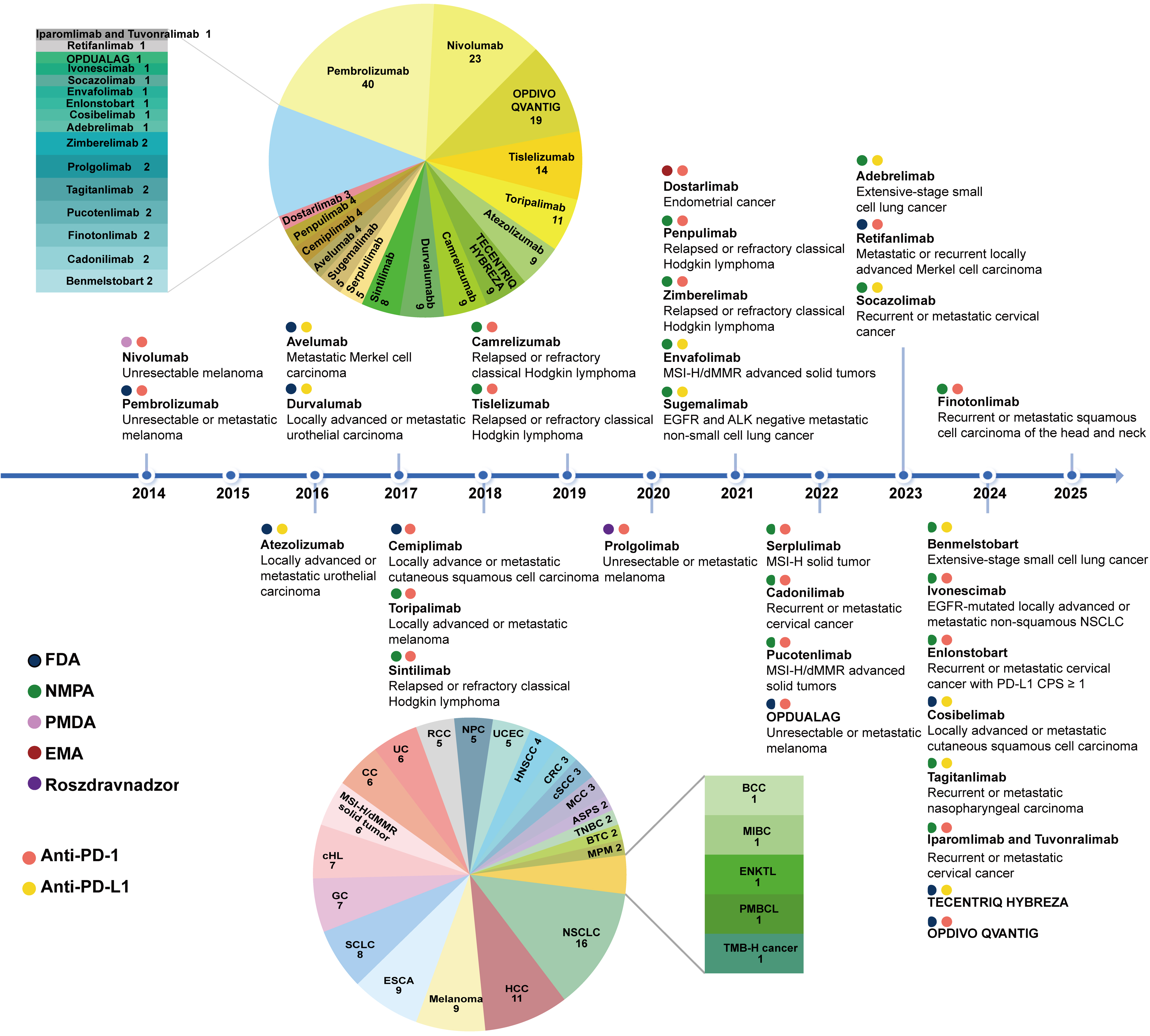
fig3
Figure 3. Timeline of PD-1/PD-L1 inhibitor approvals. The timeline illustrates the first regulatory agency to approve a PD-1/PD-L1 inhibitor and the indications for 32 PD-1/PD-L1 inhibitors. The pie chart in the upper left shows the number of approved indications for each drug. The pie chart in the lower left displays the number of available PD-1/PD-L1 inhibitors for each cancer type. PD-1: Programmed death-1; PD-L1: programmed death-ligand 1; MSI-H: microsatellite instability-high; dMMR: mismatch repair deficiency; EGFR: epidermal growth factor receptor; ALK: anaplastic lymphoma kinase; FDA: Food and Drug Administration; NMPA: National Medical Products Administration (China); PMDA: Pharmaceuticals and Medical Devices Agency (Japan); EMA: European Medicines Agency; Roszdravnadzor: Federal Service for Surveillance in Healthcare (Russia); NSCLC: non-small cell lung cancer; CPS: combined positive score; CC: cervical cancer; UC: urothelial cancer; RCC: renal cell carcinoma; NPC: nasopharyngeal carcinoma; UCEC: endometrial cancer; HNSCC: head and neck squamous cell cancer; CRC: colorectal cancer; cSCC: cutaneous squamous cell carcinoma; MCC: Merkel cell carcinoma; ASPS: alveolar soft part sarcoma; TNBC: triple-negative breast cancer; BTC: biliary tract cancer; MPM: malignant pleural mesothelioma; HCC: hepatocellular carcinoma; ESCA: esophageal cancer; SCLC: small cell lung cancer; GC: gastric cancer; cHL: classical Hodgkin lymphoma; BCC: basal cell carcinoma; MIBC: muscle-invasive bladder cancer; ENKTL: extranodal NK/T cell lymphoma; PMBCL: primary mediastinal large B-cell lymphoma; TMB-H: tumor mutational burden-high.