Application of brain organoid technology in exploring the neurotoxicity of micro/nanoplastics: a review of progress and challenges
Abstract
As an emerging environmental pollutant, micro/nanoplastics (MNPs) have been reported to accumulate in brain tissues, which may lead to risks of neurotoxicity in humans. Current traditional neurotoxicity models face difficulties in species differences and ethical limitations, whereas brain organoids derived from human cells can simulate the complex structure and function of the human brain, providing a novel model for studying the neurotoxicity of MNPs. In this study, we have reviewed the progress in applying brain organoids to assess the health risks of environmental pollutants including MNPs. MNPs can penetrate into brain organoids and cause toxic effects such as mitochondrial dysfunction, calcium signaling disruption, and abnormal neural differentiation, which may promote pathological phenotypes associated with neurodegenerative diseases. These findings are usually obtained from organoids derived from human-induced pluripotent stem cells, whereas models derived from human embryonic stem cells still require further exploration. Future perspectives are also proposed for technological innovation in brain organoids, including developing region-specific brain organoids, fetal brain organoids, and standardized brain organoid platforms to facilitate their broader application in environmental neurotoxicity assessment of environmental pollutants.
Keywords
INTRODUCTION
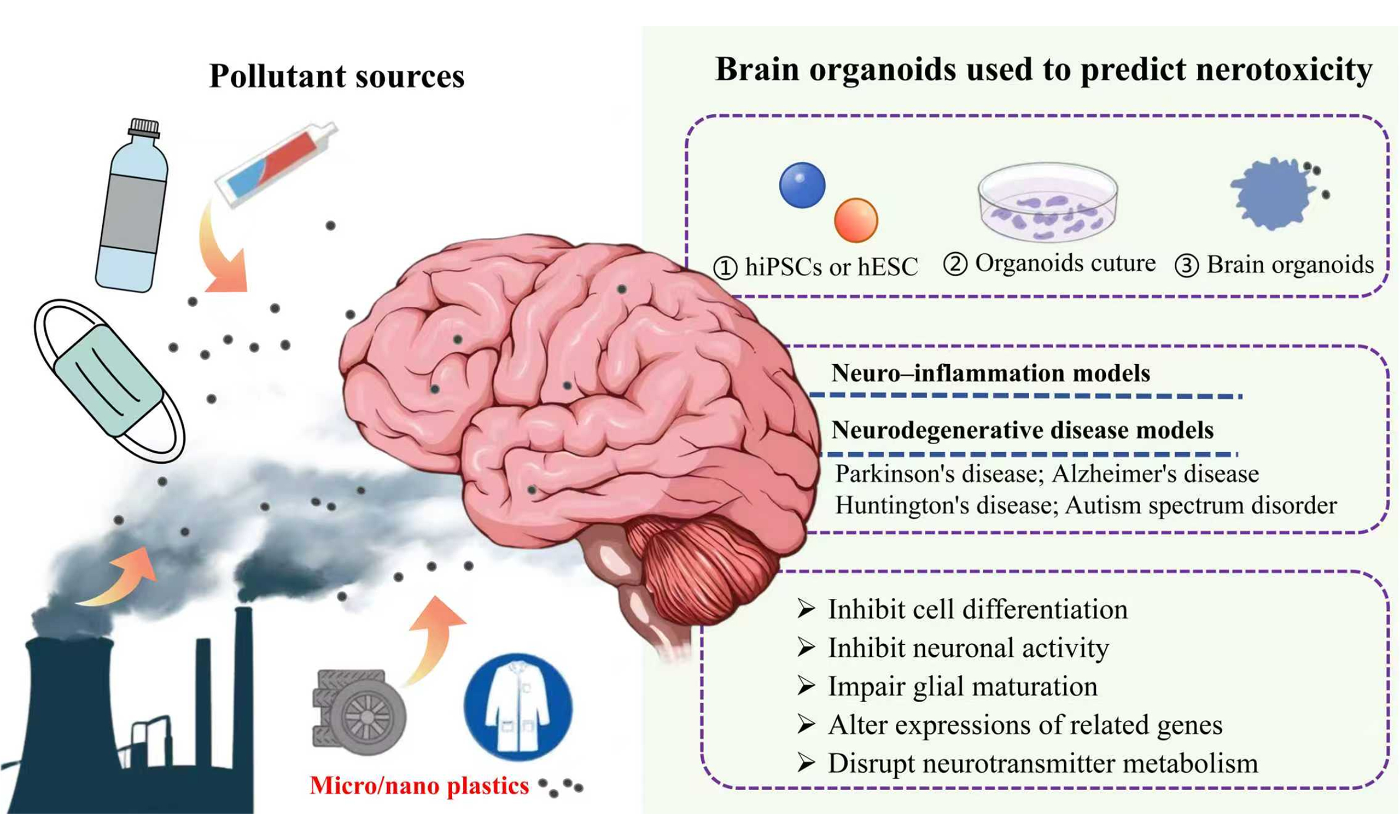
Microplastics (MPs) and nanoplastics (NPs) are emerging environmental pollutants that have been detected in various environments, including terrestrial, aquatic, and atmospheric systems[1,2]. Due to their special physicochemical properties such as small sizes and strong adsorption capacity[3,4], MPs may mix in food and be ingested and transferred to various human tissues and organs, posing potential health risks[5,6]. Current research has confirmed the presence of micro/nanoplastics (MNPs) in postmortem human brains, where their concentrations were significantly higher than those in the liver or kidney[7]. MNPs can also adsorb and transport pathogens, which can alter their bioavailability and environmental fate[8]. Moreover, MPs with sizes less than 5 mm and NPs smaller than 1 μm can easily penetrate into biological systems through ingestion, inhalation, and skin contact, leading to potential health issues[9,10].
The unique physicochemical properties of MNPs contribute to their high mobility, bioaccumulation potential, and complex interactions with organisms, posing significant environmental and health risks. Evidence has strongly suggested that MNPs can penetrate the blood-brain barrier (BBB) and accumulate in the brain[11]. MNPs can activate microglia and astrocytes, leading to chronic neuroinflammation[12]. Animal studies have shown that exposure to MNPs can result in cognitive deficits, behavioral disturbances, and pathological features resembling neurodegenerative diseases[7]. Thus, the risks that MNPs pose to human neurological health cannot be ignored[13,14].
Currently, traditional models used for studying neurotoxicity, such as animal models and two-dimensional (2D) cell cultures, face significant ethical and practical challenges. Animal models often fail to accurately replicate human physiological conditions, which may limit the relevance and applicability of research findings to human health[15]. In addition, the complex neural networks and cognitive functions of the human brain cannot be studied directly by animal models. The inability to accurately replicate human-specific responses limits their applicability in investigating neurobehavioral outcomes[16,17]. Traditional 2D models do not mimic cellular functions in tissues[18], making them unable to accurately simulate the intricate neuronal networks and dynamic complexity of the nervous system[19]. Although human samples can provide more relevant data, their usage is associated with ethical concerns including informed consent, confidentiality, and potential harm to participants[20].
The first report that created human 3D cerebral tissue arose in 2008, which is currently considered as the pioneering work of “neural organoid”[21]. In 2025, Zhao et al. cultured a multilocular brain organoid, marking the successful establishment of a 3D brain organoid culture system[22]. With the advancement of technology, brain organoids primarily sourced from human-induced pluripotent stem cells (hiPSCs) and human embryonic stem cells (hESCs) provide a promising alternative for studying neurotoxicity[23]. These three-dimensional (3D) culture systems can replicate the structural and functional complexity of human tissues, overcoming certain limitations of traditional 2D cell cultures and animal models[24,25]. They allow for the study of dynamic interactions between different cell types and the microenvironment, providing a more accurate model of human brain physiology[26]. Unlike animal models, brain organoids are derived from human pluripotent stem cells, making them more relevant for studying human-specific developmental processes and disease mechanisms[27]. In particular, brain organoids have been shown to simulate key features of human neural development and function, making them an invaluable tool for assessing the neurotoxic effects of various substances[28]. These models provide a translational platform for neurotoxicity research[29].
In this study, we outlined the application and research progress of brain organoid technology. The advances of different brain organoids in exploring the neurotoxic effects of MNPs and their associated mechanisms in causing neurodegenerative diseases were summarized. Finally, the potential challenges and prospects for future research in this field were also proposed.
LITERATURE STATISTICS
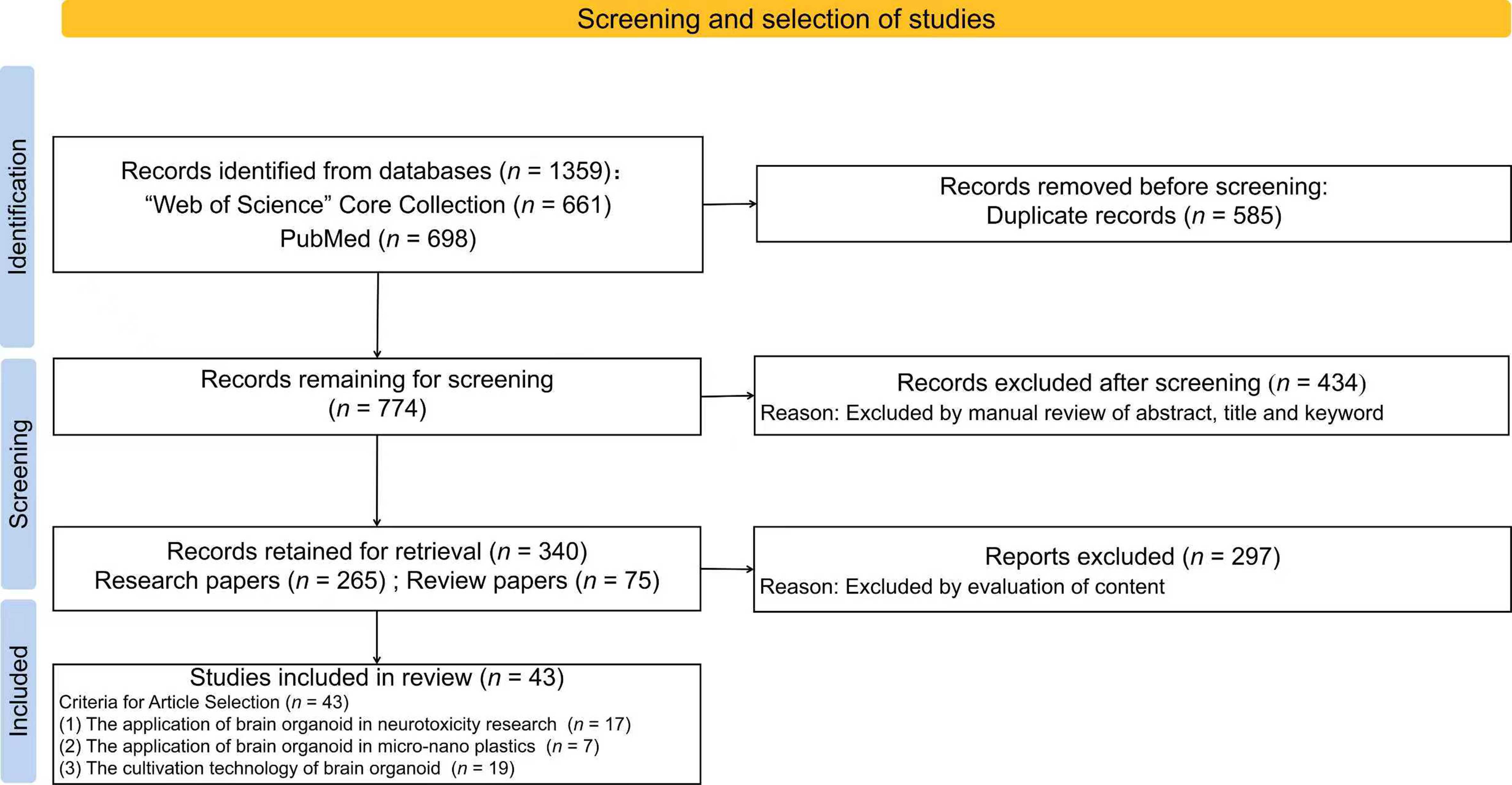
The databases of “Web of Science” (http://www.webofscience.com/) and “PubMed” (https://pubmed.ncbi.nlm.nih.gov/) were used for data collection for bibliometrics and systematic literature reviews. The literature search covered the period from January 2020 to September 2025. The search keywords included “brain organoid* OR cerebral organoid*” & “microplastic* OR nanoplastic*” & “brain organoid* OR cerebral organoid*” & “neurotox*” “brain organoid* OR cerebral organoid*” & “microplastic* OR pollution*” & “brain organoid* OR cerebral organoid*” & “Environmental health” & “brain organoid* OR cerebral organoid*” & “Health risks”. A total of 774 papers were collected following screening based on publication types (research articles and review articles) and duplicates were removed. Subsequently, literature was subjected to manual screening based on the criterion of specific papers discussing the toxic effects using brain tissue models. Finally, 43 articles were included, of which 6 were related to MNPs. The process of literature screening was shown in Figure 1.
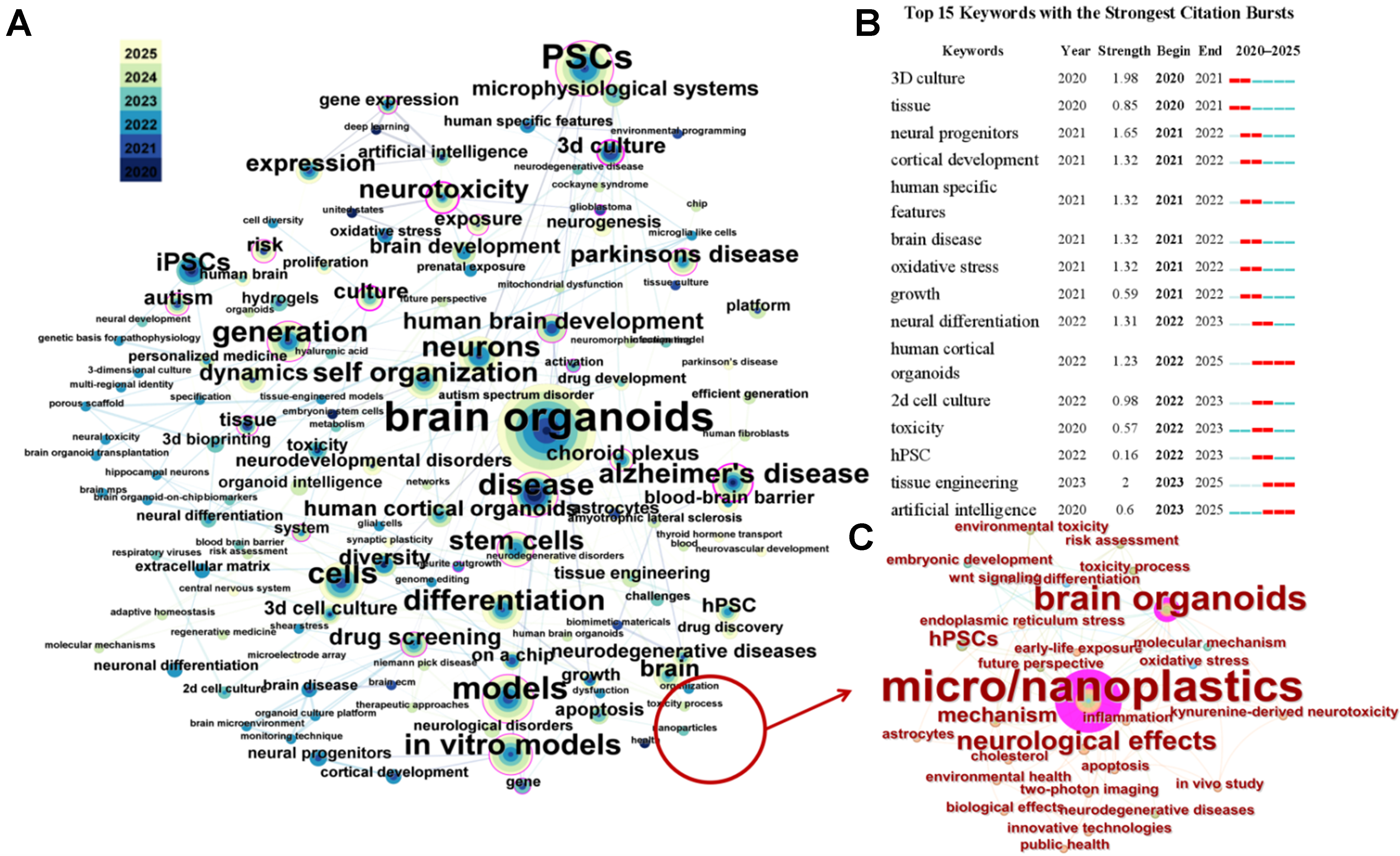
The bibliometric and visualization analysis in this study was conducted using CiteSpace 6.1. R4. Over the past five years, the number of publications on applications of brain organoid technology in toxicity studies has increased annually. According to the keyword co-occurrence analysis, the foundational technologies of brain organoids are human pluripotent stem cells and 3D culture techniques. This field has intersected with multiple areas, including neurological disease modeling, drug screening, and developmental toxicity assessment [Figure 2A]. The field of brain organoid research has gradually evolved from initial model construction and neural differentiation to the exploration of disease mechanisms. It has concentrated on using organoid models to reveal cellular heterogeneity, causes of neurodevelopmental disorders and effects of genetic mutations, as well as exploring the neurodevelopmental toxicity mechanisms of environmental pollutants in depth [Figure 2B].
Figure 2. Publications related to brain organoids in the past five years. Keywords co-occurrence network for all environmental pollutants (A) and MNPs (C) extracted from the Web of Science database. Node size represents the frequency of the keywords; color represents their distribution across years; (B) Top 15 keywords emerging. Red indicates the time period of emergence, light blue indicates that the node has not yet appeared, and dark blue indicates that the node has begun to appear. MNPs: Micro/nanoplastics.
An overview of the literature on the intersection of “micro/nanoplastics” and “brain organoids” over the past five years shows that brain organoids constructed from human pluripotent stem cells have been widely used in the study of the neurotoxic mechanisms of MNPs. These studies primarily focus on mechanisms involving oxidative stress and inflammation [Figure 2C]. Although existing research is still in its early stages, the number of publications in this field shows an upward trend.
ESTABLISHMENT OF BRAIN ORGANOIDS
A brain organoid is a self-organizing 3D tissue derived from hESCs or hiPSCs and is able to simulate the architecture and functionality of the human brain[30]. Brain organoids simulate the anatomical structure, physiological characteristics, and complex neuronal networks of the human brain, reflecting early brain tissue and cellular architecture[23]. They contain various cell types present in the human brain, including neurons, astrocytes, microglia, and oligodendrocytes[31,32]. This enables more accurate simulation of nervous system development and interactions between cell types and their microenvironments[26].
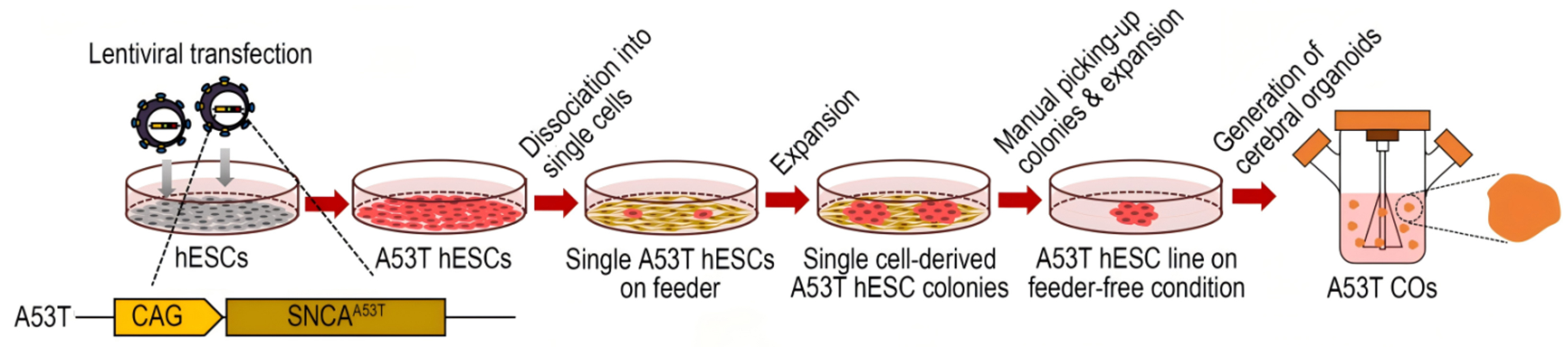
Currently, brain organoids have been derived from hiPSCs and hESCs through an intrinsic self-organization process involving embryoid body (EB) formation, neuroectoderm induction, Matrigel embedding, and culture in spinning bioreactors to yield diverse cell lineages[33]. The process typically begins with EB formation, followed by Matrigel encapsulation and agitation culture. Differentiation may occur through intrinsic self-organization or be guided by exogenous small molecules to generate region-specific brain organoids [Figure 3][34]. Organoids derived from hiPSCs generally follow a similar protocol to those derived from hESCs, although differences in cell origin and differentiation efficiency may result in variations in timing and experimental handling[35].
Figure 3. Schematic diagram of the differentiation strategies for generating hESC-derived brain organoids[34]. hESC: Human embryonic stem cell.
APPLICATION OF BRAIN ORGANOIDS IN MODELING NEUROLOGICAL DISEASES
Brain organoids have been widely used to simulate neurodevelopmental abnormalities and neurodegenerative diseases, providing important support for revealing the pathological mechanisms of related diseases and assessing the potential neurotoxic effects of exogenous substances[36].
Neuroinflammation models
The tubular organoid-on-a-chip constructed using hESCs is an innovative device designed to generate tubular organoids using 3D printed hollow mesh scaffolds. This technology is particularly useful for modeling neuroinflammation and studying neuro-immune interactions. For instance, brain organoids exposed to opioid receptor agonists effectively modeled microglia-mediated neuroinflammation, demonstrating microglia activation and subsequent homeostatic transformation[37]. The neuroinflammation organoid models have been applied to investigate the neurotoxic effects of environmental pollutants. For instance, research on human midbrain organoids derived from hiPSCs shows that perfluorooctane sulfonate can impair neurodevelopment in the brain organoid model and promote neuroinflammation, exhibiting dose-dependent neurotoxic effects[38].
Neurodegenerative disease models
Brain organoids have been used to model various neurodevelopmental and neurodegenerative diseases, including Parkinson’s disease (PD), Alzheimer’s disease (AD), Huntington’s disease (HD), and Autism spectrum disorders (ASD).
PD models
In the research on PD models, the utilization of hiPSCs for generating midbrain organoids has received widespread attention. Researchers have developed a PD model using brain organoids treated with the neurotoxin 6-hydroxydopamine to study key pathophysiological mechanisms such as α-synuclein aggregation, Lewy body formation, dopamine loss, and microglial activation[39]. This neurotoxin-induced model evaluates the neurotoxic effects on dopaminergic neurons through high-content imaging and machine learning-based analysis. It quantifies features of the number of dopaminergic neurons and neuronal complexity, providing a valuable tool for advanced in vitro modeling of PD and testing neurotoxic compounds[40]. Another strategy is to construct brain organoids that exhibit dopaminergic neurons, astrocytes, and microglia. This midbrain-specific organoid is utilized to simulate PD by comparing brain organoids carrying disease-specific mutations with healthy controls, thereby enabling the investigation of neurodegenerative pathophysiology[41]. For instance, brain organoids exposed to PM2.5 exhibited increased neuronal apoptosis and disrupted neurogenesis, indicating that environmental pollutants may have a significant impact on neurodevelopment and could predispose individuals to neurodegenerative diseases such as PD[42].
AD models
This model is typically constructed using genetically modified hiPSCs carrying familial AD mutations[43]. The AD organoids demonstrate characteristic pathological features including amyloid-β (Aβ) deposition, tau pathology, and neurodegeneration. These features are confirmed through various detection methods, including enzyme-linked immunosorbent assays, immunohistochemistry, and western blot analysis[44]. For instance, polyfluoroalkyl substances (PFAS) mixtures have been shown to induce Aβ accumulation and tau phosphorylation in brain organoids, suggesting that PFAS-induced neurotoxicity is associated with AD-like pathology and indicating the significant role of these pollutants in the development of neurodegenerative disorders[45].
HD models
This model is developed using hiPSCs from patients with HD. These cells are differentiated into medium spiny neurons and integrated into brain organoids. HD organoids exhibit calcium signaling dysregulation and elevated stromal interaction molecule 2 (STIM2) expression, which is associated with calcium influx through store-operated calcium channels[46]. Brain organoids can be used to study gene-environment interactions in HD. Thus, researchers can investigate how various environmental factors affect the onset and progression of HD[47].
ASD models
This model is derived from hiPSCs and can recapitulate early human brain development. Studies have shown that ASD-derived organoids often exhibit malformations in the ventricular zones, impaired early neuronal differentiation, and an imbalance in the excitatory–inhibitory neuron ratio, particularly a reduction in GABAergic (GABA, γ-aminobutyric acid) neurons[48,49]. As reported, the exposure of hiPSC-derived brain organoids to diesel particulate matter results in significant epigenetic and functional changes, particularly affecting mitochondrial function and cellular respiration. These disruptions are linked to altered neurodevelopment and may contribute to developmental disorders such as ASD[50].
APPLICATION OF BRAIN ORGANOIDS IN MODELING NEUROTOXICITY OF MNPS
The neurotoxicity of MNPs and related toxic mechanisms
The neurotoxicity of MNPs has been widely manifested in both in vitro and in vivo experiments. MNPs can activate glial cells including microglia and astrocytes, which leads to the release of pro-inflammatory cytokines and causes neuroinflammation and subsequent neuronal damage[51,52]. This process can trigger programmed cell death and necrosis in neuronal cells[53]. The toxic mechanism primarily relates to the oxidative damage and mitochondrial dysfunction caused by MNPs. MNPs induce oxidative stress by generating reactive oxygen species (ROS), which can damage cellular components such as DNA, proteins, and lipids[54]. Studies in vitro have demonstrated that polystyrene (PS)-NPs induce ROS production, activate nuclear factor-κB (NF-κB), and promote tumor necrosis factor α (TNF-α) secretion, leading to necroptosis in brain endothelial cells[55].
Long-term exposure to MNPs may lead to neurodegenerative diseases through mechanisms involving mitochondrial dysfunction, oxidative stress, altered signaling pathways, and impaired neuronal communication[56,57]. For instance, exposure to PS-NPs has been linked to the dysregulation of genes associated with PD and AD[58,59]. A striking animal experiment based on Caenorhabditis elegans (C. elegans) has discovered that NPs exacerbate symptoms similar to those of PD, including dopaminergic neuronal degeneration, locomotor dysfunction, and accumulation of α-synuclein aggregates[13].
However, the processes by which MNPs induce neurotoxicity are complex and influenced by factors such as their size, shape, surface charge, and chemical composition. Further research is needed on the detailed action mechanisms as well as long-term exposure effects of MNPs on the brain, including their developmental and functional impacts[53].
Brain organoids applied in the toxic research of MNPs
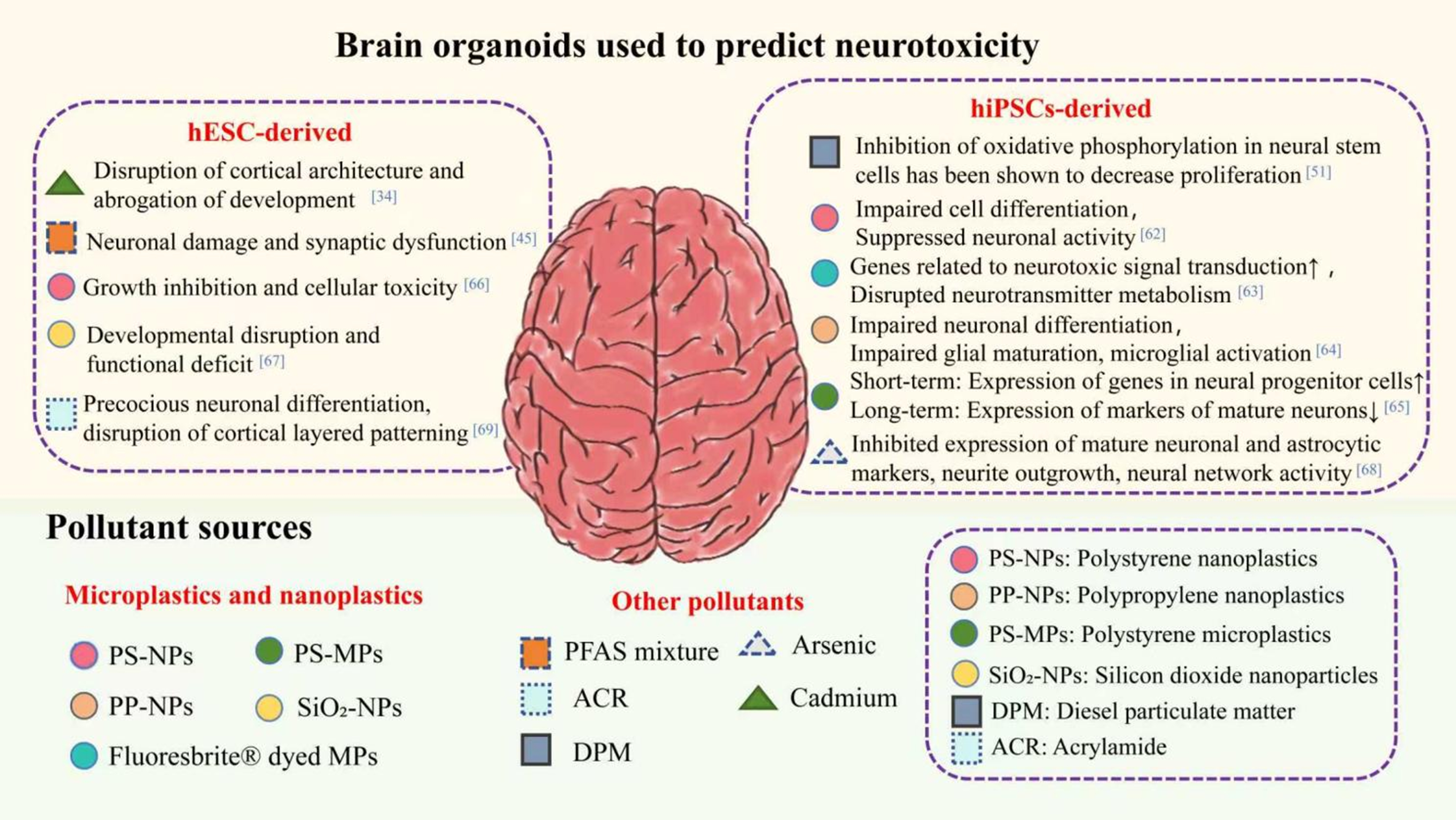
Organoid technology derived from human pluripotent or adult stem cells has broader prospects for predicting the potential health risks of MNPs to humans[60]. In recent years, brain organoids have been introduced into environmental toxicology and have gradually become an emerging tool for elucidating the neurotoxicity of environmental pollutants including MNPs[61]. Most current studies investigating the neurotoxicity of MNPs using brain organoids employ organoids derived from hiPSCs, while a smaller number of studies utilize hESCs. The applications and characteristics of brain organoids derived from these two sources in neurotoxicity studies are systematically summarized in Table 1 and Figure 4.
Figure 4. Application of brain organoids in predicting potential neurotoxic effects of pollutants and related mechanisms. hESC: Human embryonic stem cell; hiPSCs: human-induced pluripotent stem cells.
Application of brain organoid technology in exploring the neurotoxicity of MNPs
| Source | Particle characterization | Exposure duration | Endpoints | ||
| Type | Concentration | Size | |||
| hiPSCs | PS-NPs[62] | 50, 100, 200 ng/mL | 25-60 nm | 8 days | Increase apoptosis; inhibit neural differentiation |
| Fluoresbrite® dyed MPs[63] | 10 mg/mL | 50, 100 nm | 21 days | Down-regulate genes related to neurotoxic signal transduction; disrupt neurotransmitter | |
| PP-NPs[64] | 0, 10, 25, 50 μg/mL | 100 nm | 30 days | Growth inhibition; differentiation defect; impair neuronal differentiation | |
| PS-MPs[65] | 5, 50, 100 μg/mL | 1, 10 μm | 6, 26 days | Up-regulate genes in neural progenitor cells; down-regulate genetic markers of mature neurons | |
| hESCs | PS-NPs[66] | 0.025, 0.05, 0.1 mg/mL | 100 nm | 7, 14 days | Growth inhibition; cellular toxicity |
| SiO2-NPs[67] | 10, 25, 50, 100, 150 µg/mL | 20-30 nm | 10 days | Developmental disruption; functional deficit | |
hiPSCs
The hiPSC-derived brain organoids have been widely applied in exploring the neuro-inflammatory toxicity mechanisms of MNPs. For instance, PS-NPs can infiltrate hiPSC-derived brain organoids, leading to structural and functional damage[62]. Smaller NPs (50 nm) penetrate more deeply into hiPSC-derived cerebral organoids than larger ones (100 nm), which promotes the metabolism of tryptophan along the kynurenine pathway, leading to increased levels of the neurotoxic metabolites kynurenine and 3-hydroxykynurenine, ultimately triggering apoptosis and neural dysfunction[63].
The hiPSC-derived brain organoids have also been used to explore the mechanisms of neurodevelopmental inhibition caused by MNPs. For instance, maternal exposure to polypropylene (PP)-NPs significantly down-regulated the expression of neuronal markers in human brain organoids, inhibiting neuronal differentiation and cell proliferation, and ultimately leading to long-term neurobehavioral deficits in offspring[64]. In addition, exposure to environmentally relevant concentrations of PS-MPs has dual effects on the development of human-derived forebrain cortical spheroids: short-term exposure promotes cell proliferation and neural progenitor gene expression potentially through cell-MP adhesion, whereas long-term exposure induces sustained cellular stress due to particle internalization or extracellular retention, thereby disrupting the normal layered patterning and maturation of the cerebral cortex[65]. The disruption of neural development via the inhibition of key signaling pathways underlies its association with a variety of developmental neurotoxicity-related diseases[68].
hESCs
The application of hESCs in the study of neurotoxicity caused by environmental pollutants and even neurodegenerative diseases has been reported. For instance, cadmium exposure leads to significant neurodevelopmental damage and abnormalities in brain organoids derived from hESCs[34]. Chronic exposure of hESC-derived cerebral organoids to environmentally relevant concentrations of PFAS mixtures induces typical pathological features of AD[45]. Acrylamide (ACR) alters the transcriptomic profile, induces cell apoptosis, and represses neuronal differentiation in cerebral organoids, which may contribute to ACR-induced neurodevelopmental toxicity[69].
Currently, the number of studies reporting the application of hESCs in the toxicity research of MNPs is very limited. Only two publications used hESCs to explore neurotoxic mechanisms of MNPs. NPs infiltrate hESC-derived brain organoids, reducing neural cells and disrupting brain structure, and the neurotoxicity caused by NPs is mediated primarily through suppression of the Wnt/β-catenin pathway[66]. Silica nanoparticles infiltrate hESC-derived midbrain organoids, specifically suppressing dopaminergic neuron differentiation via non-cytotoxic mechanisms involving redox suppression, calcium signaling disruption, and neuroinflammation[67]. Another two publications reported the application of hESCs to establish models of cardiac developmental inhibition and retinal developmental inhibition induced by MNPs[70,71], which has some relations to the neurotoxicity caused by NPs.
Recent studies on brain organoids have shown that NPs reduce neural progenitors/neurons and perturb neurodevelopmental programs[72]. There are studies focusing on cyborg organoid technology, which cleverly integrates stretchable mesh nanoelectronic devices with tissue-like characteristics into 3D organoids, enabling precise mapping of their functions during organoid development[73]. This technology can be applied to study organoids derived from hESCs. In the field of brain organoid research, cyborg organoid technology allows for real-time monitoring of the emergence of neural network activity and the development of complex electrophysiological patterns. The application of artificial intelligence (AI)-driven data simulation analysis is anticipated to establish predictive computational models for the biological toxicity of MNPs[74]. This will help us gain deeper insights into the brain development process and the potential mechanisms behind neurodevelopmental disorders, providing new ideas for the research and treatment of neurological diseases.
Brain organoids have revealed important mechanistic insights and theoretical foundations for potential neurotoxicity, including oxidative stress, inhibition of cell proliferation, and abnormal apoptosis. However, current limitations of brain organoids pose significant challenges for their application in toxicology. Although recent research has advanced, the vascular structures in organoids often lack blood-like flow and do not form a functional BBB[75]. The absence of a functional BBB and neurovascular interactions means that brain organoids may not provide complete data on the penetration, distribution, or effects of MNPs within the brain[76]. The lack of a functional vascular system also leads to inadequate oxygen and nutrient delivery, resulting in hypoxia and limited long-term viability and maturation of organoids[77]. In addition, brain organoids often exhibit incomplete maturation of glial cells, which are crucial for maintaining homeostasis, forming myelin, and supporting neurons[78]. These limitations restrict the ability to accurately simulate the human brain’s response to neurotoxic substances[79]. Furthermore, ethical considerations and the need for regulatory approval hinder the widespread application of hESC-derived brain organoids in neurotoxicity research[80].
CONCLUSION
In summary, brain organoids could emerge as a promising tool for studying the neurotoxicity of MNPs if their technical limitations can be overcome in the future. By providing a more accurate model that better reflects human physiology, they offer significant advantages over traditional approaches. These models address the species-specific limitations of conventional 2D cell cultures and animal models, presenting great potential for a deeper understanding of MNP neurotoxicity. Among brain organoids used to investigate MNP neurotoxicity, hiPSC-derived organoids have been widely employed, whereas hESC-derived organoids remain relatively underutilized. Future research should focus on exploring the potential and unique advantages of hESC-derived organoids in neurotoxicity studies.
However, the interdisciplinary nature of brain organoid technology brings various technical and engineering challenges. Besides the limitations of brain organoids discussed above, recommendations for future research are proposed as follows:
(1) Developing region-specific brain organoids, such as cortical, midbrain, and forebrain organoids. This will help simulate the effects of MNPs on different brain regions and improve understanding of region-specific vulnerabilities.
(2) Developing fetal brain organoids that highly simulate the developmental processes of the human brain. This can accurately simulate the scenario of human fetal brain development exposed to MNPs.
(3) Developing a standardized brain organoid platform, such as tubular organoid chip devices, to enhance reproducibility and scalability. This will make it suitable for a wide range of applications in toxicity testing.
(4) In addition to revealing specific toxicity mechanisms, brain organoid technology can also be systematically applied within the framework of human health risk assessment, further enhancing its utility in modeling diseases caused by environmental pollutants.
DECLARATIONS
Authors’ contributions
Conceptualization, software and writing - original draft preparation: Sun, W.; Zhang, S.
Investigation and data curation: Nie, B.; Cong, J.; Ye, K.
Writing - review and editing: Duan, Z.
All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This study was supported by the Natural Science Foundation of China (Grant Numbers U22A20614, 42277281, and 41807487).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Peller, J. R.; Mezyk, S. P.; Shidler, S.; et al. Facile nanoplastics formation from macro and microplastics in aqueous media. Environ. Pollut. 2022, 313, 120171.
2. Ma, Y. B.; Xie, Z. Y.; Hamid, N.; et al. Recent advances in micro (nano) plastics in the environment: distribution, health risks, challenges and future prospects. Aquat. Toxicol. 2023, 261, 106597.
3. Nie, X.; Xing, X.; Xie, R.; et al. Impact of iron/aluminum (hydr)oxide and clay minerals on heteroaggregation and transport of nanoplastics in aquatic environment. J. Hazard. Mater. 2023, 446, 130649.
4. Enfrin, M.; Hachemi, C.; Hodgson, P. D.; et al. Nano/micro plastics - Challenges on quantification and remediation: a review. J. Water. Process. Eng. 2021, 42, 102128.
5. Sun, A.; Wang, W. X. Human exposure to microplastics and its associated health risks. Environ. Health. 2023, 1, 139-49.
6. Cheng, H.; Duan, Z.; Wu, Y.; et al. Immunotoxicity responses to polystyrene nanoplastics and their related mechanisms in the liver of zebrafish (Danio rerio) larvae. Environ. Int. 2022, 161, 107128.
7. Nihart, A. J.; Garcia, M. A.; El Hayek, E.; et al. Bioaccumulation of microplastics in decedent human brains. Nat. Med. 2025, 31, 1114-9.
8. Junaid, M.; Abbas, Z.; Siddiqui, J. A.; et al. Ecotoxicological impacts associated with the interplay between micro(nano)plastics and pesticides in aquatic and terrestrial environments. TrAC. Trends. Anal. Chem. 2023, 165, 117133.
9. Qiu, X.; Li, L.; Qiu, Q.; et al. Medical exposure to micro(nano)plastics: an exposure pathway with potentially significant harm to human health that should not be overlooked. Sci. Total. Environ. 2024, 957, 177743.
10. Nazeer, N.; Bhargava, A.; Soni, N.; Tiwari, R.; Ratre, P.; Mishra, P. K. Unravelling the molecular dimensions of atmospheric micro(nano)plastics: exploring potential impacts on human health and strategies for detection. Phys. Chem. Earth. 2024, 135, 103604.
11. Xie, J.; Ji, J.; Sun, Y.; Ma, Y.; Wu, D.; Zhang, Z. Blood-brain barrier damage accelerates the accumulation of micro- and nanoplastics in the human central nervous system. J. Hazard. Mater. 2024, 480, 136028.
12. Paing, Y. M. M.; Eom, Y.; Song, G. B.; et al. Neurotoxic effects of polystyrene nanoplastics on memory and microglial activation: insights from in vivo and in vitro studies. Sci. Total. Environ. 2024, 924, 171681.
13. Jeong, A.; Park, S. J.; Lee, E. J.; Kim, K. W. Nanoplastics exacerbate Parkinson’s disease symptoms in C. elegans and human cells. J. Hazard. Mater. 2024, 465, 133289.
14. Wang, Y.; Wang, J.; Cong, J.; et al. Nanoplastics induce neuroexcitatory symptoms in zebrafish (Danio rerio) larvae through a manner contrary to Parkinsonian’s way in proteomics. Sci. Total. Environ. 2023, 905, 166898.
15. Rudroff, T. Artificial intelligence as a replacement for animal experiments in neurology: potential, progress, and challenges. Neurol. Int. 2024, 16, 805-20.
16. Vorhees, C. V.; Williams, M. T.; Hawkey, A. B.; Levin, E. D. Translating neurobehavioral toxicity across species from zebrafish to rats to humans: implications for risk assessment. Front. Toxicol. 2021, 3, 629229.
17. Mazarati, A. Can we and should we use animal models to study neurobehavioral comorbidities of epilepsy? Epilepsy. Behav. 2019, 101, 106566.
18. Majumder, J.; Murphy, W. L. Neural organoids as advanced tools for neurotoxicity modeling. Curr. Res. Toxicol. 2025, 9, 100249.
19. Li, T.; Qin, X.; Ao, Q. Research progress on neural cell culture systems. Curr. Neuropharmacol. 2025, 23, 1518-32.
20. Kaawa-Mafigiri, D.; Ekusai Sebatta, D.; Munabi, I.; Mwaka, E. S. Genetic and genomic researchers’ perspectives on biological sample sharing in collaborative research in Uganda: a qualitative study. J. Empir. Res. Hum. Res. Ethics. 2023, 18, 134-46.
21. Sakaguchi, H. Self-organization and applications of neural organoids. Eur. J. Cell. Biol. 2025, 104, 151496.
22. Zhao, Y.; Wang, T.; Liu, J.; Wang, Z.; Lu, Y. Emerging brain organoids: 3D models to decipher, identify and revolutionize brain. Bioact. Mater. 2025, 47, 378-402.
23. Qian, X.; Song, H.; Ming, G. L. Brain organoids: advances, applications and challenges. Development 2019, 146, dev166074.
24. Di Stefano, J.; Di Marco, F.; Cicalini, I.; et al. Generation, interrogation, and future applications of microglia-containing brain organoids. Neural. Regen. Res. 2025, 20, 3448-60.
25. Nguyen, X. H.; Yoo, J. Current status and future prospects of toxicity assessment using organoids. Toxicol. Res. 2025, 41, 325-33.
26. Kwak, T.; Park, S. H.; Lee, S.; et al. Guidelines for manufacturing and application of organoids: brain. Int. J. Stem. Cells. 2024, 17, 158-81.
27. Vaez Ghaemi, R.; Co, I. L.; McFee, M. C.; Yadav, V. G. Brain organoids: a new, transformative investigational tool for neuroscience research. Adv. Biosyst. 2019, 3, e1800174.
28. Hongen, T.; Sakai, K.; Ito, T.; Qin, X. Y.; Sone, H. Human-induced pluripotent stem cell-derived neural organoids as a novel in vitro platform for developmental neurotoxicity assessment. Int. J. Mol. Sci. 2024, 25, 12523.
29. Cao, Y. The uses of 3D human brain organoids for neurotoxicity evaluations: a review. Neurotoxicology 2022, 91, 84-93.
30. Sun, N.; Meng, X.; Liu, Y.; Song, D.; Jiang, C.; Cai, J. Applications of brain organoids in neurodevelopment and neurological diseases. J. Biomed. Sci. 2021, 28, 30.
31. Zhang, Z.; O’Laughlin, R.; Song, H.; Ming, G. L. Patterning of brain organoids derived from human pluripotent stem cells. Curr. Opin. Neurobiol. 2022, 74, 102536.
32. Wang, M.; Gage, F. H.; Schafer, S. T. Transplantation strategies to enhance maturity and cellular complexity in brain organoids. Biol. Psychiatry. 2023, 93, 616-21.
33. Ji, X. S.; Ji, X. L.; Xiong, M.; Zhou, W. H. Modeling congenital brain malformations with brain organoids: a narrative review. Transl. Pediatr. 2023, 12, 68-78.
34. Kim, S. J.; Jung, W. H.; Choe, M. S.; Jeon, Y. S.; Lee, M. Y. Modeling synucleinopathy using hESC-derived cerebral organoids. Cells 2025, 14, 1436.
35. Wang, Y.; Wang, L.; Guo, Y.; Zhu, Y.; Qin, J. Engineering stem cell-derived 3D brain organoids in a perfusable organ-on-a-chip system. RSC. Adv. 2018, 8, 1677-85.
36. Xu, J.; Wen, Z. Brain organoids: studying human brain development and diseases in a dish. Stem. Cells. Int. 2021, 2021, 5902824.
37. Ao, Z.; Cai, H.; Wu, Z.; et al. Tubular human brain organoids to model microglia-mediated neuroinflammation. Lab. Chip. 2021, 21, 2751-62.
38. Tian, C.; Cai, H.; Ao, Z.; et al. Engineering human midbrain organoid microphysiological systems to model prenatal PFOS exposure. Sci. Total. Environ. 2024, 947, 174478.
39. Babu, H. W. S.; Kumar, S. M.; Kaur, H.; Iyer, M.; Vellingiri, B. Midbrain organoids for Parkinson’s disease (PD) - a powerful tool to understand the disease pathogenesis. Life. Sci. 2024, 345, 122610.
40. Monzel, A. S.; Hemmer, K.; Kaoma, T.; et al. Machine learning-assisted neurotoxicity prediction in human midbrain organoids. Parkinsonism. Relat. Disord. 2020, 75, 105-9.
41. Smits, L. M.; Schwamborn, J. C. Midbrain organoids: a new tool to investigate Parkinson’s disease. Front. Cell. Dev. Biol. 2020, 8, 359.
42. Han, Y.; Yu, Z.; Chen, Y.; et al. PM2.5 induces developmental neurotoxicity in cortical organoids. Environ. Pollut. 2024, 361, 124913.
43. Kim, Y.; Kim, H.; Cho, B.; et al. Modeling APOE ε4 familial Alzheimer’s disease in directly converted 3D brain organoids. Front. Aging. Neurosci. 2024, 16, 1435445.
44. Choe, M. S.; Yeo, H. C.; Kim, J. S.; et al. Simple modeling of familial Alzheimer’s disease using human pluripotent stem cell-derived cerebral organoid technology. Stem. Cell. Res. Ther. 2024, 15, 118.
45. Lu, S.; Zhu, X.; Zeng, P.; et al. Exposure to PFOA, PFOS, and PFHxS induces Alzheimer’s disease-like neuropathology in cerebral organoids. Environ. Pollut. 2024, 363, 125098.
46. Latoszek, E.; Czeredys, M. Molecular components of store-operated calcium channels in the regulation of neural stem cell physiology, neurogenesis, and the pathology of Huntington’s disease. Front. Cell. Dev. Biol. 2021, 9, 657337.
47. Smirnova, L.; Hartung, T. The promise and potential of brain organoids. Adv. Healthc. Mater. 2024, 13, e2302745.
48. Hong, Y. J.; Lee, S. B.; Choi, J.; Yoon, S. H.; Do, J. T. A simple method for generating cerebral organoids from human pluripotent stem cells. Int. J. Stem. Cells. 2022, 15, 95-103.
49. de Jong, J. O.; Llapashtica, C.; Genestine, M.; et al. Cortical overgrowth in a preclinical forebrain organoid model of CNTNAP2-associated autism spectrum disorder. Nat. Commun. 2021, 12, 4087.
50. Bilinovich, S. M.; Uhl, K. L.; Lewis, K.; et al. Integrated RNA sequencing reveals epigenetic impacts of diesel particulate matter exposure in human cerebral organoids. Dev. Neurosci. 2020, 42, 195-207.
51. Chang, X.; Li, J.; Niu, S.; Xue, Y.; Tang, M. Neurotoxicity of metal-containing nanoparticles and implications in glial cells. J. Appl. Toxicol. 2021, 41, 65-81.
52. Adamiak, K.; Sidoryk-Węgrzynowicz, M.; Dąbrowska-Bouta, B.; Sulkowski, G.; Strużyńska, L. Primary astrocytes as a cellular depot of polystyrene nanoparticles. Sci. Rep. 2025, 15, 6502.
53. Gong, J. Y.; Holt, M. G.; Hoet, P. H. M.; Ghosh, M. Neurotoxicity of four frequently used nanoparticles: a systematic review to reveal the missing data. Arch. Toxicol. 2022, 96, 1141-212.
54. Wang, Y.; Cong, J.; Kong, X.; et al. Positively charged nanoplastics destruct the structure of the PCK1 enzyme, promote the aerobic gycolysis pathway, and induce hepatic tumor risks. Environ. Sci. Technol. 2025, 59, 3013-23.
55. Shan, S.; Zhang, Y.; Zhao, H.; Zeng, T.; Zhao, X. Polystyrene nanoplastics penetrate across the blood-brain barrier and induce activation of microglia in the brain of mice. Chemosphere 2022, 298, 134261.
56. Urani, C.; Barbieri, R.; Alloisio, S.; Tesauro, M. From the environment to molecular interactions of nanoplastics: unraveling the neurotoxic impacts and the implications in neurodegenerative processes. Appl. Sci. 2024, 14, 7280.
57. Tan, C.; Kang, C.; Liu, P.; Sun, Y.; Jin, H. Polystyrene nanoplastics exposure trigger cognitive impairment mitigated by luteolin modulated glucose-6-phosphate dehydrogenase/glutathione-dependent pathway. J. Hazard. Mater. 2025, 493, 138404.
58. Bai, H.; Wu, Y.; Li, H.; et al. Cerebral neurotoxicity of amino-modified polystyrene nanoplastics in mice and the protective effects of functional food Camellia pollen. Sci. Total. Environ. 2024, 912, 169511.
59. Jeong, H.; Shanmugiah, J.; Jang, J.; Lee, D. H.; Choi, J.; Kim, J. S. Nanoplastics cause an increased risk of Parkinson’s disease compared to microplastics at environmental exposure levels. J. Hazard. Mater. Adv. 2025, 20, 100941.
60. Cong, J.; Wu, J.; Fang, Y.; et al. Application of organoid technology in the human health risk assessment of microplastics: a review of progresses and challenges. Environ. Int. 2024, 188, 108744.
61. Abdulla, A.; Yan, H.; Chen, S.; et al. A multichannel microfluidic device for revealing the neurotoxic effects of Bisphenol S on cerebral organoids under low-dose constant exposure. Biosens. Bioelectron. 2025, 267, 116754.
62. Tao, M.; Wang, C.; Zheng, Z.; et al. Nanoplastics exposure-induced mitochondrial dysfunction contributes to disrupted stem cell differentiation in human cerebral organoids. Ecotoxicol. Environ. Saf. 2024, 285, 117063.
63. Park, S. B.; Jo, J. H.; Kim, S. S.; et al. Microplastics accumulation induces kynurenine-derived neurotoxicity in cerebral organoids and mouse brain. Biomol. Ther. 2025, 33, 447-57.
64. Huang, F.; You, H.; Tang, X.; et al. Early-life exposure to polypropylene nanoplastics induces neurodevelopmental toxicity in mice and human iPSC-derived cerebral organoids. J. Nanobiotechnology. 2025, 23, 474.
65. Hua, T.; Kiran, S.; Li, Y.; Sang, Q. A. Microplastics exposure affects neural development of human pluripotent stem cell-derived cortical spheroids. J. Hazard. Mater. 2022, 435, 128884.
66. Chen, S.; Chen, Y.; Gao, Y.; et al. Toxic effects and mechanisms of nanoplastics on embryonic brain development using brain organoids model. Sci. Total. Environ. 2023, 904, 166913.
67. Kim, S.; Lee, Y.; Lee, H. A.; Lee, M. O. SiO2 nanoparticles disrupt neurodevelopmental processes in human midbrain organoids in a redox-suppressed, non-cytotoxic manner. J. Hazard. Mater. 2026, 501, 140670.
68. Wu, X.; Kreutz, A.; Dixon, D.; Tokar, E. J. Engineering human cerebral organoids to explore mechanisms of arsenic-induced developmental neurotoxicity. Toxicol. Appl. Pharmacol. 2025, 496, 117230.
69. Bu, Q.; Huang, Y.; Li, M.; et al. Acrylamide exposure represses neuronal differentiation, induces cell apoptosis and promotes tau hyperphosphorylation in hESC-derived 3D cerebral organoids. Food. Chem. Toxicol. 2020, 144, 111643.
70. Li, J.; Weng, H.; Liu, S.; et al. Embryonic exposure of polystyrene nanoplastics affects cardiac development. Sci. Total. Environ. 2024, 906, 167406.
71. Li, M.; Gao, X.; Lan, Y.; et al. Revealing the neurodevelopmental toxicity of face mask-derived microplastics to humans based on neural organoids. J. Hazard. Mater. 2025, 492, 138084.
72. Bhattacharyya, S.; Greer, M. L.; Salehi, M. Impact of micro- and nanoplastics exposure on human health: focus on neurological effects from ingestion. Front. Public. Health. 2025, 13, 1681776.
73. Lin, Z.; Wang, W.; Liu, R.; et al. Cyborg organoids integrated with stretchable nanoelectronics can be functionally mapped during development. Nat. Protoc. 2025, 20, 2528-59.
74. Huang, H.; Hou, J.; Xi, B. The need to implement innovative technologies to advance research on the biotoxicity of micro- and nanoplastics. Environ. Health. 2025, 3, 1429-37.
75. Kistemaker, L.; van Bodegraven, E. J.; de Vries, H. E.; Hol, E. M. Vascularized human brain organoids: current possibilities and prospects. Trends. Biotechnol. 2025, 43, 1275-85.
76. Depla, J. A.; Mulder, L. A.; de Sá, R. V.; et al. Human brain organoids as models for central nervous system viral infection. Viruses 2022, 14, 634.
77. Jeong, E.; Choi, S.; Cho, S. W. Recent advances in brain organoid technology for human brain research. ACS. Appl. Mater. Interfaces. 2023, 15, 200-19.
78. Makrygianni, E. A.; Chrousos, G. P. From brain organoids to networking assembloids: Implications for neuroendocrinology and stress medicine. Front. Physiol. 2021, 12, 621970.
79. Kim, S. H.; Chang, M. Y. Application of human brain organoids-opportunities and challenges in modeling human brain development and neurodevelopmental diseases. Int. J. Mol. Sci. 2023, 24, 12528.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].