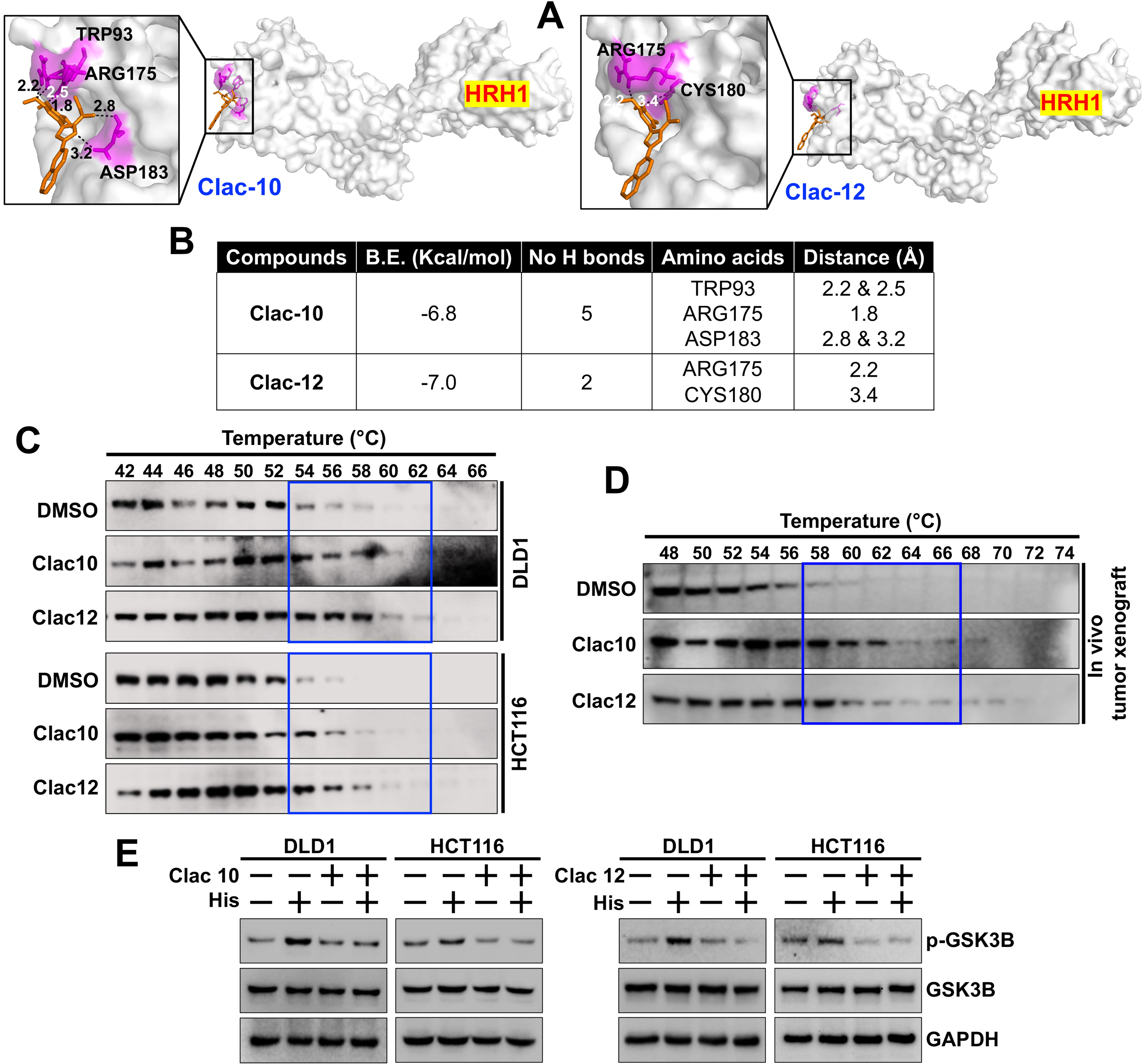
fig6
Figure 6. Clac10 (5c) and Clac12 (5e) bind to HRH1. (A) Molecular docking was used to assess the binding of 5c (Clac10) and 5e (Clac12) to the HRH1 protein cavity. Both compounds bind to the protein with binding energies of -6.8 and -7.0 kcal/mol, respectively. The figure shows cartoon and surface models for 5c (Clac10) and 5e (Clac12); (B) A summary of the docking results and consensus scores for 5c (Clac10) and 5e (Clac12) binding to HRH1 is provided; (C) The CETSA shows that 5c (Clac10) and 5e (Clac12) stabilize the HRH1 protein, suggesting potential binding; The compounds were incubated with cell lysates from HCT116 and DLD1 cell lines (C) or HCT116 Tumor xenograft lysates (D) for 4 h, followed by thermal denaturation and western blot analysis; (E) Preincubation of 5c (Clac10) and 5e (Clac12) inhibits histamine-induced GSK3B phosphorylation in CRC cells. HRH1: Histamine receptor H1; CETSA: cellular thermal shift assay; GSK3B: glycogen synthase kinase 3 beta; CRC: colorectal cancer; DMSO: dimethyl sulfoxide; p-GSK3B: phosphorylated-glycogen synthase kinase 3 beta; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.