., RGB analysis); (B) SERS detection, in which laser excitation of plasmonic nanoparticles generates an enhanced near‑field that amplifies Raman scattering from adsorbed analyte molecules, enabling fingerprint‑specific signal acquisition; (C) Fluorescence detection: Target biomarkers interact with fluorescent probes under specific light excitation to modulate emission characteristics, enabling sensitive quantitative readout; (D) ECL detection: An applied potential triggers high-energy electron-transfer reactions at the electrode surface, generating excited-state luminophores that emit light upon relaxation, thereby providing a high-sensitivity signal without the need for external optical excitation[83]. Created in BioRender. Chu Wang. (2026) https://BioRender.com/tfxxgjh, Adobe Illustrator, and Maxon Cinema 4D R21. RGB: Red-green-blue; SERS: surface-enhanced Raman scattering; ECL: electrochemiluminescence.
Colorimetric sensors
Colorimetric sensors function by generating visible color changes through enzymatic or chemical reactions, in which chromophore molecules interact with target biomarkers to change the absorption of light, producing a color change in proportion to analyte concentration[84-87]. To systematically assess this modality in the context of wearable sweat sensing, we evaluate it across several critical dimensions: its sensing mechanism and operational simplicity, sweat collection and delivery requirements, on-body signal robustness, quantitative performance, and suitability for real-time continuous monitoring. This method offers significant advantages for integrating wearables, especially by eliminating complex electronics and allowing passive, flexible and inexpensive substrates such as paper, textiles or polymers[87-89]. In terms of performance, they offer good sensitivity for major sweat metabolites and electrolytes [Table 1]; however, their quantitative accuracy is often semi-quantitative and can be compromised by uneven color development, intrinsic chroma error, and a dependence of signal intensity on both analyte concentration and sample volume[77,86,88]. For reliable on-body operation, effective sweat delivery via integrated microfluidics is often crucial to control reagent mixing and enable sequential analysis[87,89]. However, a major limitation is that chemical reactions are usually endpoint-based, which means that current wearable colorimetric systems do not allow for real-time and continuous monitoring of dynamic concentration changes[50,90]. Moreover, the compatibility of these sensors with everyday devices such as smartphones for digital imaging enhances their capability for simple, rapid in-situ analysis of sweat constituents [Figure 3A]. Data analysis can range from semi-quantitative visual comparisons to sophisticated smartphone-based digital image processing techniques, including RGB (red-green-blue)/HSB (Hue, Saturation, Brightness) extraction or the use of CIE 1976 Lab color space (CIELAB) color space and deep learning algorithms to enhance accuracy and mitigate environmental variability[50,86,87,91].
A skin-attachable paper fluidic sensor patch has been developed by printing microfluidic patterns with ceramic-based ink on cellulose paper. The patch is equipped with detection zones modified by nanoparticles for colorimetric detection of sweat analytes and for simultaneous monitoring of the sweat rate [Figure 4A][87]. The ceramic ink creates impervious, thermally stable barriers to prevent leakage, while amine-functionalized nanoparticles enhance color homogeneity and sensor sensitivity. The device employs specific enzymatic reactions for glucose, lactate, and uric acid detection, along with a pH-sensitive dye, and the color changes are analyzed through smartphone imaging in HSB and RGB color spaces. This work establishes a scalable, low-cost, and robust paper-based platform that integrates multiplexed colorimetric sensing with sweat rate measurement, providing a practical and disposable solution for wearable non-invasive health monitoring. Similarly focused on basic sensing elements, but moving towards a more complex soft form factor, Kim et al. used PDMS doped with white pigment and specific colorimetric reagents to fabricate a soft, skin-interfaced microfluidic platform for the colorimetric detection of nutrients in sweat, enabling non-invasive nutritional monitoring and transdermal nutrient delivery [Figure 4B][89]. The device features deep microreservoirs to enhance optical path length and sensitivity, and integrated passive CBVs to direct sweat flow and prevent cross-contamination. The quantitative analysis was achieved via smartphone RGB imaging, validated against laboratory methods. A key innovation is the demonstration of a simple, electronics-free, and cost-effective colorimetric platform for multiplexed nutrient sensing, highlighting the potential of sweat-based microfluidics for personalized nutrition management and at-home health monitoring.
Figure 4. (A) Schematic layout of the wearable colorimetric sensing patch and depiction of sweat flow directionality and relative flow rates within the patch. Reproduced with permission[87]. Copyright 2024, Wiley-VCH; (B) Schematic illustration of the microfluidic sensor and nutrient patch in an exploded configuration: the microchamber architecture for colorimetric analysis, the handheld device photograph, and the optical image of the assembled system situated next to a vitamin tablet. Reproduced with permission[89]. Copyright 2024, Wiley; (C) Exploded view of a skin-interfacing microfluidic system for personalized sweat-rate and sweat-chloride analysis: patterned microfluidic layer and embedded sensing layer. Reproduced with permission[76]. Copyright 2020, Science; (D) Overall workflow of the deep-learning-assisted programmable chromatic chip for evaluating sweat glucose, pH, and lactate, including an intelligent imaging device and interpretable convolutional neural network. Reproduced with permission[86]. Copyright 2022, American Chemical Society.
Building on these basic sensing platforms, considerable efforts have been made to facilitate complex physiological monitoring in the real world and in ambulatory settings. For example, Baker et al. employed multilayer thin-film polymers to develop a flexible, skin-interfaced wearable microfluidic system for the real-time monitoring of regional sweating rate and chloride content in sweat in sports science applications [Figure 4C][76]. The device consisted of two primary microchannels: Microchannel 1 incorporated an orange dye that dissolved upon sweat entry for volumetric measurement, while Microchannel 2 was pre-loaded with a chloride-sensitive assay which produced a purple color with intensity proportional to sweat chloride concentration. A smartphone application captured images for automatic analysis. This work represents an innovation by seamlessly integrating a low-cost, manufacturable microfluidic platform with ubiquitous smartphone capabilities for in-situ colorimetric analysis. It successfully transitions sweat analytics from laboratory environments to real-world applications and serves as a foundational model for field-deployable optical sweat sensors. Advancing beyond traditional colorimetric methods, the incorporation of artificial intelligence (AI) has opened new avenues for highly accurate and automated data interpretation. Liu et al. utilized sodium alginate-calcium chloride gel capsules immobilized with various enzymatic/chemical indicators to fabricate a programmable colorimetric sensor, which was coupled with a deep learning algorithm designed for the precise distinguishing and quantification of pH levels, glucose, and lactate in human sweat [Figure 4D][86]. The gel capsules functioned as programmable sensing pixels containing specific reagents that induced distinct color changes. A convolutional neural network (CNN) model was trained on an extensive dataset of colorimetric images, achieving 100% accuracy and a Class Activation Mapping (CAM) analysis was applied to demonstrate the inner working mechanism of CNN operation[86]. By integrating the customizable colorimetric platform with the interpretable deep learning framework, this end-to-end strategy becomes an example of AI to overcome the limitations of traditional technologies and provides an important theoretical reference for future sensor design.
The development of colorimetric sensing is further illustrated by its innovative integration with different sensing modes and readout mechanisms to create a multi-functional system. For instance, a wireless, skin-interfaced platform was developed by integrating a thermal flow sensor with a straight PDMS fluid passage and a Bluetooth Low Energy system-on-chip, for real-time analysis of sweat flow rate and cumulative loss. This platform allows for optional integration into advanced microfluidic systems designed for the colorimetric sensing of pH, chloride, creatinine, and glucose[92]. Operating on a non-contact thermal sensing principle, the platform was engineered to be reversibly integrated with a microfluidic module containing reservoirs pre-loaded with colorimetric assays for sequential analysis. By integrating wireless digital readout of sweat parameters and advanced colorimetric chemistry into one device, this multifunctional, multimodal platform signifies a major advance in the development of fully integrated “lab-on-skin” systems.
While colorimetric sensors offer significant advantages such as suitability, low cost, and simplicity for in situ and multiplexed analyses, they face several challenges. These include the inability to perform real-time continuous monitoring, a typically semi-quantitative nature, susceptibility to environmental interference, and limitations in accuracy due to factors such as uneven color development and signal dependence on both concentration and sample volume[50,60,77,86,88]. Colorimetric sensing provides a simple, low-cost, and electronics-free pathway for wearable sweat analysis, enabling immediate perceptibility and user-friendly operation[93]. However, compared to more complex optical techniques such as fluorescence or SERS, it trades some quantitative accuracy and temporal resolution for unmatched simplicity and cost-effectiveness. This makes it particularly ideal for disposable, single-use patches aimed at periodic health screening or personalized hydration and nutrition management, where continuous data streaming is not the primary requirement[76,85,89].
SERS seznsors
SERS technology offers higher specificity and multiplexing capabilities by acquiring the unique fingerprint information of molecules. SERS utilizes the localized surface plasmon resonance (LSPR) effect on the surface of noble metal particles and the gaps between them to enhance the Raman scattering signal of adsorbed molecules by approximately 6 to 9 orders of magnitude, even down to the single-molecule level[67,70,94]. Compared to the colorimetric method, SERS provides a direct molecular identification without the need for chemical markers or enzymatic reactions and is less likely to interfere with complex sweat matrices[94,95]. The basic principle of SERS relies on the enhancement of the Raman signal, generated by specific molecular vibrational/rotational modes through the high local electric field produced by LSPR. This enables label-free and highly specific identification of target analytes [Figure 3B][65,67,96]. The technique offers advantages such as ultra-high sensitivity, resistance to photobleaching, minimal interference from water, and the ability to analyze multiple components simultaneously in a single measurement[55,94,95,97]. With the development of flexible substrates and portable Raman spectrometers, SERS is now being incorporated into wearable devices for noninvasive, real-time analysis of biofluids[95,98]. When evaluated within the comparative framework established for colorimetry, SERS reveals distinct trade-offs: it offers higher specificity and multiplexing capability, but its on-board reliability is questioned due to the need for a uniform and mechanically stable plasmonic substrate, and its integration often relies on external optical equipment. For effective wearable integration, SERS substrates must not only provide dense and uniform plasmonic hotspots for signal enhancement but also maintain mechanical compliance and stability under skin deformation[71,94]. This is often achieved through flexible materials such as paper, textiles, and nanostructured polymers, which can conform to skin curvature and withstand repeated bending[66,70]. Furthermore, efficient sweat collection and delivery are essential for reliable SERS-based sensing. Microfluidic architectures are frequently incorporated to enable controlled fluid transport, prevent cross-contamination between samples, and facilitate sequential or time-resolved sampling[67,99]. Nevertheless, the realization of robust in vivo SERS sensing continues to face significant challenges, including the scalable fabrication of homogeneous nanostructures, preservation of signal fidelity under mechanical strain, and mitigation of interference from the complex biochemical composition of sweat[70,99]. Based on these fundamental considerations, innovative designs have been developed to translate SERS into practical wearable formats.
Building on these intrinsic advantages, SERS has been effectively adapted into immunoassays for sensitive biomarker detection. For example, a SERS-based immunoassay (SERSI) was developed to analyze cortisol in biological fluids using Au NRs as SERS nanotags and gold-coated glass slides or magnetic beads as capture substrates functionalized with cortisol antibodies [Figure 5A][78]. In the magnetically-assisted SERSI modality, competition between free cortisol and SERS nanotags for binding sites on antibody-decorated magnetic beads resulted in an increase in the SERS signal in the supernatant, which is proportional to cortisol concentration. This enabled quantification across a linear range of 12.5-400 ng/mL, with a detection limit (LOD) of 7 ng/mL. This method demonstrated high selectivity against structurally similar hormones, excellent repeatability, and was successfully validated against standard techniques. This work presents a highly reliable and sensitive SERS immunoassay platform, highlighting its potential as an effective platform for in situ and real-time detection of cortisol-related disorders and human stress levels, and establishing a crucial methodological foundation for subsequent SERS biosensing development. To transition toward wearable applications, research has focused on integrating SERS with flexible, low-cost substrates. Mogera et al. employed Au NRs immobilized on chromatography paper integrated with a flexible microfluidic system and SERS to fabricate a wearable sensor for continuous, label-free quantification of uric acid in sweat [Figure 5B][67]. The paper microfluidic layer enabled efficient sweat collection and transport via capillary action while simultaneously quantifying sweat loss and sweat rate, and the gold nanostars served as stable plasmonic substrates that provided significant SERS enhancement for highly sensitive (LOD: 1 µM) and specific uric acid detection using a ratiometric approach. This work innovatively combines label-free SERS detection with paper-based microfluidics in a flexible and stretchable wearable format, effectively addressing the environmental instability of traditional enzymatic/antibody-based sensors and demonstrating considerable potential for advancing reliable, continuous metabolite monitoring in point-of-care health applications.
Figure 5. (A) Illustration of two SERS immunoassay approaches: magnetic-bead suspensions bearing cortisol antibodies for magnetically assisted detection, and gold-coated glass substrates functionalized with cortisol antibodies for rigid-support detection. Reproduced with permission[78]. Copyright 2020, Elsevier; (B) Conceptual, overhead, and stacked representations of the wearable plasmonic paper microfluidic system for sweat collection, storage, and in situ SERS measurement. Reproduced with permission[67]. Copyright 2022, Science; (C) Schematic illustration depicting the fabrication process of the Au NSs/ion-track-etched polycarbonate membrane-based SERS substrate and its subsequent detection procedure. Reproduced with permission[94]. Copyright 2023, American Chemical Society; (D) Schematic diagram of the stacked structure of functional layers, an integrated microfluidic plasmonic device enabling biofluid flow, storage and SERS analysis, alongside a system-level block diagram of the internal functional modules within the portable Raman analyzer. Reproduced with permission[99]. Copyright 2022, Nature Publishing Group; (E) Schematic illustration of the fabrication and initiation of the flexible nanoplasmonic SERS patch comprising plasmonic fluid-channel layer and dermal-contact layer. Reproduced with permission[71]. Copyright 2025, Nature Publishing Group; (F) Schematic diagram of the sweat collection principle of Janus-based wearable textiles and the generation of inverse Raman signals by microstructured optical fiber cores. Reproduced with permission[66]. Copyright 2023, American Chemical Society. 3D: 3-dimensional; SERS: surface-enhanced Raman scattering; PDMS: polydimethylsiloxane.
In addition to disposable paper-based systems, enhancing the performance and reusability of SERS substrates has emerged as a key focus area for sustainable wearable sensing technologies. Gui et al. fabricated a flexible and nanoporous SERS substrate by synthesizing Au NRs in situ on an ion-track-etched polycarbonate (PC) membrane aimed at facilitating sweat enrichment and metabolite analysis [Figure 5C][94]. The nanoporous structure of the PC membrane enabled efficient analyte enrichment, while the Au NRs provided dense and uniform plasmonic hotspots for significant Raman signal enhancement. The substrate showed highly reproducible SERS signals for methylene blue, along with excellent mechanical flexibility (withstanding 2,000 bending cycles) and long-term stability. This work showcases the integration of high-performance SERS substrates with flexible porous membranes, providing a material design strategy for achieving stable and reproducible non-invasive sweat monitoring. The evolution of SERS sensing further extends to sophisticated system integration with microfluidics and intelligent data processing for dynamic and multi-parameter monitoring. For example, He et al. used Ag nanomushroom arrays integrated into a PDMS microfluidic platform to fabricate a wearable SERS sensing device for portable and refreshable detection of sweat analytes such as pH, lactate, and urea [Figure 5D][99]. This work integrated epidermal microfluidics with portable SERS analysis, offering a reusable, high-resolution sensing platform for personalized healthcare, and highlights the potential of microfluidic SERS systems for continuous, non-invasive sweat analysis.
Advancing beyond traditional sensing paradigms, the incorporation of chronological sampling and machine learning has opened new avenues for intelligent metabolic monitoring. Jeon et al. developed an all-flexible chronoepifluidic nanoplasmonic patch that integrated plasmonic nanostructures within a microfluidic sequential sampler for label-free and chronological analysis of sweat biomarkers [Figure 5E][71]. The sensor featured plasmonic Ag nanoislands formed on an ultrathin fluorocarbon-coated PDMS membrane via low-temperature solid-state dewetting, creating large-area, uniform electromagnetic hotspots. The microfluidic sampler utilized CBVs to collect and spatially separate sweat samples over time. Combined with machine learning, the patch enabled the quantitative analysis of lactate, uric acid, and tyrosine, and successfully captured dynamic metabolic changes induced by exercise and dietary intake. This work represents a significant advancement in manufacturing high-performance nanostructures on flexible substrates and provides new ideas for addressing signal stability issues in continuous, long-term monitoring using wearable SERS sensors. Innovation in SERS wearable technology also extends to novel architectures that decouple fluid handling from detection. Han et al. combined a Janus textile for unidirectional sweat collection with a grapefruit microstructured optical fiber decorated with Ag nanoparticles as a SERS probe for sweat analysis [Figure 5F][66]. The Janus textile features a superhydrophobic skin side and patterned superhydrophilic zones for directional sweat transport, while the optical fiber extracts nanoliter sweat volumes via capillary action and provides length-distributed plasmonic hotspots for SERS enhancement. The system detected urea and sodium lactate at very low concentrations with high uniformity and multiplexing capability, illustrating a creative merger of textile-based microfluidics and optical SERS for practical wearable operation.
Despite the successful integration of SERS with flexible platforms, there are still challenges in translating this technology into robust and widely applicable wearable systems. The main obstacle is the difficulty of producing scalable SERS substrates with dense, homogeneous and mechanically stable plasmonic hotspots[71,94]. Signal reproducibility can be compromised by the oxidation of silver-based nanomaterials, variations in nanoparticle spacing under skin-induced strain, and interference from competing molecules present in sweat[66,70,99]. For system integration, SERS sensing requires external optical excitation and spectroscopic analysis. The current reliance on portable Raman spectrometers imposes constraints in terms of device cost, overall size and easy integration in a separate, low-profile patch[95,98]. In addition, detection efficiency may be limited in individuals with low sweating capacity and long-term considerations regarding the biocompatibility of nanomaterials and laser safety require careful consideration[88,97]. Thus, SERS is particularly suited for applications demanding high-specificity, multi-analyte profiling, such as metabolic monitoring, hormone detection and drug screening, although its wide use in wearable formats is dependent on progress in portable spectroscopy and the stability of substrates[95,98].
Fluorescence sensors
Fluorescence-based sensing is another powerful optical method for wearable sweat analysis, being distinguished by its high sensitivity, suitability for quantitative and multiplexed detection, and inherent compatibility with flexible, skin-interfaced platforms[100,101]. The basic principle is to detect fluorescence signal changes, such as intensity, emission wavelength or lifetime, emitted by chemical probes when they are activated by light and when they interact with target analytes[100,102]. Common fluorescence detection mechanisms include fluorescence quenching (e.g., dynamic quenching of quinine sulfate by chloride ions)[79], fluorescence enhancement (e.g., sodium binding to crown ether derivatives)[102], wavelength shifts, and changes in fluorescence lifetime[79,102]. Energy transfer mechanisms such as Förster resonance energy transfer (FRET) and the inner filter effect (IFE), as well as enzyme-coupled reactions (e.g., glucose sensing via H2O2-mediated quenching of fluorescein)[79], are also used to achieve high specificity and multiplexing capability for metabolites such as glucose, lactate, chloride, and pH[54,79]. Compared to colorimetric approaches, fluorometric approaches offer higher intrinsic sensitivity for multiplexing, which results in lower LODs and broader linear ranges for different analytes [Table 1][79,100].
A prime example of this integrated approach is the smart wearable sweat patch developed by Ardalan et al., which utilized cellulose-based paper discs and cotton-thread microfluidics for the fluorimetric sensing of glucose, lactate, pH, chloride, and sweat volume [Figure 6A][79]. Each biomarker was measured via a distinct fluorescent mechanism, and a custom 3D-printed smartphone module with a UV-LED and optical filter captured the signals for quantification via a dedicated app. This work demonstrated a low-cost (~ $0.03 per patch), disposable platform, underscoring the potential of combining simple microfluidics with smartphone fluorimetry for accessible multi-analyte sensing. Extending the concept of multiplexed ion detection, Sekine et al. integrated specific fluorescent probes for chloride, sodium, and zinc into a soft microfluidic device for sequential sweat analysis during exercise [Figure 6B][102]. The device used CBVs to direct sweat into pre-loaded assay reservoirs. A smartphone attachment with appropriate optical filters enabled fluorescence imaging, with signals normalized to an internal fluorescent reference (Rhodamine 110). This system achieved accuracy comparable to standard laboratory techniques in human studies, establishing a practical, low-cost platform for fluorometric sweat ion analysis.
Figure 6. (A) Digital photograph of the IoT-integrated cellulose microfluidic fluorescent patch constructed from medical tape, cotton thread, and paper. Reproduced with permission[79]. Copyright 2020, Elsevier; (B) Schematic of a microfluidic platform with in situ fluorescence analysis: collection and retention of chloride, sodium, and zinc with corresponding chemical-probe fluorescence signals. Reproduced with permission[102]. Copyright 2018, The Royal Society of Chemistry; (C) Schematic representation of a soft microfluidic device for fluorescent measurement of amino acid concentrations in sweat via enzymatic reactions. Reproduced with permission[103]. Copyright 2025, The Royal Society of Chemistry; (D) Schematic illustration of the fabrication of a wearable hydrogel sensing patch embedded with upconversion optical probes and its multichromatic response to urea by inner-filter effect. Reproduced with permission[54]. Copyright 2023, American Chemical Society; (E) Schematic representation of the process of luminescent silicon-loaded carbon quantum dots and bimetallic nanoparticles for red-to-blue fluorescence detection of glucose in sweat. Reproduced with permission[104]. Copyright 2020, American Chemical Society; (F) Schematic diagram of the principle of Tb-DPA MOF-based detection of human sweat pH. Reproduced with permission[105]. Copyright 2024, Elsevier. CQD: Carbon quantum dot; DPA: 2,6-pyridinedicarboxylic acid; IoT: internet of things; MOF: metal-organic framework.
In addition to ions, fluorescence is particularly effective for monitoring metabolites via enzymatic pathways. Cho et al. employed smartphone-linked enzymatic fluorometric assays (for total amino acid, alanine, and lysine) incorporated into a skin-interfaced, flexible microfluidic patch, to investigate amino acid loss in eccrine sweat during exercise [Figure 6C][103]. The device featured grouped microreservoirs filled via CBVs for sequential sampling. The assays, based on coupled enzymatic reactions producing a fluorescent signal (excitation/emission: 535/587 nm), exhibited high sensitivity, low LODs, and linear responses across physiological ranges. This system provided a cost-effective, non-invasive method for real-time monitoring of amino acid turnover, emphasizing sweat’s role in metabolite loss and the importance of dietary management during prolonged exercise. In a different optical design focused on a single metabolite, Hu et al. developed a wearable hydrogel patch embedding an UCNPs functionalized with p-DMAC for urea sensing based on the inner inner filter effect (IFE) [Figure 6D][54]. The reaction between urea and p-DMAC under acidic conditions generated a product that absorbed the green emission of UCNPs, causing a ratiometric color shift from yellow to red under 980 nm near infrared (NIR) excitation. This NIR excitation strategy minimized background autofluorescence from skin and ambient light. A portable 3D-printed reader integrating a smartphone, a miniaturized laser, and an optical filter enabled in situ quantitative analysis, demonstrating a robust approach for anti-interference monitoring in complex environments.
The pursuit of higher accuracy has driven the development of advanced ratiometric fluorescence systems. For example, Cui et al. fabricated a wearable skin pad using luminescent porous silicon (PSi) particles loaded with carbon quantum dots (CQDs) and decorated with bimetallic (Au/Ag) nanoparticles (BiM), co-immobilized with glucose oxidase (GOX) in a chitosan film on a PU membrane, for the noninvasive and real-time visual detection of sweat glucose [Figure 6E][104]. In this ratiometric fluorescent nanohybrid (BiM-CQDs@PSi), the PSi acts as a red-emissive, oxidizable carrier, CQDs serve as the blue-emissive donor, and BiM enhances both the initial fluorescence of PSi and catalyzes its H2O2-triggered oxidation. The working principle relies on an initial FRET from CQDs to PSi, quenching blue emission and showing red fluorescence. Upon contact with sweat glucose, GOX-generated H2O2 oxidizes PSi, weakening FRET, decaying red fluorescence, and recovering blue emission, resulting in a visible red-to-blue color shift. The transition kinetics are proportional to glucose concentration, enabling semiquantitative analysis via smartphone imaging and RGB analysis, which was successfully demonstrated in clinical tests to distinguish diabetic hyperglycemia from normal levels. Similarly, for pH monitoring, Chen et al. developed a skin-attachable ratiometric sensor using a dual-emissive Tb-based metal-organic framework (Tb-DPA MOF, where DPA = 2,6-pyridinedicarboxylic acid)[105]. The H+-induced collapse of the MOF structure differentially affected the emissions of the ligand (DPA at 380 nm) and the Tb3+ center (544 nm), providing a stable and reversible fluorescence response across the physiologically relevant sweat pH range (3.50-6.70). The sensor demonstrated excellent long-term stability and was successfully used for real-time sweat pH monitoring during exercise.
Fluorescence sensing occupies a distinct niche in the wearable optical sensing landscape. However, translating fluorescence into robust, widely deployable wearables faces challenges, including the inability for real-time continuous monitoring in some endpoint-based designs[79], potential temperature sensitivity affecting enzyme activity or assay stability[79,103], and limited detection sensitivity for certain analytes such as zinc[102]. Signal integrity can be compromised by photobleaching risks requiring light-shielding measures[102], as well as interference from autofluorescence and background signals, especially for short-wavelength-excited probes[100]. Furthermore, the irreversible and non-reusable nature of most assays necessitates single-use formats[103]. Fluorescence is particularly well-suited for applications demanding sensitive, quantitative, and multiplexed tracking of key metabolites and electrolytes, especially when integrated with advanced strategies such as NIR excitation to minimize background noise[54]. Future advancements focusing on improved stability, interference resistance, and continuous monitoring capability will be key to developing more robust fluorescence-based wearable systems.
ECL sensors
ECL is emerging as a promising but nascent optical sensing technique that uniquely combines electrochemical and luminescent processes for highly sensitive bio-sensing applications. Unlike other optical methods, ECL operates without an external light source and offers intrinsic low background, high sensitivity, and precise spatiotemporal control of light emission[106]. The basic principle of ECL involves the generation of excited-state luminophores by exergonic electron transfer reactions at the surface of the electrode, which then relax and emit photons [Figure 7A][107]. This process can be initiated through various mechanisms. One commonly used system involves coreactant pathways, such as the classic tris(2,2′-bipyridine)ruthenium(II) ([Ru(bpy)3]2+)/trialkylamine pair, in which amine-containing analytes participate in a redox cycle to regenerate the excited state of [Ru(bpy)3]2+. Another common approach is the enzyme-coupled ECL, where enzymatic reactions generate coreactants such as nicotinamide adenine dinucleotide (NADH) or H2O2 that interact with ECL luminophores (e.g., [Ru(bpy)3]2+ or luminol) to produce light[108,109]. From a performance point of view, ECL presents several compelling advantages including high sensitivity (e.g., LODs down to pM levels for lactate) [Table 1], wide dynamic range, and minimal interference from ambient light due to its self-emissive nature[106,107,110].
Figure 7. (A) Schematic diagrams of a stainless steel electrode for ECL detection using the luminol/H2O2 system and the mechanism of photoinduced ECL at silicon electrodes protected by nanoscale SiOX and Ni-stabilizing thin films. Reproduced with permission[107]. Copyright 2021, American Chemical Society; (B) Schematic illustration of the Ru-PEI@SiO2 synthesis and the assembly route of the flexible MIP-ECL skin sensor: electrochemical conversion, surface immobilization, MIP layer imprinting, sensor elution, and subsequent detection of epidermal analytes. Reproduced with permission[106]. Copyright 2019, The Royal Society of Chemistry; (C) Schematic diagram of neurotransmitter monitoring using a stretchable photonic-crystal-hydrogel-enhanced ECL platform, together with machine learning-assisted detection and classification. Reproduced with permission[108]. Copyright 2025, American Chemical Society; (D) Schematic depiction of rGO@hydrogel used for monitoring motion behavior and evaluating multiple physiological indicators in sweat, including urea, lactate, and chloride. Reproduced with permission[111]. Copyright 2023, Elsevier. ECL: Electrochemiluminescence; TEOS: tetraethyl orthosilicate; PAM: polyacrylamide; PVA: polyvinyl alcohol; O-β-CD: oxidized β-cyclodextrin; c-BPE: closed bipolar electrode.
Integrating ECL into wearable devices demands flexible and robust architectures that can maintain electrical and optical performance under deformation. Pioneering designs have focused on merging ECL-active components with soft, stretchable materials such as flexible conductive substrates (e.g., Au nanotube networks, carbon textiles) functionalized with ECL luminophores[106]. Hydrogels are particularly attractive as they can serve dual roles as sweat-absorbing reservoirs and ECL electrode matrices[111]. ECL presents several compelling advantages for wearable sweat analysis. It combines the high sensitivity, wide dynamic range, and ease of multiplexed analysis inherent to chemiluminescence with the good stability and tunability of electrochemical methods[107]. Crucially, its operation without an external light source eliminates ambient light interference and simplifies device architecture. Compared to traditional electrochemical sensors, ECL offers superior sensitivity for non-electroactive analytes and minimizes interference from electroactive species in complex biofluids such as sweat[106]. These attributes, coupled with the potential for simple, low-cost instrumentation, make ECL a promising candidate for next-generation wearable diagnostics[110,112].
A pioneering example is the flexible ECL platform developed by Chen et al. for wearable sweat analysis [Figure 7B][106]. They immobilized highly luminescent ruthenium-polyethyleneimine (Ru-PEI@SiO2) nanospheres on stretchable gold nanotube networks, followed by a coating of an elastic molecularly imprinted polymer (MIP) for effective detection. This design enabled in situ sampling and quantification of urea and lactate in human sweat with high selectivity and excellent mechanical stability, achieving LODs of 15.0 μM and 16.7 μM, respectively. This study translated the ECL mechanism into a practical wearable format, establishing a novel paradigm for non-invasive metabolic sensing. Advancing the integration of ECL with flexible optics, other researchers have developed composite hydrogel electrodes for enhanced performance. Chen et al. designed an ECL electrode using a stretchable photonic-crystal hydrogel (PCH), which synergistically combined a polyacrylamide (PAM-PVA)-(O-β-CD)-Gel conductive hydrogel with a poly latex-sphere photonic crystal nanomatrix for enhanced neurotransmitter detection in sweat [Figure 7C][108]. The mechanochromic PCH allowed dynamic tuning of its photonic bandgap under strain to match the emission wavelength of the [Ru(bpy)3]2+ luminophore, resulting in a signal amplification of up to 9-fold. This platform achieved simultaneous qualitative and quantitative analysis of 11 different neurotransmitters with 100% classification accuracy in artificial sweat and serum, leveraging linear discriminant analysis of the ECL responses. This work represents a significant innovation by merging stretchable optics with ECL sensing, creating a versatile and powerful platform for multiplexed biomarker detection in wearable healthcare applications.
Beyond metabolite sensing, ECL platforms are being engineered to combine biochemical analysis with physical sensing for holistic health monitoring. Hu et al. utilized rGO- and luminol (Lum)-decorated self-healing hydrogels to construct a sweat sensor incorporating a closed bipolar electrode (c-BPE) sweat detection unit and a strain sensing unit for simultaneous motion monitoring and multiplexed sweat biomarker analysis such as chloride ions, lactic acid, and urea [Figure 7D][111]. In this platform, the rGO@hydrogel serves as a strain sensor with excellent stretchability (up to 400%), adhesiveness to skin, and stable electrical responses to body movements, while its porous 3D structure enables in-situ sweat collection and storage; subsequently, it spontaneously self-heals with Lum@hydrogel to form a c-BPE system, where the rGO@hydrogel (cathode) containing sweat analytes and the Lum@hydrogel (anode) enable ECL detection through anode signal fluctuations correlated to cathode reactions, achieving accurate quantification without direct sample-reagent contact. The platform demonstrated rapid response to physical activities, high stability, prolonged luminescence duration, and reliable on-body detection of sweat components, validated against commercial kits. Complementing these integrated systems, enzymatic ECL biosensors highlight the technique’s potential for extreme sensitivity. Cai et al. developed such a biosensor by co-immobilizing pyruvate oxidase (PYOD) and dehydrogenase (LDH) with their cofactor nicotinamide adenine dinucleotide (NAD+) on a carbon nanotube-modified electrode, using luminol as the emitter[110]. The enzymatic oxidation of lactate produced hydrogen peroxide, which strongly enhanced the luminol ECL signal. This coupling yielded an ultra-sensitive lactate sensor with a remarkably wide linear range and a LOD down to 8.9 × 10-12 mol/L, successfully applied to track lactate dynamics in athlete sweat during exercise.
ECL faces several challenges that need to be addressed to achieve widespread adoption of the wearable. The main obstacles are the instability of luminescence on flexible substrates and the incompatibility of traditional rigid electrodes with conforming skin contact[108]. High operating voltages in some systems raise concerns regarding power consumption and on-body safety[113]. The integration of multiple functional units (e.g., sensing, fluidics, and electronics) on a single flexible substrate often increases system complexity and can degrade overall performance[111]. In addition, ECL signals can be susceptible to interference from co-existing species in real sweat samples[110].
Other optical sensors
In addition to colorimetric, SERS, fluorescence, and ECL methods, optical fiber-based sensing represents a distinct and promising approach for wearable sweat analysis, operating on fundamentally different principles. The core mechanism relies on light guidance via total internal reflection within the optical fiber, where a portion of the electromagnetic field extends as an evanescent wave at the core-cladding interface. By modifying the fiber through tapering, side-polishing, or cladding removal, this evanescent field can be exposed to interact with the surrounding sweat. Changes in the local refractive index due to biomarkers modulate the guided light’s characteristics (intensity, wavelength, phase), enabling sensitive detection[114,115]. Diverse sensing architectures are built on this foundation, including Mach-Zehnder interferometers, surface plasmon resonance structures, and Fiber Bragg Gratings (FBG), each transducing specific physical or chemical changes into optical signals[115-117]. Hybrid designs, such as combining a semi-distributed interferometer (SDI) with an FBG, allow simultaneous, decoupled monitoring of sweat biomarkers and skin temperature, effectively eliminating cross-sensitivity[118].
Optical fiber sensors offer unique advantages for wearability, including complete immunity to electromagnetic interference, excellent biocompatibility, and minimal invasiveness due to their small diameter (~ 125 µm). Their flexibility allows seamless integration into textiles, garments, or flexible substrates, enabling conformal skin contact without compromising comfort[119]. Performance-wise, they achieve high sensitivity, fast response, and low cross-sensitivity. For instance, a fiber-optic pH sensor demonstrated a sensitivity of -27.06 nm/pH with a response time of 15-20 s and negligible temperature cross-sensitivity[117]. A surface plasmon resonance (SPR)-based cholesterol sensor achieved a LOD as low as 0.0051 µM[116]. Integration strategies include embedding fibers in soft polymers such as PDMS, weaving them into fabrics, or incorporating them into functional hydrogels. Readout systems are increasingly miniaturized, employing LEDs, photodiodes, and microcontrollers, with data transmitted wirelessly via Bluetooth. For simpler outputs, smartphone-based colorimetric or intensity analysis combined with machine learning enables practical on-body use[119,120].
Advancing towards multifunctional integration, Zhang et al. fabricated a wearable hydrogel optical fiber sensor via photopolymerization molding for simultaneous human posture and sweat pH monitoring [Figure 8A][120]. The sensor features a polyethylene glycol diacrylate (PEGDA) hydrogel core and a calcium alginate hydrogel cladding. Posture sensing is achieved through bending-induced optical path changes, while sweat pH sensing relies on the fluorescence intensity variation of Rhodamine B. The sensor exhibits excellent biocompatibility, flexibility (withstanding 700 bending cycles), and low optical loss (0.6 dB·cm-1), demonstrating the potential of multifunctional wearable optical fibers for integrated physiological monitoring. Building on this need for enhanced chemical detection, Lei et al. developed a tapered fiber Mach-Zehnder interferometer sensor functionalized with a chitosan/polyacrylic acid polyelectrolyte film for sweat pH detection[117]. The film’s swelling degree varies with pH, altering its refractive index and causing a wavelength shift in the interference spectrum. The sensor achieved a high sensitivity of -27.06 nm/pH within the physiologically relevant sweat pH range of 5.0-7.5, with low temperature cross-sensitivity (0.012 nm/°C), showing a high-performance design for wearable metabolite detection.
Figure 8. (A) Schematic of a dual-function hydrogel optical fiber (HOF) sensor. The fiber features a step-index core-cladding structure doped with phenol red, enabling simultaneous monitoring of sweat pH and joint motion. Reproduced with permission[120]. Copyright 2024, American Chemical Society; (B) Diagram of a reflective tapered optical fiber sensor for SERS-based lactate analysis. The fiber tip is chemically etched and decorated with Ag NPs to create high-density hotspots for rapid and sensitive Raman detection. Reproduced with permission[121]. Copyright 2025, Elsevier; (C) Illustration of a fiber optic SPR biosensor for cholesterol detection. The sensing probe is fabricated via electroless silver plating on the fiber core and functionalized with cholesterol oxidase for specific enzymatic recognition. Reproduced with permission[116]. Copyright 2025, Elsevier. APTES: (3-aminopropyl)triethoxysilane; β-CD: β-cyclodextrin; CHOL: cholesterol; SPR: surface plasmon resonance; EDC: 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide; NHS: N-hydroxysuccinimide; SERS: surface-enhanced Raman scattering.
Further advancing sensitivity and specificity for trace metabolite detection, Wang et al. fabricated a reflective tapered optical fiber SERS sensor using Ag NPs via chemical modification techniques for lactic acid detection in sweat [Figure 8B][121]. The tapered geometry enhances the evanescent field, while the nanoparticles provide strong electromagnetic enhancement. The sensor achieved a LOD of 2.67 × 10-8 M for lactic acid and successfully detected lactic acid Raman peaks in real sweat samples, highlighting its potential for highly sensitive wearable Raman-based monitoring. Despite high sensitivity, real-world sweat monitoring often involves complex environments where temperature fluctuations and multiple interfering factors must be decoupled to ensure accurate metabolite quantification. To address such multi-parameter challenges, Karipbayeva et al. developed a SDI assisted by a fiber Bragg grating (FBG) for dual-parameter sensing[118]. The SDI is sensitive to external refractive index (biomarker concentration), while the FBG provides precise temperature measurement. This combination enables low-cross-talk, simultaneous monitoring of sweat biomarkers (e.g., azelaic acid) and temperature, offering a robust platform for applications in complex physiological environments. Expanding the detection to low-concentration lipid metabolites in sweat, Zhang et al. fabricated a fiber-optic SPR cholesterol biosensor using a polydopamine and β-cyclodextrin functional film [Figure 8C][116]. The sensor operates via SPR wavelength shifts induced by cholesterol binding. It achieved a high sensitivity of 133.83 nm/µM and a LOD of 0.0051 µM, presenting a significant innovation for the non-invasive, label-free monitoring of trace cholesterol in sweat and sebum.
Despite the distinct advantages of fiber-optic sensing in terms of mechanical stability and operational simplicity, translating this technology into robust, wearable platforms presents several significant challenges. Key obstacles include the miniaturization of readout electronics, as commercial interrogators for FBGs or spectrometers remain bulky and power-intensive[115]. The integration of brittle silica fibers with soft, flexible substrates poses risks of delamination and fracture under mechanical deformation, while the scalable fabrication of sensitive elements such as fiber tapers and gratings contributes to increased production costs[116]. For implantable applications, ensuring long-term biocompatibility and resistance to biofouling necessitates advanced material engineering strategies[119]. Future development will focus on novel materials such as hydrogel-based fibers, multifunctional sensor networks, and integration with AI and the internet of things (IoT) for intelligent health monitoring[119]. Through continued innovation, fiber-optic sensing is poised to complement established optical methods, offering quantitative, real-time capability distinct from semi-quantitative colorimetry, yet with simpler instrumentation than SERS and greater stability than fluorescence or ECL systems.
CONCLUSION AND OUTLOOK
The vision of wearable optical patches for continuous, non-invasive sweat sensing represents a paradigm shift in personalized health monitoring. As detailed in this review, significant advancements have been made across colorimetric, SERS, fluorescence, ECL, and optical fiber-based platforms, demonstrating their unique advantages in sensitivity, specificity, and integration with soft, wearable form factors. However, the path from compelling laboratory prototypes to reliable, commercially viable, and clinically trusted devices is complex, necessitating a concerted effort to address complicated scientific, engineering, and clinical challenges. The fundamental hurdle lies in bridging the knowledge gap between sweat composition and systemic physiology. While sweat is rich in metabolites, electrolytes, and trace biomarkers such as hormones and proteins, the mechanisms governing their secretion and partitioning from blood remain poorly characterized for many analytes[19]. This incomplete understanding directly impacts the clinical interpretation of sensor data. Correlations between sweat analyte concentrations and blood levels or specific health states can be confounded by individual variations in sweat gland density, secretory rate, and biological pathways, leading to potentially conflicting or non-comparable results across studies[19]. Therefore, establishing the true diagnostic value of optical sweat sensing requires extensive, large-scale population studies that correlate multimodal sensing data with gold-standard clinical measurements to build robust, physiologically meaningful interpretation models[3,9].
Beyond physiological interpretation, the practical operation of optical sensors on the dynamic, heterogeneous environment of human skin presents huge technical obstacles. The nature of sweat secretion, such as low volume, irregular without stimulation, and prone to evaporation, challenges the core requirement of consistent sample delivery to the optical sensing zone[3]. This issue is acute for sensing modalities that require precise reagent mixing or controlled reaction times. For instance, in colorimetric and enzymatic fluorescence assays, irregular sweat flow can lead to delayed response, incomplete reagent mixing, or signal saturation, compromising the accuracy of endpoint measurements. For mechanisms such as colorimetry and fluorescence that rely on chemical reactions, irregular sweat flow can lead to delayed response, incomplete reagent mixing, or signal saturation. Furthermore, the on-body environment is a source of significant noise. Ambient light interference is a major concern for colorimetric and fluorescence-based sensors, where it can distort color interpretation or swamp weak fluorescence signals. This necessitates careful optical shielding or the use of NIR-excited probes (e.g., UCNPs in fluorescence) to minimize background[43,54,100]. For SERS and fluorescence, signal integrity is further threatened by skin-derived contaminants (e.g., lipids and dead cells), which can foul the sensitive plasmonic nanostructures or quench fluorescent probes, leading to signal drift or loss. Temperature fluctuations pose another challenge, as they may alter enzymatic reaction kinetics crucial for many colorimetric and fluorescent assays, and can also affect the refractive index sensing mechanism in optical fiber-based platforms, requiring effective temperature compensation strategies[117,118]. Motion artifacts and mechanical stress from daily activities threaten the integrity of both the delicate optical components and the fluidic networks designed to transport sweat. For SERS sensors, maintaining the uniformity and enhancement factor of plasmonic nanostructures under repeated stretching or bending is a critical yet unsolved challenge, as deformation can alter inter-particle gaps and thus the local electromagnetic field[70,71,94]. Similarly, the mechanical robustness of thin fluorescent films and the interfacial adhesion between rigid optical fibers and soft substrates are key concerns for long-term wearable fluorescence and fiber-optic sensors, respectively[100,115]. Ensuring long-term mechanical robustness, stable adhesion to moist skin, and effective encapsulation against environmental intrusions are therefore critical, yet non-trivial, design goals[9], especially for systems containing rigid electronic components (common in ECL and active readout modules) or fragile silica fibers[115,122].
The quest for a fully autonomous, user-friendly device introduces system-level integration challenges that are particularly acute for active optical methods. A self-contained “lab-on-skin” patch would ideally house not only the sensing chemistry but also miniaturized light sources (e.g., UV LEDs, laser diodes for SERS/fluorescence), photodetectors, signal processing circuits, a power source, and wireless communication modules-all within a thin, soft, and stretchable architecture, which remains a significant engineering aspiration. The inherent need for external optical components differentiates these modalities: colorimetry often relies on a separate smartphone, whereas fluorescence, SERS, and ECL require integrated light sources or electrodes. This integration directly trades off against device miniaturization, flexibility, and power autonomy[122]. Power consumption is a critical constraint; while colorimetry can leverage passive smartphone imaging, techniques such as fluorescence, SERS, ECL, and active optical fiber sensing require energy for excitation or electrochemical triggering. This makes power management a central design criterion, especially for continuous monitoring applications. Advances in flexible energy storage (e.g., thin-film batteries, supercapacitors) and, more promisingly, the development of self-powered systems that harvest energy from body motion or from the sweat biofluid itself (e.g., biofuel cells) are essential research directions to enable truly continuous, maintenance-free monitoring[9,122].
Looking ahead, the future evolution of this field will be driven by convergence and intelligence. Material innovation will focus on enhancing the stability and performance of optical elements: developing photobleaching-resistant and environmentally stable fluorescent probes, engineering SERS substrates with not only high enhancement factors but also superior mechanical resilience to maintain hotspot uniformity under deformation, creating robust, stretchable, and high-conductivity composites for ECL electrodes, and advancing novel flexible waveguide materials (e.g., advanced hydrogels) for optical fiber sensors to improve durability and skin compatibility[94,108,119,123]. Sensing mechanisms will increasingly leverage data science. The integration of machine learning and AI with optical readouts is a powerful trend. Deep learning algorithms can transform smartphone-captured colorimetric images into highly accurate quantitative data, overcoming limitations of visual interpretation[86]. Similarly, advanced spectral analysis powered by machine learning can deconvolute complex SERS or fluorescence signatures for reliable multiplexed detection, even in the presence of biological background, and can assist in decoupling cross-sensitivity in multi-parameter fiber-optic sensors[71,86,118]. The ultimate wearable platform will likely be multimodal, seamlessly combining optical sweat metabolite sensing with physical sensors for contextual parameters such as sweat rate and skin temperature. This fusion of data streams will enable sophisticated algorithms to compensate for confounding variables (e.g., correcting analyte concentration for sweat rate variations) and extract more profound, actionable health insights[16].
The final leap from advanced prototype to widespread application depends on scalable manufacturing, rigorous validation, and clear demonstration of utility. Economical, high-throughput fabrication techniques (e.g., roll-to-roll printing, injection molding) must be adapted for the precision required in making microfluidics and nanostructured optical components[9]. This includes the reproducible, large-scale fabrication of uniform SERS-active nanostructures, the precise patterning of microfluidic channels for controlled sweat delivery across all platforms, and the cost-effective integration of micro/nano-optical features for fiber-based sensors[9,65,115]. Most importantly, the field must move beyond proof-of-concept studies in controlled settings to conduct extensive, real-world clinical trials. These trials are necessary to validate sensor accuracy against established clinical standards, to define the clinical relevance of sweat-based biomarkers for specific conditions (from athletic performance to metabolic disorders), and to earn regulatory approval and user trust[9,123]. The collaborative synergy between material scientists, optical engineers, data analysts, and clinical researchers will be the cornerstone of translating the remarkable potential of flexible optical sweat sensors into tangible tools that empower individuals with real-time, meaningful knowledge about their own physiology, ultimately paving the way for a more proactive and personalized approach to healthcare.
DECLARATIONS
Acknowledgments
We thank Chu Wang for assistance with the creation of Figures 1-3 and the Graphical Abstract using BioRender.com (created in BioRender; 2026; https://BioRender.com/dtltj3c, https://BioRender.com/w58hd9a, https://BioRender.com/tfxxgjh, and https://BioRender.com/1qnvpvm).
Authors’ contributions
Conceptualization and supervision and project administration: Dong, X.; Qu, L.-L.; Guo, Y.
Investigation, original draft, and figure editing: Shuai, Y.; Wu, X.; Cao, C.; Chen, L.
Manuscript revision: Shuai, Y.; Qu, L. -L.; Guo, Y.; Cao, C.; Chen, L.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Yunlong Lake Laboratory of Deep Underground Science and Engineering Project (104024004), Jiangsu Provincial Innovation Capacity Building Program (BM2022009 and BM2023017), Key Project of Basic Research Program of Jiangsu Province (BK20243036), and Basic Research Program of Jiangsu Province (BK20231527).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Manjakkal, L.; Yin, L.; Nathan, A.; Wang, J.; Dahiya, R. Energy autonomous sweat-based wearable systems. Adv. Mater. 2021, 33, e2100899.
2. Karpova, E. V.; Karyakina, E. E.; Karyakin, A. A. Wearable non-invasive monitors of diabetes and hypoxia through continuous analysis of sweat. Talanta 2020, 215, 120922.
4. Sharma, A.; Badea, M.; Tiwari, S.; Marty, J. L. Wearable biosensors: an alternative and practical approach in healthcare and disease monitoring. Molecules 2021, 26, 748.
5. Smith, A. A.; Li, R.; Tse, Z. T. H. Reshaping healthcare with wearable biosensors. Sci. Rep. 2023, 13, 4998.
6. Xu, J.; Fang, Y.; Chen, J. Wearable biosensors for non-invasive sweat diagnostics. Biosensors 2021, 11, 245.
7. Song, Z.; Zhou, S.; Qin, Y.; et al. Flexible and wearable biosensors for monitoring health conditions. Biosensors 2023, 13, 630.
8. Vo, D. K.; Trinh, K. T. L. Advances in wearable biosensors for healthcare: current trends, applications, and future perspectives. Biosensors 2024, 14, 560.
9. Ghaffari, R.; Yang, D. S.; Kim, J.; et al. State of sweat: emerging wearable systems for real-time, noninvasive sweat sensing and analytics. ACS. Sens. 2021, 6, 2787-801.
10. Nonaka, T.; Wong, D. T. W. Saliva diagnostics. Annu. Rev. Anal. Chem. (Palo. Alto. Calif). 2022, 15, 107-21.
11. Song, M.; Bai, H.; Zhang, P.; Zhou, X.; Ying, B. Promising applications of human-derived saliva biomarker testing in clinical diagnostics. Int. J. Oral. Sci. 2023, 15, 2.
12. Ghelli, F.; Panizzolo, M.; Garzaro, G.; et al. Inflammatory biomarkers in exhaled breath condensate: a systematic review. Int. J. Mol. Sci. 2022, 23, 9820.
13. Szunerits, S.; Dӧrfler, H.; Pagneux, Q.; et al. Exhaled breath condensate as bioanalyte: from collection considerations to biomarker sensing. Anal. Bioanal. Chem. 2023, 415, 27-34.
14. Dos Santos, C. C.; Lucena, G. N.; Pinto, G. C.; Júnior, M. J.; Marques, R. F. Advances and current challenges in non‐invasive wearable sensors and wearable biosensors - a mini‐review. Med. Devices. Sens. 2020, 4, e10130.
15. Ma, C. B.; Shang, X.; Sun, M.; et al. Emerging multifunctional wearable sensors: integrating multimodal sweat analysis and advanced material technologies for next-generation health monitoring. ACS. Sens. 2025, 10, 2388-408.
16. Luo, D.; Sun, H.; Li, Q.; Niu, X.; He, Y.; Liu, H. Flexible sweat sensors: from films to textiles. ACS. Sens. 2023, 8, 465-81.
17. Pour, S. R. S.; Calabria, D.; Emamiamin, A.; et al. Microfluidic-based non-invasive wearable biosensors for real-time monitoring of sweat biomarkers. Biosensors 2024, 14, 29.
18. Xuan, X.; Pérez-Ràfols, C.; Chen, C.; Cuartero, M.; Crespo, G. A. Lactate biosensing for reliable on-body sweat analysis. ACS. Sens. 2021, 6, 2763-71.
19. Davis, N.; Heikenfeld, J.; Milla, C.; Javey, A. The challenges and promise of sweat sensing. Nat. Biotechnol. 2024, 42, 860-71.
20. Jo, S.; Sung, D.; Kim, S.; Koo, J. A review of wearable biosensors for sweat analysis. Biomed. Eng. Lett. 2021, 11, 117-29.
21. Yang, D. S.; Ghaffari, R.; Rogers, J. A. Sweat as a diagnostic biofluid. Science 2023, 379, 760-1.
22. Marvelli, A.; Campi, B.; Mergni, G.; et al. Sweat chloride assay by inductively coupled plasma mass spectrometry: a confirmation test for cystic fibrosis diagnosis. Anal. Bioanal. Chem. 2020, 412, 6909-16.
23. Aloke, C.; Egwu, C. O.; Aja, P. M.; et al. Current advances in the management of diabetes mellitus. Biomedicines 2022, 10, 2436.
24. Huang, T.; Liang, Z.; Wang, K.; Miao, X.; Zheng, L. Novel insights into athlete physical recovery concerning lactate metabolism, lactate clearance and fatigue monitoring: a comprehensive review. Front. Physiol. 2025, 16, 1459717.
25. Knezevic, E.; Nenic, K.; Milanovic, V.; Knezevic, N. N. The role of cortisol in chronic stress, neurodegenerative diseases, and psychological disorders. Cells 2023, 12, 2726.
26. Pulopulos, M. M.; Baeken, C.; De Raedt, R. Cortisol response to stress: The role of expectancy and anticipatory stress regulation. Horm. Behav. 2020, 117, 104587.
27. Ghaffari, R.; Rogers, J. A.; Ray, T. R. Recent progress, challenges, and opportunities for wearable biochemical sensors for sweat analysis. Sens. Actuators. B. Chem. 2021, 332, 129447.
28. Heng, W.; Yang, G.; Kim, W. S.; Xu, K. Emerging wearable flexible sensors for sweat analysis. Bio-des. Manuf. 2022, 5, 64-84.
29. Choi, D. H.; Kitchen, G. B.; Stewart, K. J.; Searson, P. C. The dynamic response of sweat chloride to changes in exercise load measured by a wearable sweat sensor. Sci. Rep. 2020, 10, 7699.
30. Lihong, Z.; Qiuping, L. Advancements and obstacles in sweat-based biosensors for health monitoring. Crit. Rev. Anal. Chem. 2025, 1-32.
31. Yu, H.; Sun, J. Sweat detection theory and fluid driven methods: a review. Nanotechnol. Precis. Eng. 2020, 3, 126-40.
32. Brasier, N.; Eckstein, J. Sweat as a source of next-generation digital biomarkers. Digit. Biomark. 2020, 3, 155-65.
33. Tang, Y.; Li, X.; Lv, H.; Wang, W.; Zhi, C.; Li, H. Integration designs toward new‐generation wearable energy supply‐sensor systems for real‐time health monitoring: a minireview. InfoMat 2020, 2, 1109-30.
34. Johnson, K. B.; Wei, W. Q.; Weeraratne, D.; et al. Precision medicine, AI, and the future of personalized health care. Clin. Transl. Sci. 2021, 14, 86-93.
35. Iqbal, S. M. A.; Mahgoub, I.; Du, E.; Leavitt, M. A.; Asghar, W. Advances in healthcare wearable devices. NPJ. Flex. Electron. 2021, 5, 9.
36. Hu, M.; Wang, Z.; Zhang, L.; Lin, S.; Liao, J. A microfluidic patch for wireless wearable electrochemical detection of sweat metabolites. Sens. Actuators. B. Chem. 2025, 422, 136604.
37. Bilbao, E.; Garate, O.; Rodríguez Campos, T.; et al. Electrochemical sweat sensors. Chemosensors 2023, 11, 244.
38. Liu, G.; Wang, H.; Zhang, P.; et al. A wearable multi-parameter electrochemical detection and signal processing system for real-time sweat analysis. Mikrochim. Acta. 2025, 192, 604.
39. Childs, A.; Mayol, B.; Lasalde-Ramírez, J. A.; Song, Y.; Sempionatto, J. R.; Gao, W. Diving into sweat: advances, challenges, and future directions in wearable sweat sensing. ACS. Nano. 2024, 18, 24605-16.
40. Gao, N.; Xu, G.; Chang, G.; Wu, Y. From lab to life: self-powered sweat sensors and their future in personal health monitoring. Adv. Sci. (Weinh). 2025, 12, e2409178.
41. Wang, X.; Shen, C.; Zhou, C.; Bu, Y.; Yan, X. Methods, principles and applications of optical detection of metal ios. Chem. Eng. J. 2021, 417, 129125.
42. Kumar, S.; Singh, R. Recent optical sensing technologies for the detection of various biomolecules: review. Opt. Laser. Technol. 2021, 134, 106620.
43. Wang, J.; Luo, Y.; Zhou, Z.; Xiao, J.; Xu, T.; Zhang, X. Epidermal wearable optical sensors for sweat monitoring. Commun. Mater. 2024, 5, 77.
44. Qin, J.; Jiang, S.; Wang, Z.; et al. Metasurface micro/nano-optical sensors: principles and applications. ACS. Nano. 2022, 16, 11598-618.
45. Min, J.; Tu, J.; Xu, C.; et al. Skin-interfaced wearable sweat sensors for precision medicine. Chem. Rev. 2023, 123, 5049-138.
46. Vasanth, A.; Ashok, A.; Do, T. N.; Phan, H. P. Advancements in flexible porous nanoarchitectonic materials for biosensing applications. Adv. Colloid. Interface. Sci. 2025, 339, 103439.
47. Ariati, R.; Sales, F.; Souza, A.; Lima, R. A.; Ribeiro, J. Polydimethylsiloxane composites characterization and its applications: a review. Polymers. (Basel). 2021, 13, 4258.
48. Teixeira, I.; Castro, I.; Carvalho, V.; et al. Polydimethylsiloxane mechanical properties: a systematic review. AIMS. Mater. Sci. 2021, 8, 952-73.
49. Choi, J.; Xue, Y.; Xia, W.; et al. Soft, skin-mounted microfluidic systems for measuring secretory fluidic pressures generated at the surface of the skin by eccrine sweat glands. Lab. Chip. 2017, 17, 2572-80.
50. Koh, A.; Kang, D.; Xue, Y.; et al. A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 2016, 8, 366ra165.
51. Lima, R. A. The impact of polydimethylsiloxane (PDMS) in engineering: recent advances and applications. Fluids 2025, 10, 41.
52. Kan, X.; Shi, E.; Zhu, C.; Wang, Q.; Qu, L.; Dong, X. Binary solvent-induced ultra-adhesive gel for underwater motion and water flow detection. ACS. Appl. Polym. Mater. 2025, 7, 6523-32.
53. Qu, X.; Wang, Q.; Gan, D.; Sun, H.; Ni, Z.; Dong, X. Tissue-adaptable hydrogel for mechanically compliant bioelectronic interfaces. Nano. Lett. 2025, 25, 4759-66.
54. Hu, B.; Kang, X.; Xu, S.; Zhu, J.; Yang, L.; Jiang, C. Multiplex chroma response wearable hydrogel patch: visual monitoring of urea in body fluids for health prognosis. Anal. Chem. 2023, 95, 3587-95.
55. Wang, W.; Chen, Y.; Xiao, C.; et al. Flexible SERS wearable sensor based on nanocomposite hydrogel for detection of metabolites and pH in sweat. Chem. Eng. J. 2023, 474, 145953.
56. Sun, H.; Qu, X.; Wang, Q.; Guo, Y.; Dong, X. Dynamic regulation of interfacial adhesion in biomedical hydrogels. Chem. Soc. Rev. 2026, 55, 469-503.
57. Qu, X.; Wang, Q.; Sun, H.; et al. Molecular competition induced Janus hydrogel bioelectronic interface for electroceutical modulation. Nat. Commun. 2025, 17, 455.
58. Chao, F.; Cao, C.; Xu, Y.; et al. Sprayable hydrogel for pH-responsive nanozyme-derived bacteria-infected wound healing. ACS. Appl. Mater. Interfaces. 2025, 17, 5921-32.
59. Gan, D.; Wang, Q.; Zhang, X.; et al. Congeneric and robust adhesive epidermal patch for anti-interference physiological signal recognition. Adv. Funct. Mater. 2024, 34, 2411588.
60. Shao, Z.; Chen, H.; Wang, Q.; et al. Melt electrowriting ordered TPU microfibrous mesh for on-demand colorimetric wearable sweat detection. IEEE. Sensors. J. 2022, 22, 18560-6.
61. Sheng, F.; Zhao, C.; Zhang, B.; Tan, Y.; Dong, K. Flourishing electronic textiles towards pervasive, personalized and intelligent healthcare. Soft. Sci. 2024, 4, 2.
62. Hou, Z.; Liu, X.; Tian, M.; et al. Smart fibers and textiles for emerging clothe-based wearable electronics: materials, fabrications and applications. J. Mater. Chem. A. 2023, 11, 17336-72.
63. Zhang, Y.; Wang, H.; Lu, H.; Li, S.; Zhang, Y. Electronic fibers and textiles: recent progress and perspective. iScience 2021, 24, 102716.
64. Zhang, S.; Tan, R.; Xu, X.; Iqbal, S.; Hu, J. Fibers/textiles-based flexible sweat sensors: a review. ACS. Materials. Lett. 2023, 5, 1420-40.
65. Koh, E. H.; Lee, W. C.; Choi, Y. J.; et al. A wearable surface-enhanced raman scattering sensor for label-free molecular detection. ACS. Appl. Mater. Interfaces. 2021, 13, 3024-32.
66. Han, Y.; Fang, X.; Li, H.; Zha, L.; Guo, J.; Zhang, X. Sweat sensor based on wearable janus textiles for sweat collection and microstructured optical fiber for surface-enhanced raman scattering analysis. ACS. Sens. 2023, 8, 4774-81.
67. Mogera, U.; Guo, H.; Namkoong, M.; Rahman, M. S.; Nguyen, T.; Tian, L. Wearable plasmonic paper-based microfluidics for continuous sweat analysis. Sci. Adv. 2022, 8, eabn1736.
68. Khan, S. M.; Nassar, J. M.; Hussain, M. M. Paper as a substrate and an active material in paper electronics. ACS. Appl. Electron. Mater. 2020, 3, 30-52.
69. Eskandari, V.; Sahbafar, H.; Zeinalizad, L.; Marashipour, R.; Hadi, A. A review of paper-based substrates as surface-enhanced Raman spectroscopy (SERS) biosensors and microfluidic paper-based SERS platforms. J. Comput. Appl. Mech. 2022, 53, 142-56.
70. Chung, M.; Skinner, W. H.; Robert, C.; et al. Fabrication of a wearable flexible sweat pH sensor based on SERS-active Au/TPU electrospun nanofibers. ACS. Appl. Mater. Interfaces. 2021, 13, 51504-18.
71. Jeon, J.; Lee, S.; Chae, S.; et al. All-flexible chronoepifluidic nanoplasmonic patch for label-free metabolite profiling in sweat. Nat. Commun. 2025, 16, 8017.
72. Fan, F.; Chen, L.; Wang, Y.; et al. Damage-free dry transfer printing of ultrathin films with on-demand interfacial adhesion: principles and applications. Soft. Sci. 2025, 5, 52.
73. Chen, S.; Qiao, Z.; Niu, Y.; et al. Wearable flexible microfluidic sensing technologies. Nat. Rev. Bioeng. 2023, 1, 950-71.
74. Ayuso, J. M.; Virumbrales-Muñoz, M.; Lang, J. M.; Beebe, D. J. A role for microfluidic systems in precision medicine. Nat. Commun. 2022, 13, 3086.
75. Ray, T. R.; Choi, J.; Bandodkar, A. J.; et al. bio-integrated wearable systems: a comprehensive review. Chem. Rev. 2019, 119, 5461-533.
76. Baker, L. B.; Model, J. B.; Barnes, K. A.; et al. Skin-interfaced microfluidic system with personalized sweating rate and sweat chloride analytics for sports science applications. Sci. Adv. 2020, 6, eabe3929.
77. Vaquer, A.; Barón, E.; de la Rica, R. Wearable analytical platform with enzyme-modulated dynamic range for the simultaneous colorimetric detection of sweat volume and sweat biomarkers. ACS. Sens. 2021, 6, 130-6.
78. Villa, J. E. L.; Garcia, I.; Jimenez de Aberasturi, D.; Pavlov, V.; Sotomayor, M. D. P. T.; Liz-Marzán, L. M. SERS-based immunoassay for monitoring cortisol-related disorders. Biosens. Bioelectron. 2020, 165, 112418.
79. Ardalan, S.; Hosseinifard, M.; Vosough, M.; Golmohammadi, H. Towards smart personalized perspiration analysis: an IoT-integrated cellulose-based microfluidic wearable patch for smartphone fluorimetric multi-sensing of sweat biomarkers. Biosens. Bioelectron. 2020, 168, 112450.
80. Li, H.; Ma, Y.; Huang, Y. Material innovation and mechanics design for substrates and encapsulation of flexible electronics: a review. Mater. Horiz. 2021, 8, 383-400.
81. Tu, J.; Yeom, J.; Ulloa, J. C.; et al. Stressomic: A wearable microfluidic biosensor for dynamic profiling of multiple stress hormones in sweat. Sci. Adv. 2025, 11, eadx6491.
82. Luo, Y.; Abidian, M. R.; Ahn, J. H.; et al. Technology roadmap for flexible sensors. ACS. Nano. 2023, 17, 5211-95.
83. Deng, M. Z.; Zhong, M. Y.; Li, M. L.; et al. Research progress on electrochemiluminescence nanomaterials and their applications in biosensors - a review. Anal. Chim. Acta. 2025, 1361, 344148.
84. Kim, J.; Oh, S.; Yang, D. S.; et al. A skin-interfaced, miniaturized platform for triggered induction, capture and colorimetric multicomponent analysis of microliter volumes of sweat. Biosens. Bioelectron. 2024, 253, 116166.
85. Liu, S.; Yang, D. S.; Wang, S.; et al. Soft, environmentally degradable microfluidic devices for measurement of sweat rate and total sweat loss and for colorimetric analysis of sweat biomarkers. EcoMat 2022, 5, e12270.
86. Liu, Z.; Li, J.; Li, J.; et al. Explainable deep-learning-assisted sweat assessment via a programmable colorimetric chip. Anal. Chem. 2022, 94, 15864-72.
87. Zheng, X. T.; Goh, W. P.; Yu, Y.; et al. Skin-attachable ink-dispenser-printed paper fluidic sensor patch for colorimetric sweat analysis. Adv. Healthc. Mater. 2024, 13, e2302173.
88. Cheng, Y.; Feng, S.; Ning, Q.; et al. Dual-signal readout paper-based wearable biosensor with a 3D origami structure for multiplexed analyte detection in sweat. Microsyst. Nanoeng. 2023, 9, 36.
89. Kim, J.; Wu, Y.; Luan, H.; et al. A skin-interfaced, miniaturized microfluidic analysis and delivery system for colorimetric measurements of nutrients in sweat and supply of vitamins through the skin. Adv. Sci. (Weinh). 2022, 9, e2103331.
90. Ghaffari, R.; Choi, J.; Raj, M. S.; et al. soft wearable systems for colorimetric and electrochemical analysis of biofluids. Adv. Funct. Mater. 2019, 30, 1907269.
91. Xi, P.; He, X.; Fan, C.; et al. Smart Janus fabrics for one-way sweat sampling and skin-friendly colorimetric detection. Talanta 2023, 259, 124507.
92. Kwon, K.; Kim, J. U.; Deng, Y.; et al. An on-skin platform for wireless monitoring of flow rate, cumulative loss and temperature of sweat in real time. Nat. Electron. 2021, 4, 302-12.
93. Sung, D.; Han, S.; Kim, S.; et al. Electrophoretic digital colorimetry integrated with electrochemical sweat sensor. Sci. Adv. 2025, 11, eadu2142.
94. Gui, X.; Xie, J.; Wang, W.; et al. Wearable and flexible nanoporous surface-enhanced raman scattering substrates for sweat enrichment and analysis. ACS. Appl. Nano. Mater. 2023, 6, 11049-60.
95. Li, G.; Fan, X.; Tang, X.; et al. Challenges and prospects of personalized healthcare based on surface-enhanced Raman spectroscopy. Research. (Wash. D. C). 2024, 7, 0572.
96. Liu, X.; Ma, J.; Jiang, P.; et al. Large-scale flexible surface-enhanced Raman scattering (SERS) sensors with high stability and signal homogeneity. ACS. Appl. Mater. Interfaces. 2020, 12, 45332-41.
97. Xie, L.; Zeng, H.; Zhu, J.; et al. State of the art in flexible SERS sensors toward label-free and onsite detection: from design to applications. Nano. Res. 2022, 15, 4374-94.
98. Laing, S.; Sloan-Dennison, S.; Faulds, K.; Graham, D. Surface enhanced Raman scattering for biomolecular sensing in human healthcare monitoring. ACS. Nano. 2025, 19, 8381-400.
99. He, X.; Fan, C.; Luo, Y.; Xu, T.; Zhang, X. Flexible microfluidic nanoplasmonic sensors for refreshable and portable recognition of sweat biochemical fingerprint. NPJ. Flex. Electron. 2022, 6, 60.
100. Lee, G. H.; Moon, H.; Kim, H.; et al. Multifunctional materials for implantable and wearable photonic healthcare devices. Nat. Rev. Mater. 2020, 5, 149-65.
101. Shin, Y.; Teresa Gutierrez-wing, M.; Choi, J. Review - recent progress in portable fluorescence sensors. J. Electrochem. Soc. 2021, 168, 017502.
102. Sekine, Y.; Kim, S. B.; Zhang, Y.; et al. A fluorometric skin-interfaced microfluidic device and smartphone imaging module for in situ quantitative analysis of sweat chemistry. Lab. Chip. 2018, 18, 2178-86.
103. Cho, S. H.; Cho, S.; Lv, Z.; et al. Soft, wearable, microfluidic system for fluorometric analysis of loss of amino acids through eccrine sweat. Lab. Chip. 2025, 25, 1647-55.
104. Cui, Y.; Duan, W.; Jin, Y.; Wo, F.; Xi, F.; Wu, J. Ratiometric fluorescent nanohybrid for noninvasive and visual monitoring of sweat glucose. ACS. Sens. 2020, 5, 2096-105.
105. Chen, Q.; Li, S.; Tu, X.; Zhang, X. Skin-attachable Tb-MOF ratio fluorescent sensor for real-time detection of human sweat pH. Biosens. Bioelectron. 2024, 263, 116606.
106. Chen, M. M.; Cheng, S. B.; Ji, K.; et al. Construction of a flexible electrochemiluminescence platform for sweat detection. Chem. Sci. 2019, 10, 6295-303.
107. Ma, X.; Gao, W.; Du, F.; et al. Rational design of electrochemiluminescent devices. Acc. Chem. Res. 2021, 54, 2936-45.
108. Chen, Y.; Chen, J.; Luo, Y.; et al. Stretchable photonic crystal-enhanced ECL for sweat neurotransmitter detection. Anal. Chem. 2025, 97, 19525-31.
109. Ferraraccio, L. S.; Bertoncello, P. Electrochemiluminescence (ECL) biosensor based on tris(2,2'-bipyridyl)ruthenium(II) with glucose and lactate dehydrogenases encapsulated within alginate hydrogels. Bioelectrochemistry 2023, 150, 108365.
110. Cai, X.; Yan, J.; Chu, H.; Wu, M.; Tu, Y. An exercise degree monitoring biosensor based on electrochemiluminescent detection of lactate in sweat. Sens. Actuators. B. Chem. 2010, 143, 655-9.
111. Hu, Y.; Li, J.; Liu, J.; Yu, X.; Yang, J.; Li, Y. A simple yet multifunctional sensing platform inspired by healing-assembly hydrogels serving motion and sweat monitoring. Sens. Actuators. B. Chem. 2023, 378, 133173.
112. Sun, Y.; Li, P.; Zhu, Y.; et al. In situ growth of TiO2 nanowires on Ti3C2 MXenes nanosheets as highly sensitive luminol electrochemiluminescent nanoplatform for glucose detection in fruits, sweat and serum samples. Biosens. Bioelectron. 2021, 194, 113600.
113. Kwon, D. K.; Myoung, J. M. Wearable and semitransparent pressure-sensitive light-emitting sensor based on electrochemiluminescence. ACS. Nano. 2020, 14, 8716-23.
114. Gomez, D. Fibre optic humidity and pH sensing for healthcare and sports applications. Sens. Actuators. B. Chem. 2018, 254, 887-95. https://eprints.nottingham.ac.uk/55406/ (accessed 2026-2-27).
115. Duarah, R.; Torné-Morató, H.; Zhang, G.; et al. Next-generation wearable optical sensors for personalized health and point-of-care diagnostics-a systematic review. Adv. Healthc. Mater. 2026, e04419.
116. Zhang, H.; Chen, S.; Fan, X.; et al. The fiber optic SPR cholesterol biosensor based on electroless silver plating film. Optics. . Laser. Technol. 2025, 192, 113907.
117. Lei, Y.; Gong, Z.; Li, Y.; et al. Highly Sensitive physiological sensor based on tapered fiber-optic interferometer for sweat pH detection. IEEE. Sensors. J. 2023, 23, 11627-34.
118. Karipbayeva, K.; Blanc, W.; Tosi, D. Optical fiber semi-distributed interferometer assisted by an FBG for thermorefractometry and sweat sensing. IEEE. Sensors. J. 2023, 23, 14161-6.
119. Tu, D.; Tang, Y.; Huang, Y.; et al. Next-generation wearable/implanted sensors based on fiber optic and its application: from in vitro to in vivo. ACS. Sens. 2025, 10, 3818-39.
120. Zhang, Z.; Li, K.; Li, Y.; Zhang, Q.; Wang, H.; Hou, C. Dual-function wearable hydrogel optical fiber for monitoring posture and sweat pH. ACS. Sens. 2024, 9, 3413-22.
121. Wang, R.; Yang, X.; Tian, F.; et al. Highly sensitive and rapid Raman detection of lactic acid in human sweat based on reflective tapered optical fiber. Optics. Laser. Technol. 2025, 188, 112975.
122. Xiang, L.; Zeng, X.; Xia, F.; Jin, W.; Liu, Y.; Hu, Y. Recent advances in flexible and stretchable sensing systems: from the perspective of system integration. ACS. Nano. 2020, 14, 6449-69.
123. Yang, Y.; Gao, W. Wearable and flexible electronics for continuous molecular monitoring. Chem. Soc. Rev. 2019, 48, 1465-91.
124. Wang, D.; Xu, G.; Zhang, X.; et al. Dual-functional ultrathin wearable 3D particle-in-cavity SF-AAO-Au SERS sensors for effective sweat glucose and lab-on-glove pesticide detection. Sens. Actuators. B. Chem. 2022, 359, 131512.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






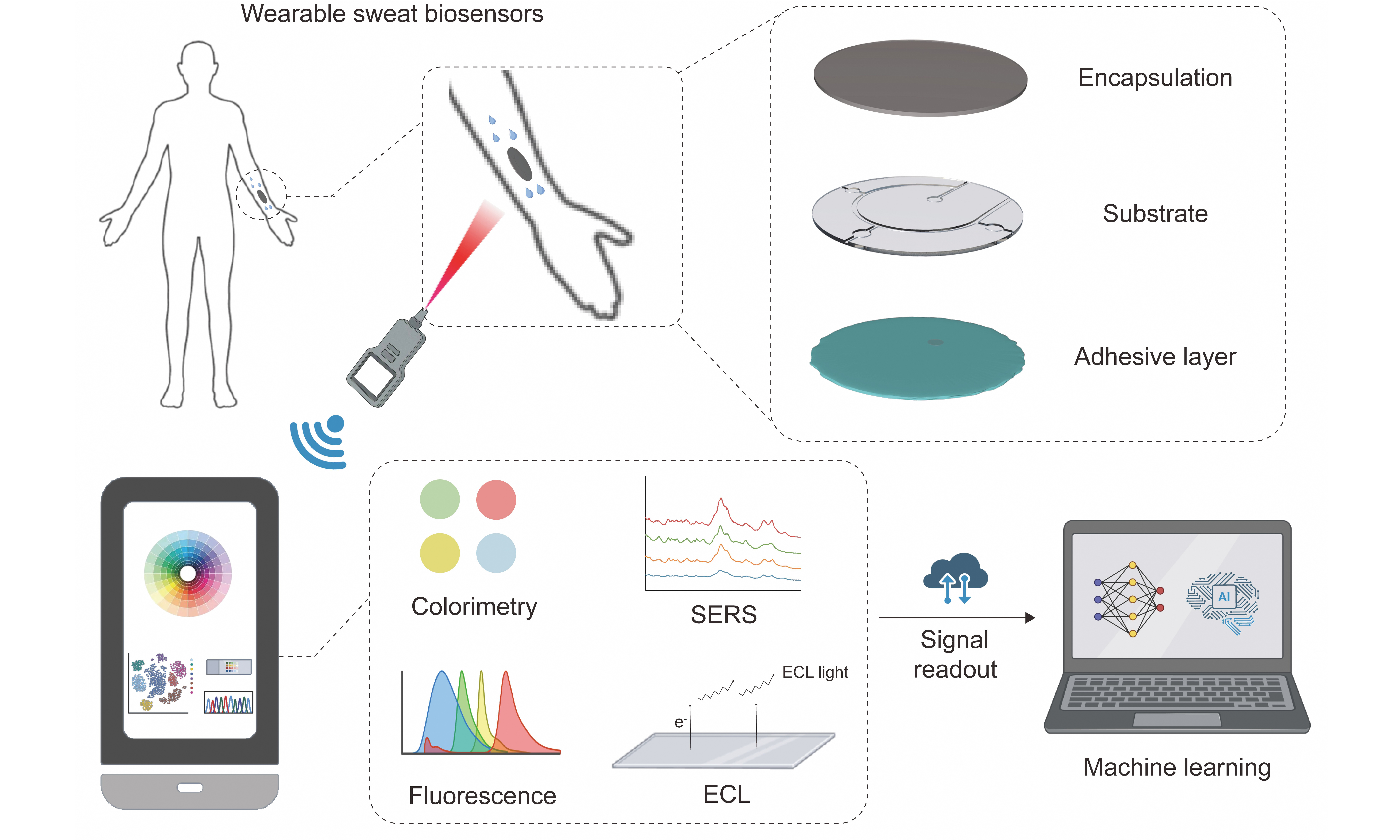
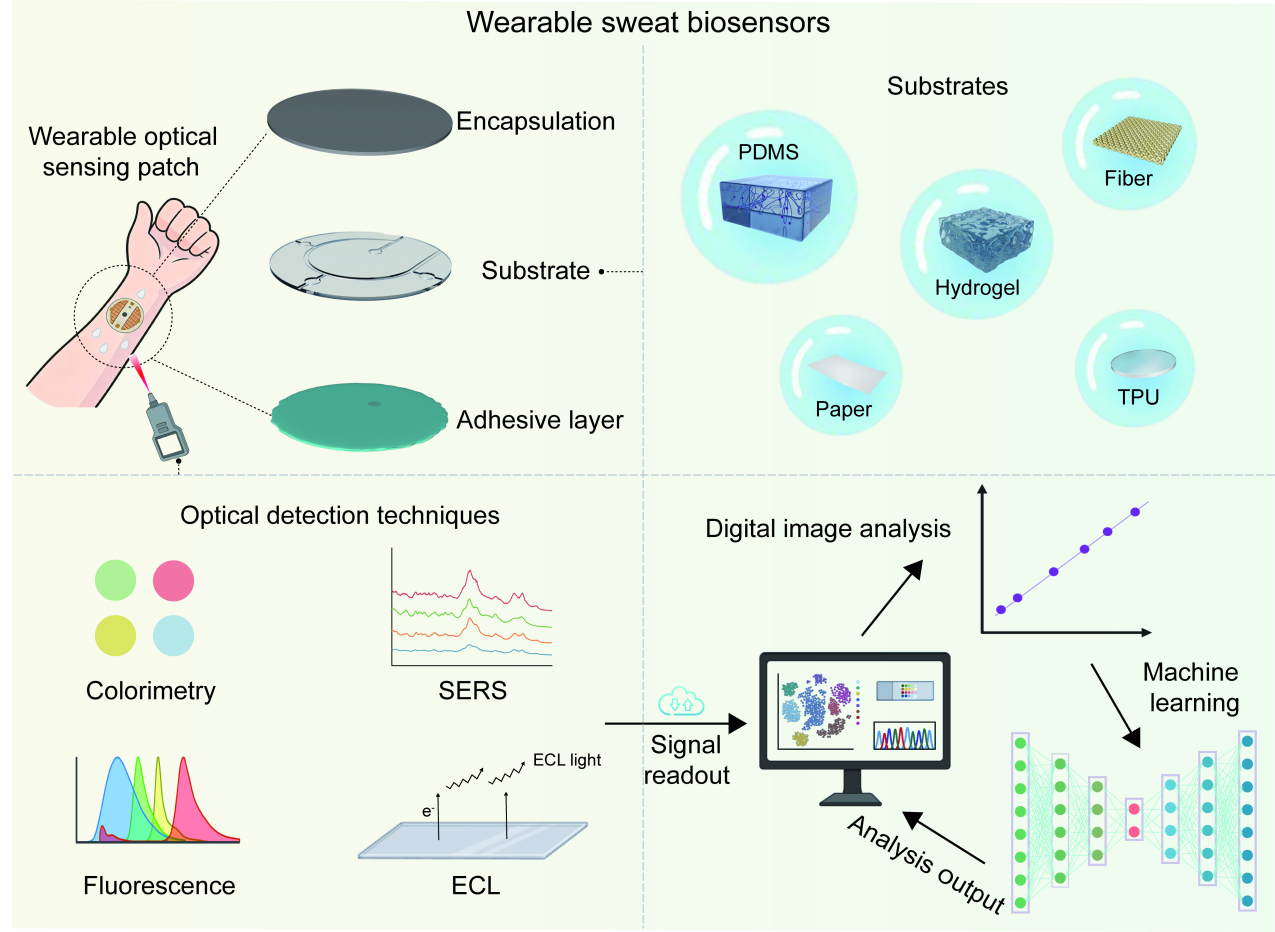
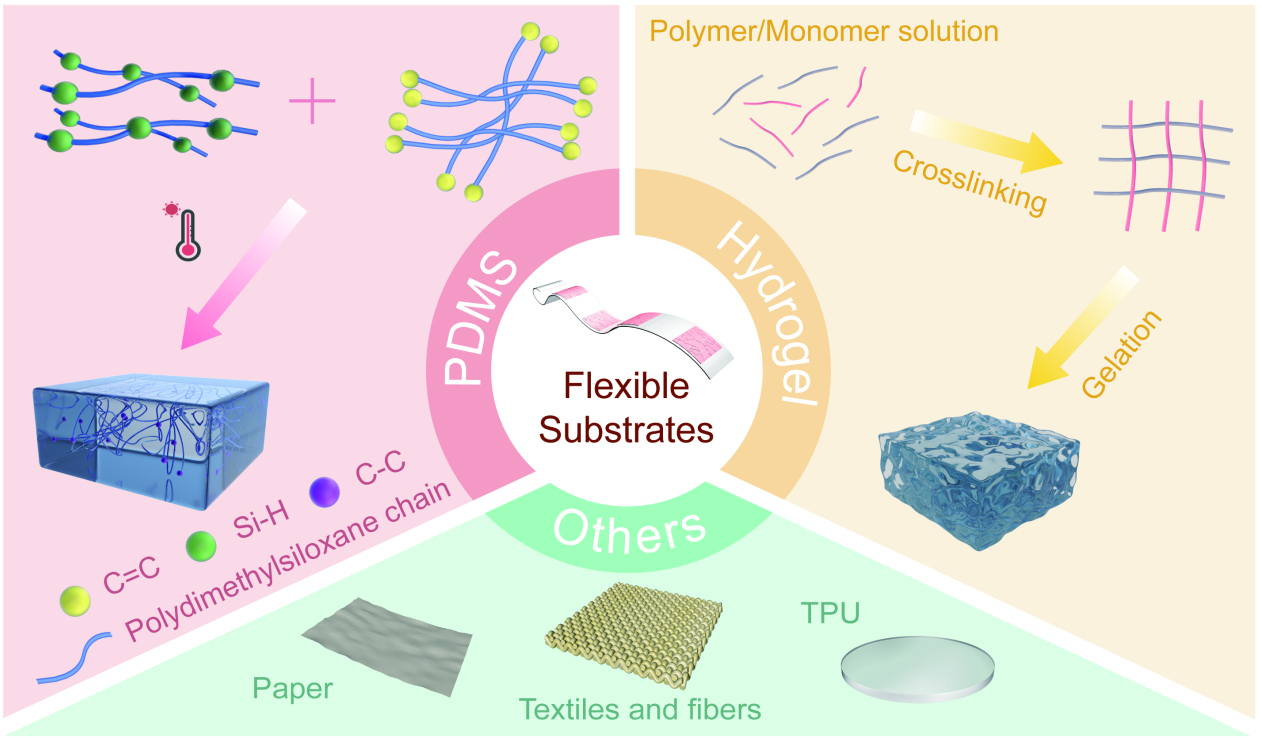
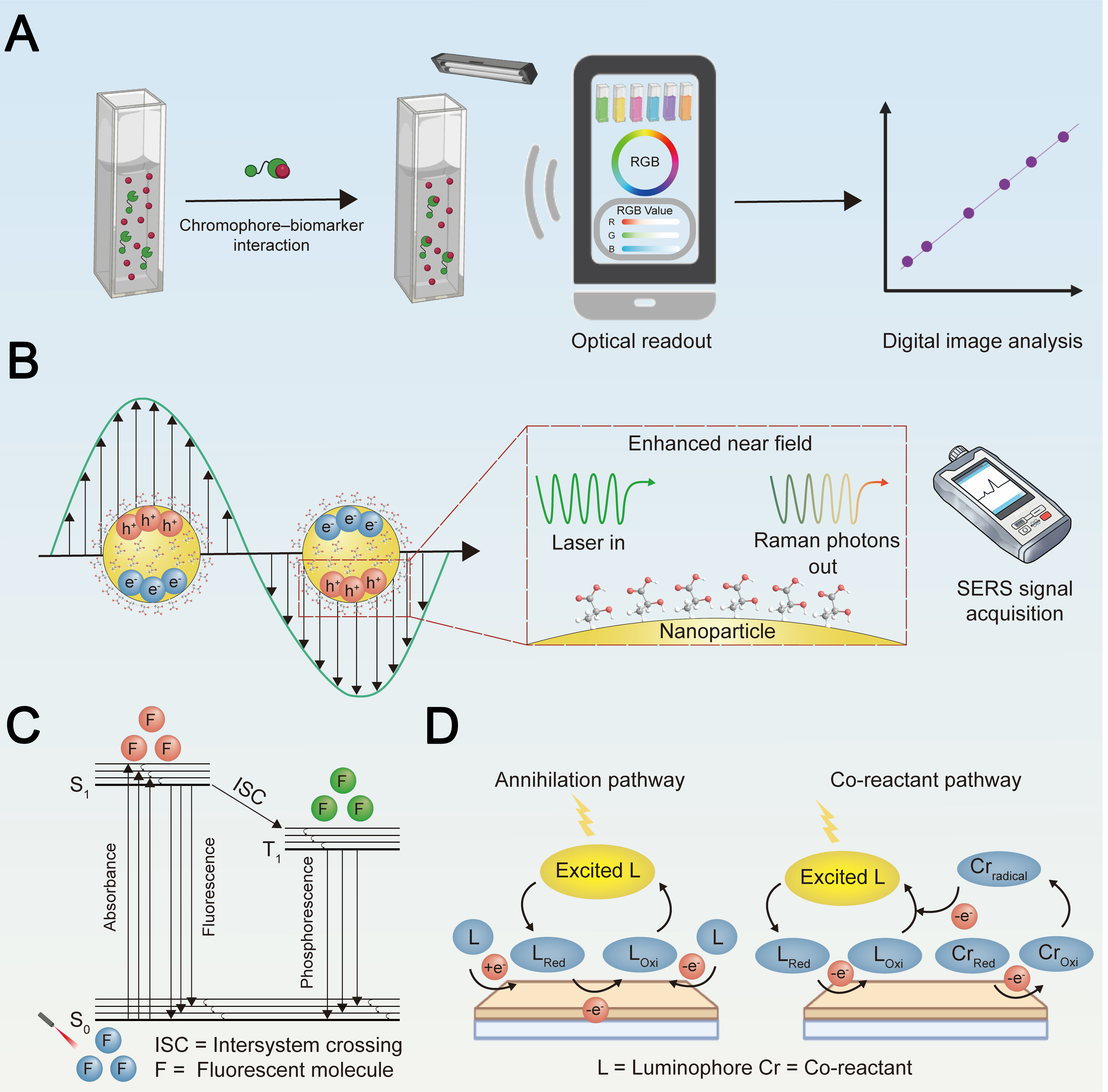
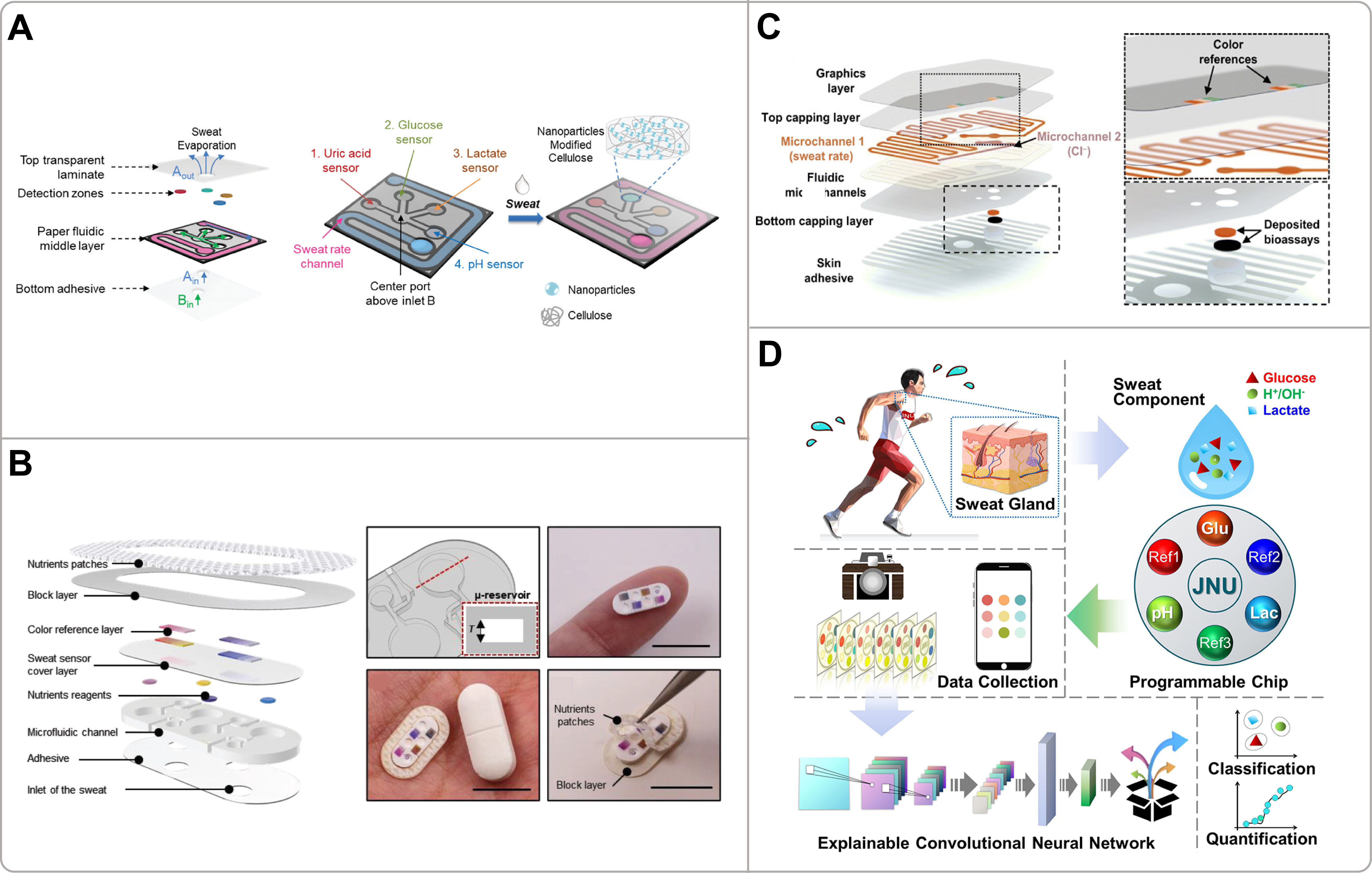
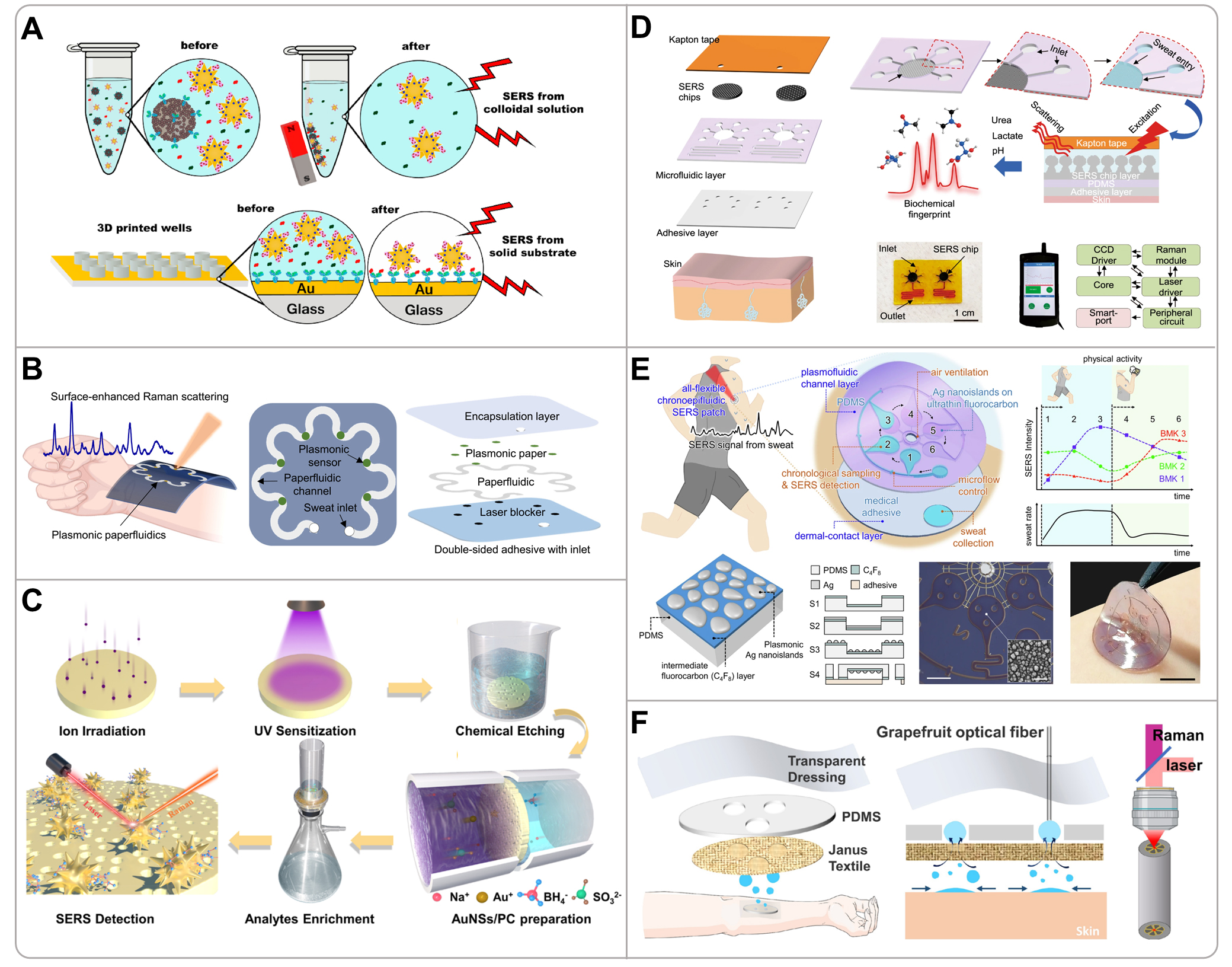
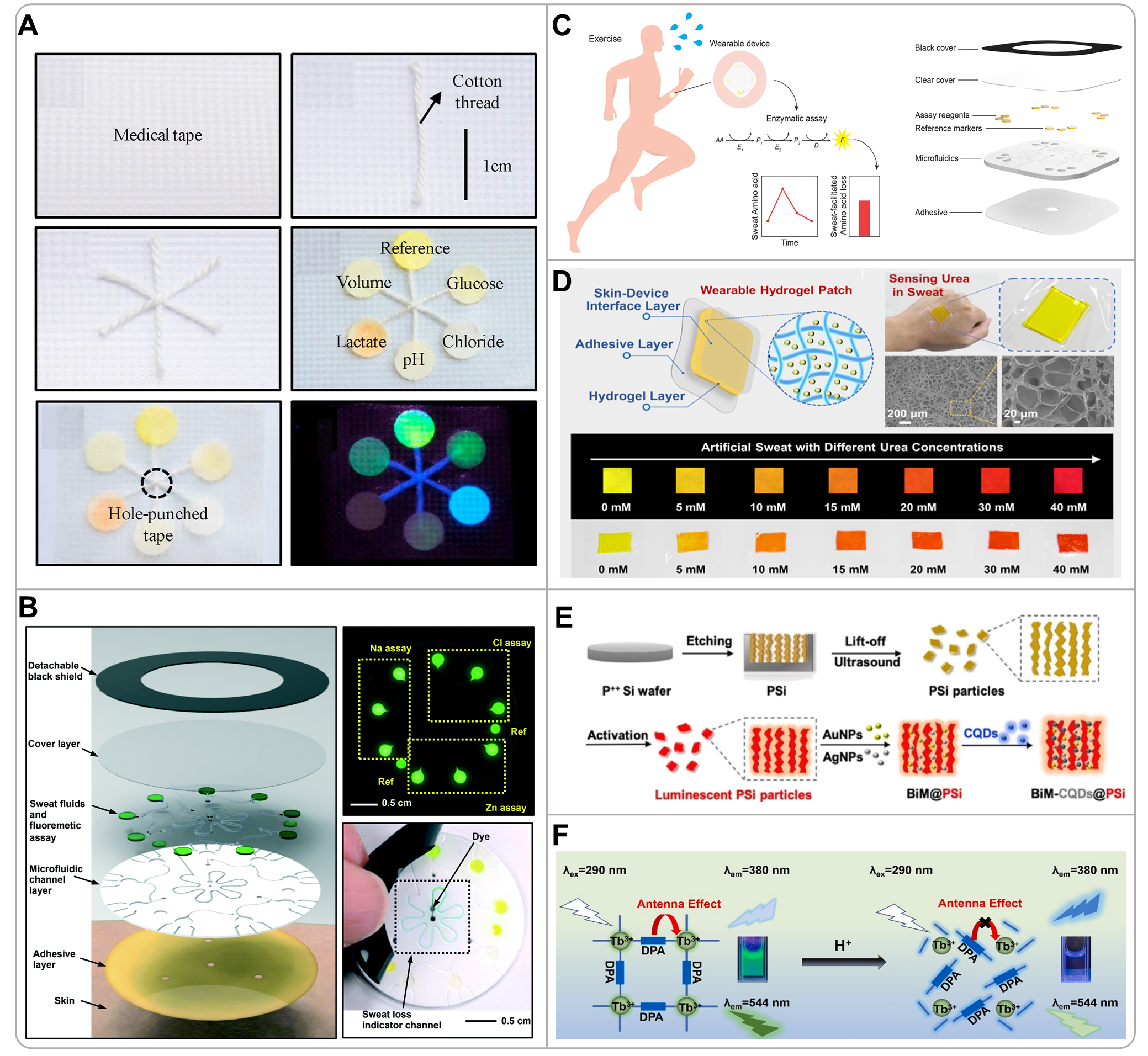
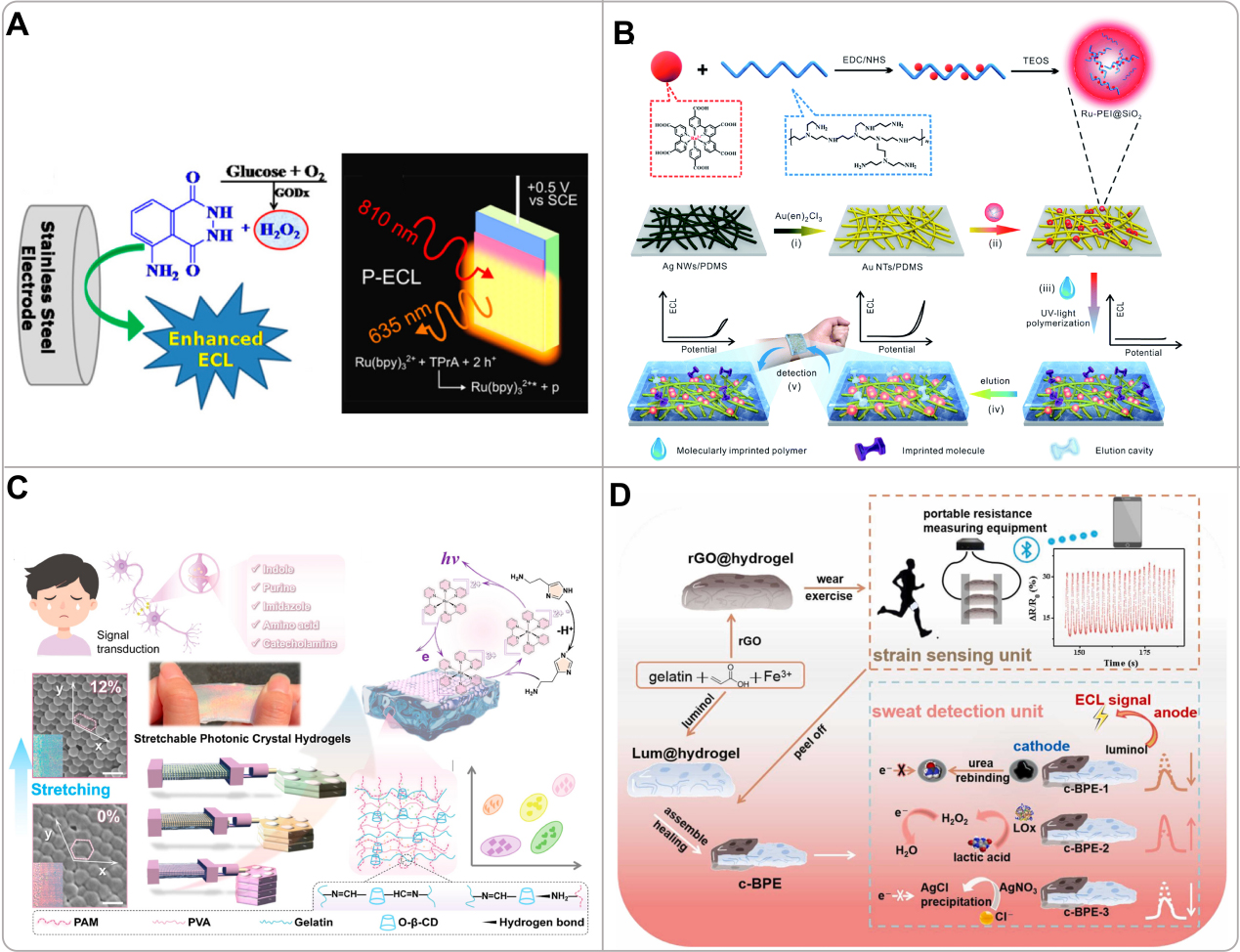
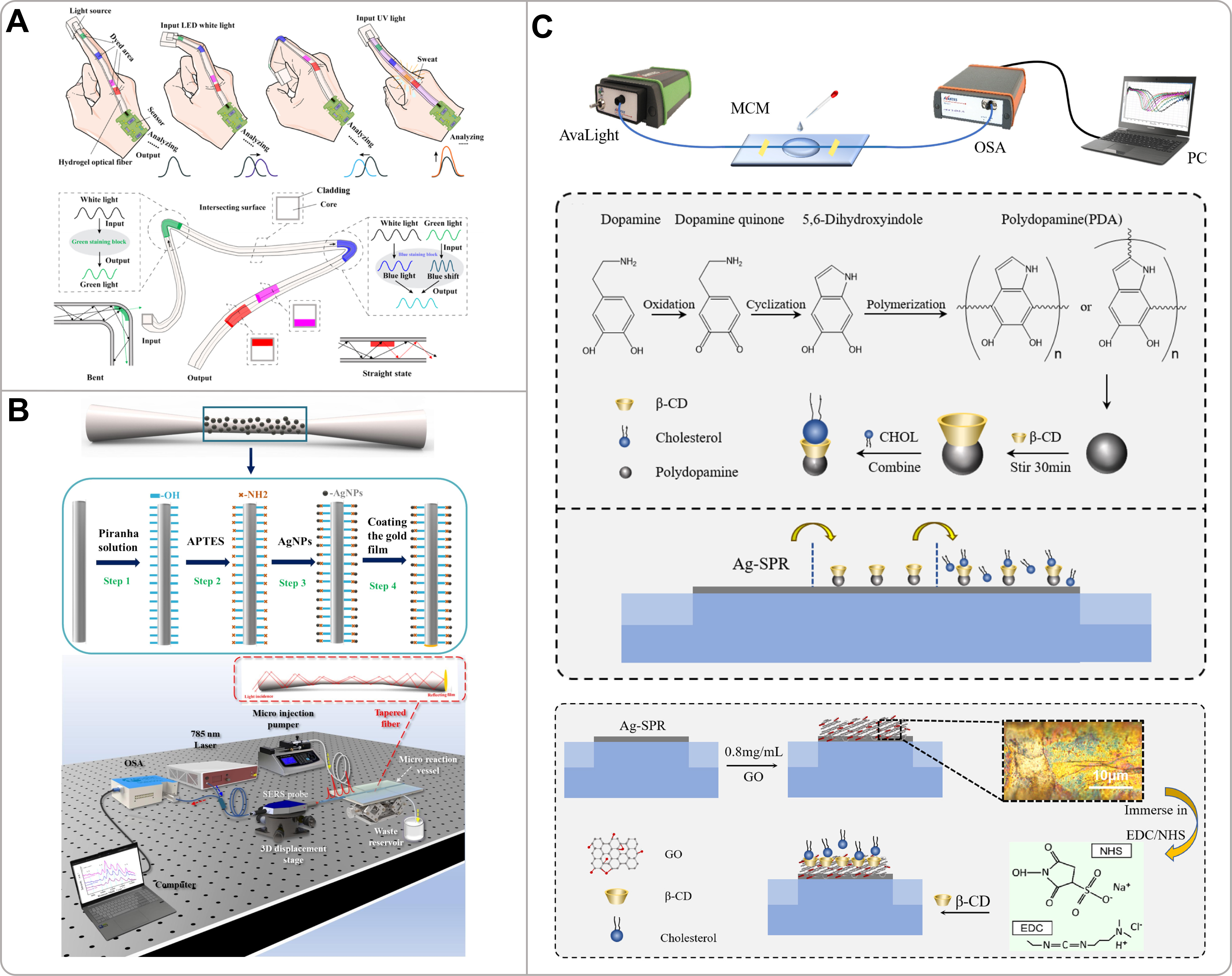












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].