Olfactory-inspired nanofluidic sensor: molecular recognition and transport in confined space
Abstract
The olfactory system of biological intelligence, which enables accurate discernment of vital cues within complex odor mixtures, provides inspiration for the development of rapid and sensitive sensors. Olfactory perception is mediated by ionic fluxes that convert chemical information into electrical signals. Olfactory-inspired sensors utilize molecular recognition and transport in confined spaces to achieve macroscopic readouts of chemical information, holding significant potential to balance sensitivity, selectivity, and stability - long-standing challenges in traditional sensing. These sensors are expected to enable miniaturization, low power consumption, and reliable detection in real scenarios characterized by complex compositions, strong background interference, and extremely low target concentrations. This review provides development strategies for olfactory-inspired nanofluidic sensors and summarizes their bioinspired mechanisms, fabrication methods, and sensing applications. Finally, it highlights key challenges and potential future directions essential for advancing olfactory-inspired sensing technologies.
Keywords
INTRODUCTION
Chemical sensing, transforming complex and abstract chemical signals into concrete and processable information, extends the limits of human perception and serves as the base for the fields of bioscience, food safety, public safety, environmental protection, medical diagnostics, etc. For chemical sensing, the key technical bottleneck remains the balance of selectivity, sensitivity, and stability. Furthermore, significant attention must be given to addressing interference when facing complex environments and achieving dynamic sensing in situ. Olfactory perception of living organisms offers insights for addressing the above challenges. Dogs can track a single target within complex odor backgrounds; moths can detect pheromones from kilometers away; and humans can effortlessly distinguish and remember thousands of scents. Through sophisticated strategies, organisms resolve fundamental trade-offs in molecular recognition, including selectivity versus broad-spectrum response, stability versus sensitivity, and power consumption versus intelligence.
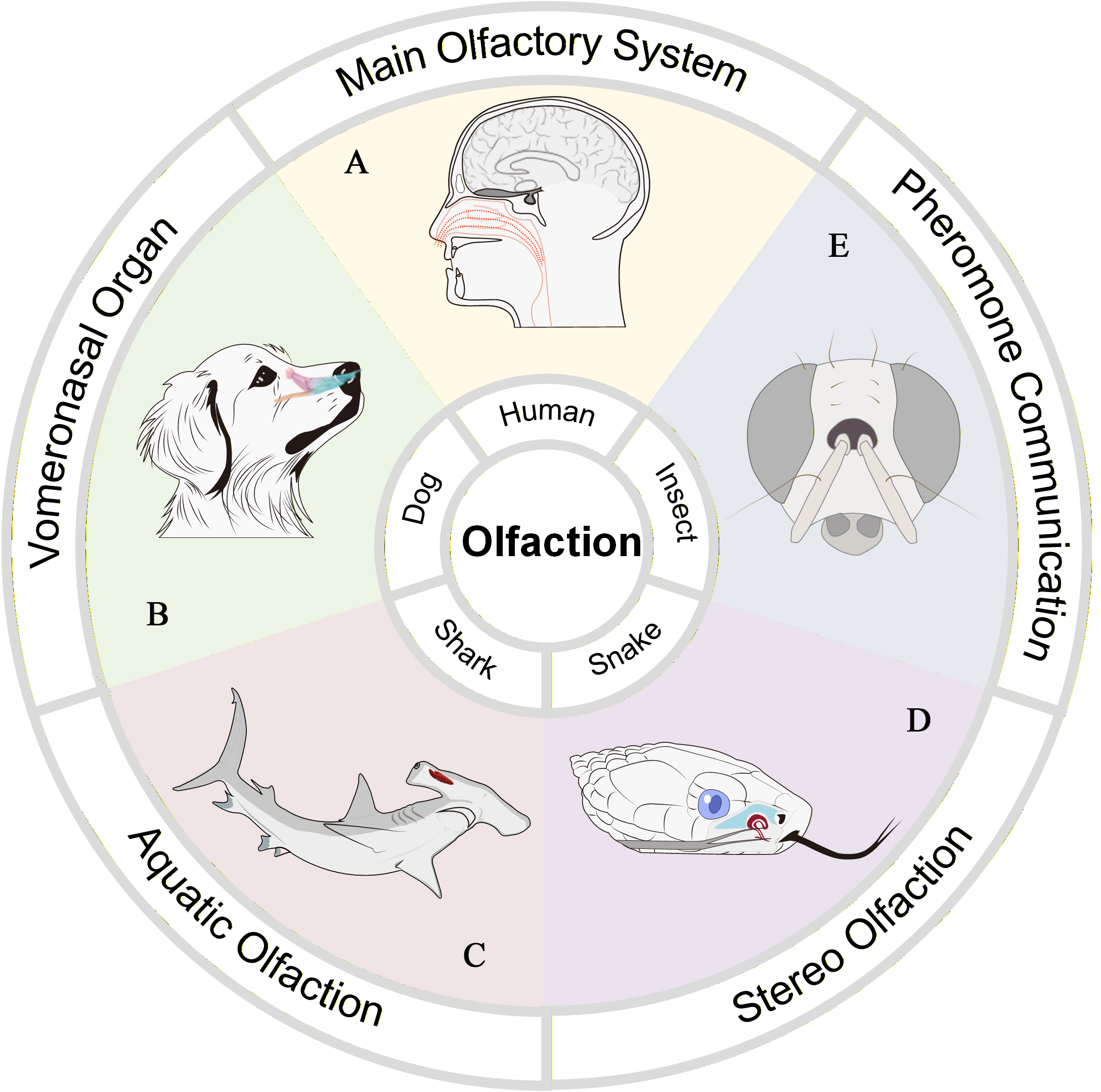
Olfactory systems exhibit significant evolutionary differentiation in structure, sensitivity, and functional specialization through adaptation to different environmental conditions, behavioral requirements, and ecological niches[1,2] [Figure 1]. As human ancestors came to rely more heavily on vision and social intelligence during evolution, the significance of olfaction correspondingly diminished, with the consequence that both the olfactory organs and the repertoire of olfactory receptor genes underwent simplification. Human olfaction[3,4] primarily relies on the neural pathway of the main olfactory system [Figure 1A], which is essential for detecting the vast majority of environmental odors. This system originates in the olfactory epithelium located at the roof of the nasal cavity, where olfactory sensory neurons extend cilia into the nasal mucus. When odorant molecules bind to receptors on these cilia, an electrical signal is triggered and transmitted via the olfactory nerve directly to the olfactory bulb at the base of the brain. From the olfactory bulb, information is primarily relayed to the primary olfactory cortex, which in turn projects rapidly to brain regions closely associated with memory and emotion[5]. In many mammals, there exists an accessory olfactory system known as the vomeronasal organ (VNO) (also called Jacobson’s organ). It is primarily responsible for detecting non-volatile molecules and plays a key role in regulating reproductive and social behaviors. However, genetic and anatomical evidence indicates that the human VNO largely degenerates after the fetal stage. Canines possess the functional VNO[6,7], which lies along the right-cephalic side of the septal organ (SO), is symmetrical on both sides and functions as an additional site for odor detection. Its neurons do not connect to the main olfactory bulb (MOB); instead, signals are transmitted via the separate vomeronasal nerve directly to the accessory olfactory bulb (AOB) in the brain [Figure 1B]. Aquatic organisms[8-10] have evolved to be well-adapted to their underwater tracking environments [Figure 1C]. They detect water-soluble molecules via olfactory epithelia located between inflow and outflow nostrils. For example, hammerhead sharks have evolved paired olfactory organs[9,10]. The flow within this system begins at the anterior nostril, moves through branching interlamellar channels, collects into a central excurrent channel or gallery, and is finally expelled through the posterior nostril. Both nostrils are located on the anterior margin of the head, with the olfactory epithelium situated between them. The wide separation of the nostrils suggests that hammerhead sharks may more effectively localize odor sources through klinotaxis. Furthermore, other species have evolved more specialized olfactory mechanisms. Snakes[11], for example, employ stereo olfaction by using a forked tongue to collect chemical samples from different spatial points [Figure 1D], which are then analyzed by the VNO for spatial tracking. The snake’s tongue[12], devoid of taste buds, is a specialized chemosensory organ that samples chemical cues via rapid flicking motions from both air and substrates, and delivers the collected chemicals to the VNO. The two tongue tips can sample independently from slightly different spatial points, achieving spatial olfactory analysis. Insects[13,14] rely on highly sensitive antennae covered with olfactory sensilla densely packed with receptor proteins specialized for pheromone recognition [Figure 1E]. This forms an efficient chemical communication system crucial for mating, trail marking, alarm signaling, and coordinating group activities. Organisms have evolved highly sensitive and selective olfactory systems through adaptation, with diverse evolutionary strategies effectively addressing varied environmental constraints.
Figure 1. Different olfactory patterns. (A) The main olfactory system of humans[3]; (B) The vomeronasal organ of a dog. This figure is quoted with permission from MDPI[7]; (C) The aquatic olfaction of sharks. This figure is quoted with permission from The PLOS One[10]; (D) The stereo olfaction of snakes; (E) The pheromone communication of insects.
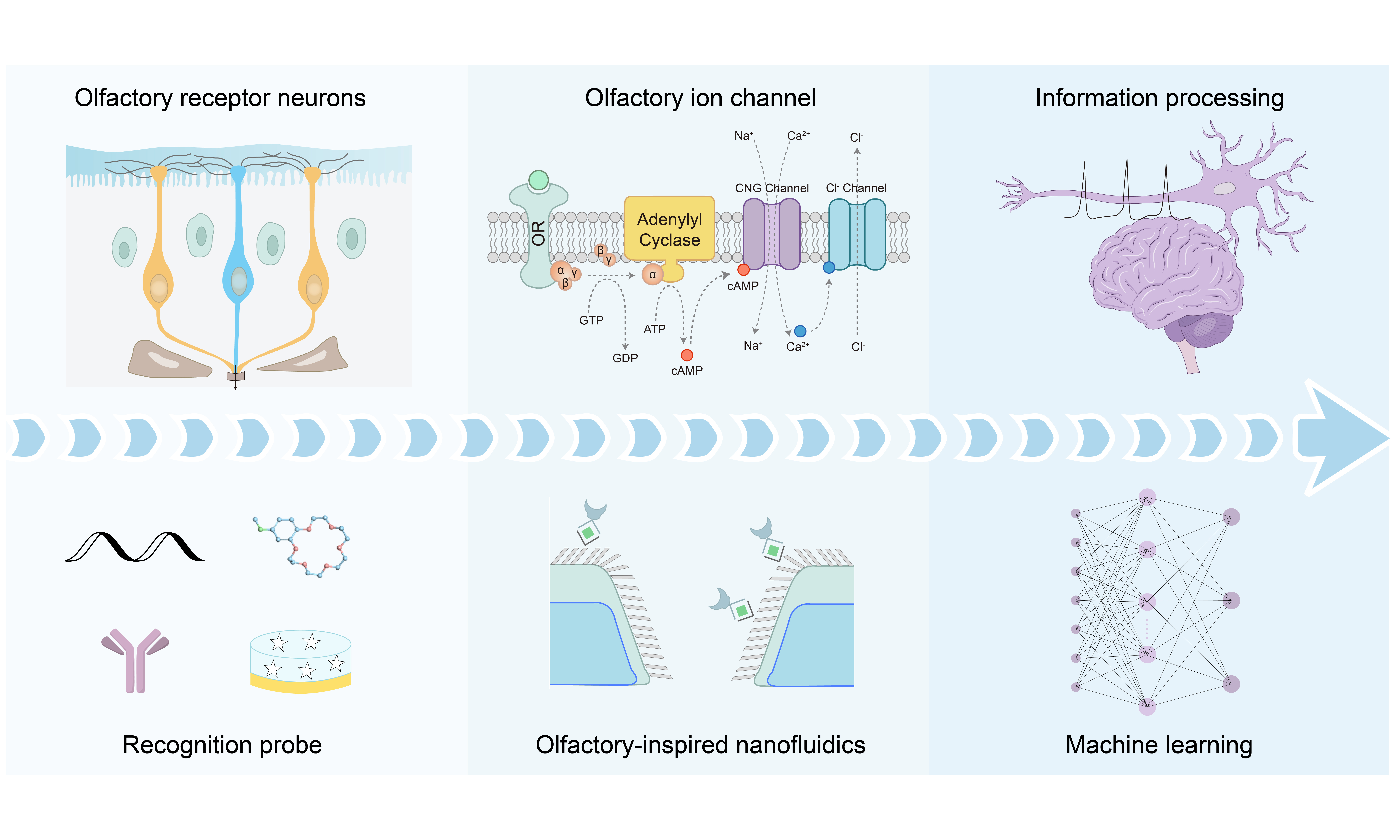
Olfactory-inspired sensing technology involves learning in terms of appearance, mechanism, structure, and signal processing from organisms. Its scope of detection is not limited to odor molecules but extends to the recognition and differentiation of substances in various complex states. The diverse olfactory types and the structure of olfactory receptors in organisms offer substantial inspiration for the design of olfactory-inspired sensors. The main olfactory system’s odor-distinguishing receptors and layered information encoding inspire cross-reactive sensor arrays, with memristors advancing integrated signal processing[15]. The VNO’s pheromone detection mechanism, which requires close contact for large, polar signaling molecules, broadens olfactory-inspired sensor design for liquid-phase detection of large biomolecules and highly polar compounds in bodily fluids or aqueous samples. Aquatic organisms such as hammerhead sharks, with specialized olfactory flow architectures, inspire microfluidic-integrated sensors that boost response speed and measurement consistency. Snakes’ stereo olfaction enables spatial olfactory analysis, inspiring differential sensing arrays for three-dimensional (3D) substance detection. Insects’ high-efficiency pheromone communication, featuring rapid, highly specific receptor responses, motivates the design of fast, selective sensors with engineered recognition elements, ideal for trace screening of drugs, explosives and disease biomarkers. The olfactory mechanism follows a shared logic of signal transduction[13]: odorant molecules bind to specific receptors, triggering ion channel opening and membrane depolarization. Molecular recognition and ion transport through protein channels embedded in the membrane are the critical bridge between chemical and electrical signals. The keys to olfactory-inspired sensing lie in the design of recognition probes and the functionalization of confined space for controllable ion transport. Nanofluidics sensors offer a paradigm for olfactory-inspired sensing that aligns more closely with the essence of biological olfactory perception. Nanofluidics is the discipline that explores the principles of mass transport in the nanoconfined spaces. Nanochannels and nanopores are the key building blocks for fabricating nanofluidic sensors. Due to the high surface-area-to-volume ratio, significant surface charge, and overlapping electric double layers, fluids in confined spaces exhibit fundamentally distinct behaviors compared to those at the macroscopic scale[16-18]. These confined channels possess selectivity, gating and rectification properties, thereby enabling precise monitoring of microenvironmental changes at the microscopic level. The emergence of a growing number of olfactory-inspired sensors drives the need for researchers to fabricate diverse functional nanofluidics. To address the limitations of traditional nanofluidic sensors (lack of specific recognition capability) and conventional bioinspired sensors (poor stability caused by biomolecular inactivation), olfactory-inspired nanofluidic sensors have been developed as a promising sensing platform, which integrates the superior stability of nanofluidic sensors with the specific recognition capability of bioinspired sensors.
OLFACTORY-INSPIRED NANOFLUIDIC SENSOR
Olfactory receptor and signaling pathways in vertebrates and insects
Olfaction is crucial for organismal survival. Over more than 600 million years[19] of independent evolution, the olfactory systems of vertebrates and insects have developed different molecular mechanisms to meet distinct ecological demands.
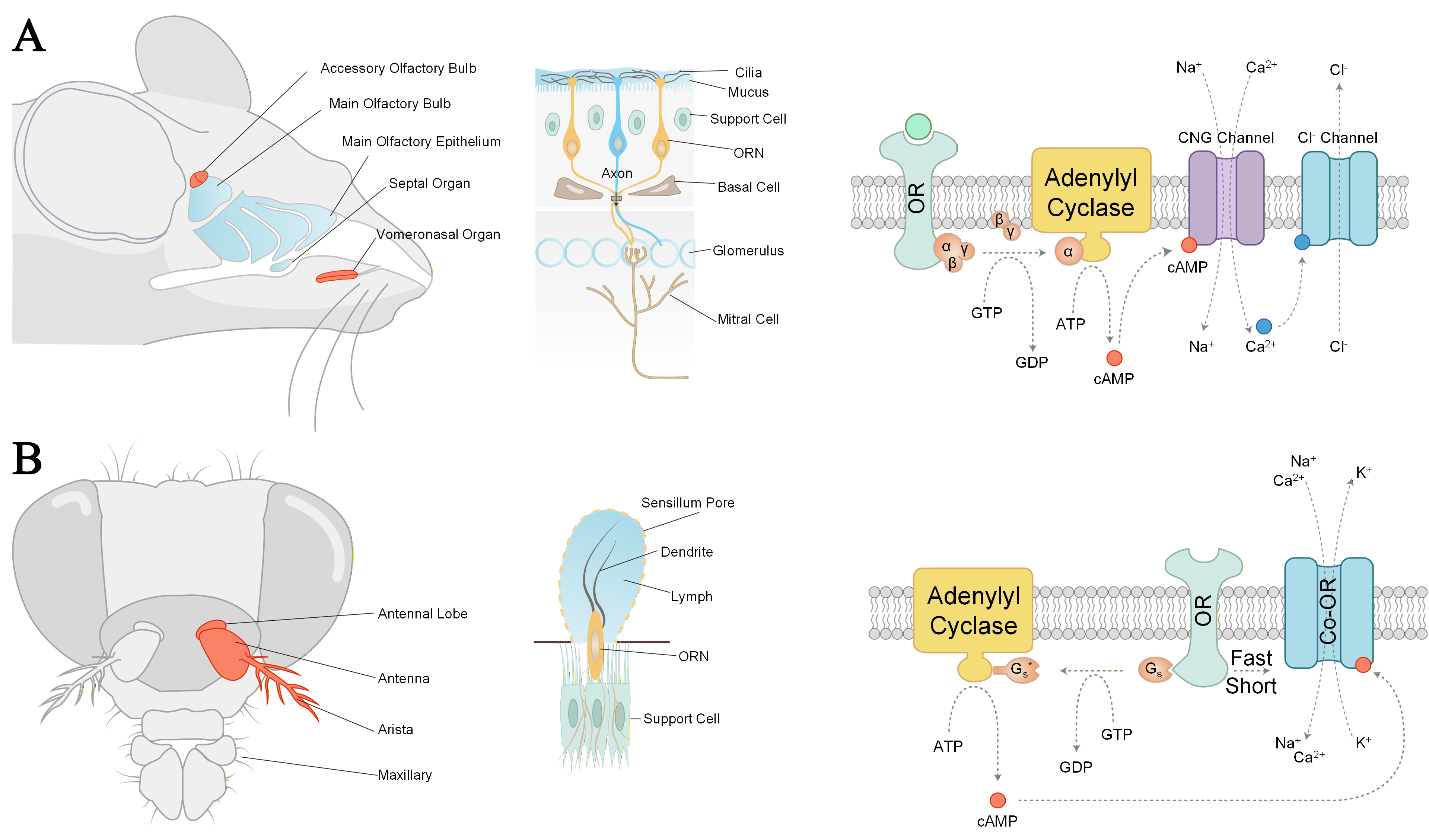
The main olfactory system [Figure 2A] is responsible for detecting a broad spectrum of environmental odors. This system originates in the olfactory epithelium located at the roof of the nasal cavity, where olfactory sensory neurons extend cilia into the nasal mucus[5]. When odorant molecules bind to receptors on these cilia, an electrical signal is triggered and transmitted via the olfactory nerve directly to the olfactory bulb at the base of the brain. From the olfactory bulb, information is primarily relayed to the primary olfactory cortex, which in turn projects rapidly to brain regions closely associated with memory and emotion, such as the amygdala and hippocampus[5]. The activation of most olfactory receptor neurons (ORNs) involves a canonical cyclic adenosine monophosphate (cAMP) signaling pathway[13]. Ligand-bound receptors activate a specific G protein. A single activated membrane receptor can, in turn, activate dozens of G protein molecules, each of which stimulates an adenylyl cyclase molecule[5]. This enzyme, primarily Adenylate Cyclase type III (ACIII), catalyzes the conversion of abundant intracellular adenosine triphosphate (ATP) to cAMP, thereby amplifying and integrating the initial binding event. The resulting cAMP directly activates cyclic nucleotide-gated (CNG) channels, allowing the influx of cations such as Na+ and Ca2+[21]. An inactive ORN typically maintains a resting membrane potential of approximately -65 mV. The opening of CNG channels and the subsequent influx of Na+ and Ca2+ depolarize the cell, making the interior less negative. Once the depolarization reaches a threshold, an action potential is generated. This action potential then transports along the axon to synapse with secondary neurons in the olfactory bulb. Notably, the Ca2+ entering through CNG channels activates nearby chloride channels. ORNs maintain an unusually high intracellular Cl- concentration, so the activation of these chloride channels results in Cl- efflux. This efflux leaves a net positive charge on the membrane, further depolarizing the cell and thereby enhancing the strength of the excitatory response.
Figure 2. Different olfactory receptor structures and signaling pathways in vertebrates and insects. (A) The vertebrate nasal cavity (left) contains several olfactory subsystems: the main olfactory epithelium (MOE), the SO, the VNO, the MOB and the AOB. ORNs in the MOE (middle) have cilia that extend into the mucus. Signal transduction in mammalian ORNs (right)[13,20]; (B) In insects, olfaction occurs in the antenna and the maxillary palp which are covered with the sensilla (left). Insect ORNs share morphological similarities with vertebrate ORNs (middle). Ionotropic mechanisms for olfaction in insects (right)[13,20]. ORN: Olfactory receptor neuron; OR: olfactory receptor; cAMP: cyclic adenosine monophosphate; GTP: guanosine triphosphate; GDP: guanosine diphosphate; SO: septal organ; VNO: vomeronasal organ; MOB: main olfactory bulb; AOB: accessory olfactory bulb.
Vertebrates employ a modulatory pathway centered on G protein-coupled receptors and the second messenger cAMP, achieving high sensitivity and discrimination. In contrast, insects have evolved ligand-gated ion channel receptors[13], establishing a direct chemical-to-electrical signal conversion mechanism that enables rapid behavioral responses on a millisecond timescale [Figure 2B]. Before binding to odorant receptors on the cilia, odor molecules must first pass through the sensillum pores and the lymph. Given that the lymph is water-soluble to better accommodate ion transmembrane transport, odorant binding proteins present within it assist in the transport of certain hydrophobic odor molecules. For hydrophilic odor molecules, a type of chemosensory protein facilitates their delivery close to the corresponding receptors, thereby preventing a decrease in response sensitivity due to low concentration resulting from diffusion. In many insects, odorant detection involves a dual-pathway mechanism[22]. At low odorant concentrations, binding to the olfactory receptor triggers a G protein-coupled cascade that synthesizes cAMP. This second messenger then gates the ion channel formed by Or83b (a universal co-receptor), providing essential signal amplification for high sensitivity. At high odorant concentrations, a direct, fast ionotropic gating of the receptor complex is predominantly activated, enabling rapid neural responses[13]. Despite their apparent diversity, olfactory mechanisms share a common logic of signal transduction. Odorant molecules are selectively transported to olfactory receptors and, upon specific binding, trigger the opening of ion channels in the cell membrane. This leads to membrane depolarization and the generation of transmissible and recognizable electrical signals to the brain. Ionic currents serve as the critical bridge between chemical signals and electrical signals, playing an essential role in molecular sensing.
Olfactory-inspired nanofluidic sensor
The key to achieving olfactory-inspired nanofluidic sensing is regulation of molecular recognition and mass transport in confined spaces. Confined spaces, which mimic the nanoscale microenvironments of biological olfactory systems, play a critical role in enhancing the accuracy and efficiency of these two processes: molecular recognition enables the specific binding between target analytes and functional sites, while mass transport governs the efficient diffusion, adsorption, and desorption of target molecules between the bulk phase and the recognition sites. Olfactory-inspired nanofluidic sensing aims to replicate the extraordinary capabilities of biological olfactory systems, such as high sensitivity to trace odor molecules, excellent specificity for distinguishing structurally similar analytes, and rapid response-recovery cycles. By fabricating confined nanochannels and functionalizing their interfaces to regulate transport, nanofluidic devices can achieve the olfactory sensing capabilities of biological systems. The theoretical foundation has been established to a certain extent[23]. The following discussion will focus on material design, nanoarchitecture design, and the optimization of recognition mechanisms and recognition probes.
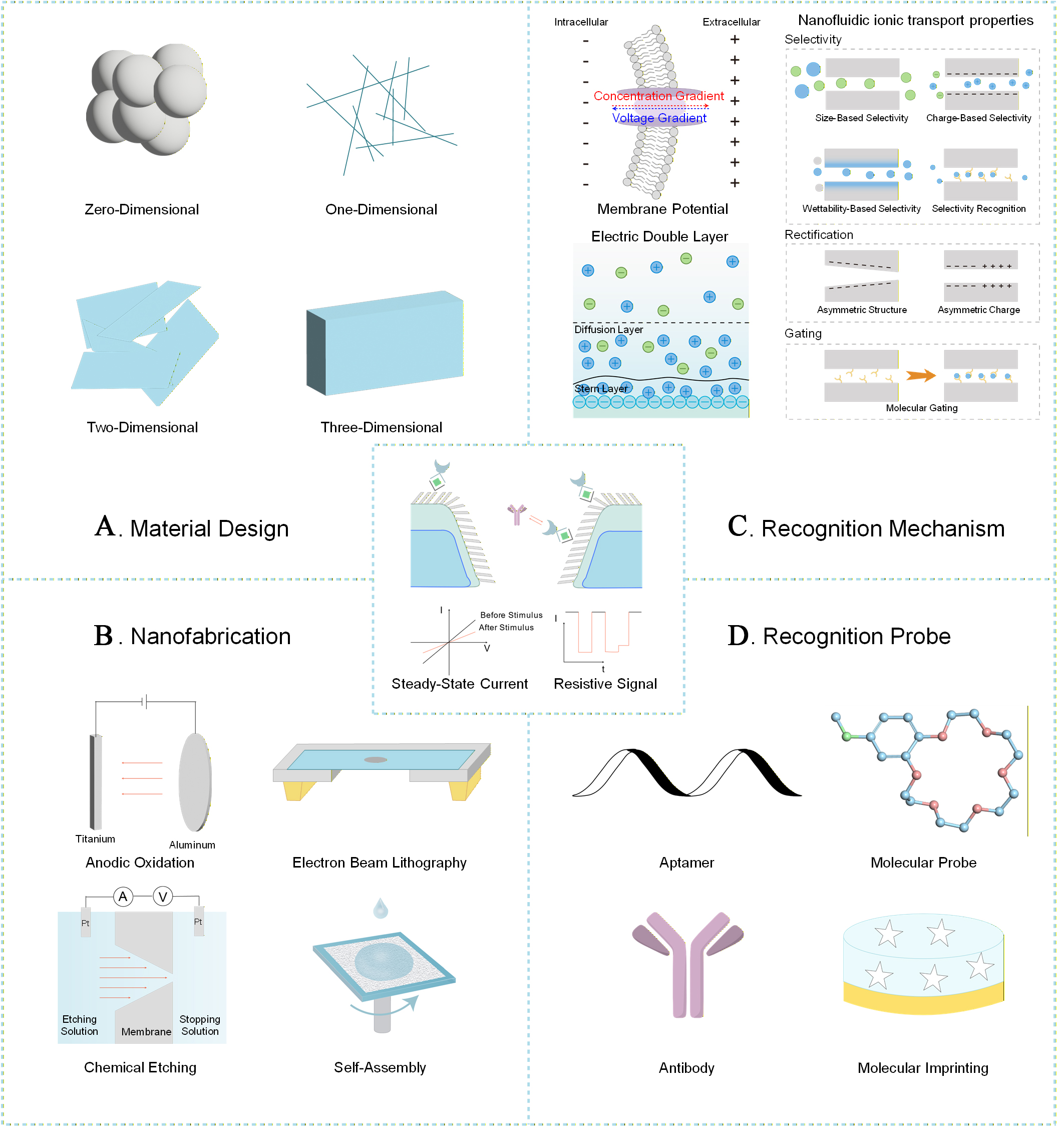
The fabrication of these nanofluidic devices relies on the convergence of dimensional material design and nanofabrication technologies. The engineered nanochannels and nanopores can thereby modulate ion transport properties akin to those of biological systems[24]. From the perspective of material design[25], zero-dimensional (0D) nanoparticles[26] or quantum dots[27] are often employed as functional sites. They can be anchored to the channel interior via surface modifications to regulate local charge or introduce specific recognition sites. One-dimensional (1D) nanowires[28] or nanotubes can serve either as sacrificial templates for channel fabrication or as transport channels, offering unique confinement effects. Two-dimensional (2D) materials[29] are notable for their atomic-level thickness and tunable surface properties. 3D frameworks[30] or interconnected networks are designed to construct biomimetic, high-throughput complex transport systems [Figure 3A]. Figure 3B illustrates typical fabrication methods for solid-state nanochannels and nanopores to precisely optimize three critical structural parameters: channel geometry (including shape, aspect ratio, and spatial distribution), pore size (ranging from sub-nanometers to hundreds of nanometers), and membrane thickness. Among these core techniques, electron-beam lithography[31] is renowned for its ultra-high spatial resolution, which enables the fabrication of nanochannels and nanopores with well-defined, complex geometric configurations. This precise patterning capability is crucial for controlling channel geometry and ensuring consistent performance across fabricated devices. Chemical etching[32], by contrast, offers remarkable flexibility and scalability. By adjusting key parameters such as etching time, temperature, and reagent concentration, researchers can fine-tune pore size and precisely control channel depth, making it suitable for large-scale fabrication of nanopores with uniform dimensions. Anodic oxidation[33] is particularly effective for constructing ordered arrays of solid-state nanochannels, typically performed on metal substrates (e.g., aluminum, titanium, or tantalum) to induce the formation of a porous oxide layer through electrochemical reactions; the thickness of the oxide membrane (and thus the channel length) can be precisely regulated by modulating the anodization voltage and duration, which is beneficial for applications requiring uniform fluid transport or molecular sieving. Nanoscale self-assembly[34] provides a low-cost, bottom-up approach for fabricating nanochannels and nanopores, leveraging the intrinsic self-organizing behavior of nanomaterials to spontaneously form ordered nanostructures without the need for complex lithographic equipment.
Figure 3. Fundamentals of nanofluidic sensor. (A) Material design; (B) Nanofabrication; (C) Recognition mechanism; (D) Recognition probe.
Sensing technologies based on nanofluidics rely on the unique properties of matter at the nanoscale. As biological perception, which is fundamentally based on an electrical potential imbalance across the cell membrane, solid-state nanochannels/pores also rely on this underlying principle. At charged solid-liquid interfaces, an electrical double layer (EDL) is typically present[35,36], which confers selectivity, rectification, and gating properties to nanofluidics. Nearly two centuries of theoretical advancement have significantly enriched our understanding of interfacial charge dynamics. Stern proposed the Gouy-Chapman-Stern (GCS) model in 1924, dividing the EDL into a compact Stern layer and an outer diffuse layer. The Stern layer includes the inner Helmholtz plane (IHP) formed by specifically adsorbed ions, of specifically adsorbed ions and the outer Helmholtz plane (OHP) of solvated ions held by electrostatic attraction. Beyond the Stern layer, the diffuse layer extends into the bulk, with ion distributions governed by a balance of electrostatic and entropic forces[37]. This classical model effectively describes charge behavior at conductive solid-liquid interfaces and can be tuned electrochemically. The EDL forms due to electrostatic interaction with electrolyte ions. In nanochannels with dimensions comparable to the Debye length, EDL overlap leads to a surface-charge-dominated transport regime filled with net charge, which causes their distinctive ionic phenomena. The thickness of the EDL is approximately equal to the Debye length (λD)[35]:
where
Confined nanochannels, as a core component of nanofluidic sensing systems, have attracted extensive attention due to their unique confinement effect, which can significantly amplify intermolecular interactions. Specifically, the interactions between the inner surface of confined nanochannels and target molecules or ions can be flexibly regulated by tailoring the physicochemical properties of the channel interior, including surface charge density, surface hydrophilic-hydrophobic property and channel gating behavior. Such regulation further enables precise control over ion transport and mass transfer within the channels, which can be converted into reproducible electrical signals (e.g., conductance, current, voltage, and impedance) or transport signals (e.g., ion/molecule transport rate, flux, and selectivity) for the quantitative detection of target analytes. The unique properties of confined spaces, including selectivity, rectification, and gating properties, can effectively reflect and amplify molecular recognition events [Figure 3C]. Selectivity[38] of nanofluidics is defined as the ability to permit the passage of specific ions or molecules through a confined space while excluding others. This property can be tuned through the physical dimensions of the pores and the design of their surface functionalization, thereby enabling molecular sieving based on size, charge, wettability, and specificity. Ionic current rectification[39] refers to the directional asymmetry of ion transport through nanochannels, achieved by introducing asymmetry in either the channel geometry or the surface charge distribution. The binding of target molecules alters the local charge density or geometry, thereby significantly modulating the rectification characteristics. This mechanism effectively amplifies a chemical binding event into a readily detectable change in the electrical signal, enhancing detection sensitivity. Ion-gating behavior[40] refers to the stimulus-gated transport characteristics exhibited by nanochannels or nanopores functionalized with stimuli-responsive molecules. This property can be used not only for the direct detection of the stimuli themselves (e.g., ion concentration) but also, more importantly, for converting specific recognition events (e.g., aptamer-target binding) into a switch between open and closed states of the channel. This enables detection with high signal-to-noise ratio and excellent reproducibility.
Recognition probes [Figure 3D] are the core functional elements that confer specificity and intelligent responsiveness to bio-inspired nanofluidic sensors. Molecular probes[41] are designed with special functional groups that recognize specific ions or small molecules. Aptamers[42], which are single-stranded DNA or RNA oligonucleotides obtained through in vitro selection, offer high affinity, specificity, and ease of chemical synthesis and modification. Target binding often induces a significant conformational change in aptamers. Antibodies[43], renowned for their exceptional specificity, are commonly employed for detecting biomacromolecules such as proteins and pathogens. Molecularly imprinted polymers (MIPs)[44], serving as artificial antibodies, provide superior chemical stability. They are synthesized in the presence of a template molecule; after template removal, complementary cavities that match in shape, size, and functional groups to the target are formed within the polymer network. Their application in nanofluidic sensors shows distinct advantages, particularly in detecting small molecules and targets that are difficult to raise antibodies against. These probes are precisely functionalized on the inner surfaces or entrance regions of the nanochannels[45]. Their specific binding events with target analytes alter key physicochemical parameters, such as the effective channel size, surface charge, or wettability, which are sensitively transduced into detectable changes in ionic current, potential, or rectification signals. This mechanism enables highly selective and sensitive detection of the target. The diverse recognition elements, based on their distinct preparation principles and intrinsic properties, offer versatile design strategies for nanofluidic sensors.
Applications of olfactory-inspired nanofluidic sensors
Olfactory-inspired sensors can effectively amplify the signal of binding events, thereby enhancing sensitivity. In terms of specific recognition, artificially designed recognition probes[46,47] (e.g., aptamers and MIPs) are introduced into the sensor system, enabling the sensors to achieve organism-like high selectivity toward target analytes. Moreover, compared with traditional bioinspired sensors relying on natural biomaterials (e.g., enzymes and antibodies), olfactory-inspired nanofluidic sensors exhibit enhanced chemical stability. This advantage is mainly attributed to the precise design of channel materials, which can effectively avoid the common issues of biomolecular inactivation and degradation under harsh detection conditions (e.g., extreme pH and high temperature). In addition, the response speed of these sensors can be further optimized by regulating mass transfer processes, rendering them more suitable for practical application scenarios. Therefore, olfactory-inspired nanofluidic sensors based on confined nanochannels hold great potential in the field of high-performance chemical and biological sensing.
Olfactory-inspired nanofluidic sensors have achieved significant progress[48]; a large number of related studies have demonstrated the great application potential of these sensors in environmental monitoring, medical diagnosis, food safety detection, and other important fields. The key performance parameters involved herein include response time, signal reproducibility, sensitivity and limit of detection, specificity and selectivity, linear detection range, regenerability, and long-term stability, each of which plays a crucial role in determining the overall performance and practical applicability of the sensor. Response time[49] is defined as the time required for the sensor signal (e.g., ionic current, conductance, current rectification ratio) to reach 90%/95% of the maximum steady-state response upon exposure to target odor molecules, which serves as a core indicator to evaluate the rapid recognition capability. Specifically, the magnitude of response time is closely associated with the mass transfer rate in nanochannels, the binding rate of the recognition process, the signal transduction efficiency at the nanochannel interface, and the efficiency of signal processing and transmission. Consequently, a shorter response time is conducive to meeting the requirements of on-site rapid detection and real-time monitoring, which are essential for practical applications. Among these parameters, signal reproducibility[50] is a prerequisite for the quantitative detection and batch application of olfactory-inspired nanofluidic sensors. Its evaluation is based on repeatability and reproducibility, both quantified by the relative standard deviation (RSD); notably, a smaller RSD indicates superior reproducibility. This parameter is closely related to factors such as the uniformity of nanochannel device fabrication, the stability and uniformity of recognition element immobilization, and the stability of the signal baseline. Therefore, the performance of sensors in this regard can be significantly enhanced through the development of high-precision, batch-compatible nanochannel fabrication technologies, the improvement of the stability and distribution uniformity of immobilized recognition elements, the establishment of standardized detection systems, the optimization of signal calibration, and the mitigation of baseline drift. Additionally, parameters related to molecular recognition[51] are also critical for evaluating sensor performance, as they directly determine the sensor’s ability to accurately identify target odor molecules. To elaborate, sensitivity is defined as the rate of signal change of the sensor with the concentration of target odor molecules, which is measured by the slope of the calibration curve. Selectivity, by contrast, refers to the capability of the sensor to generate a much higher response to target odor molecules than to interfering molecules, quantified by the selectivity coefficient (response to target molecules/response to interfering molecules). The linear detection range, meanwhile, is the concentration interval where the sensor signal exhibits a linear relationship with the target molecule concentration, which should strike a balance between trace detection and the detection of medium-to-high concentrations in real samples. Regenerability describes the ability of the sensor to recover its initial recognition performance after simple treatment, evaluated by the number of reusable cycles, while long-term stability refers to the sensor’s capacity to maintain its recognition capability under specific storage conditions, quantified by the signal retention rate after a certain storage period.
Studies based on ionic current achieve the transduction from molecular recognition events into the electrical signals, thereby establishing the theoretical foundation for biomimetic sensing. When target molecules interact specifically with the recognition elements modified on the inner surface of the nanochannel, it will cause changes in the surface charge density, wettability, or steric hindrance of the channel, thereby regulating the transport rate and transport amount of ions in the channel[52]. These changes can be accurately captured as changes in ionic current intensity, current-voltage curves, or other electrical parameters, thereby realizing the quantitative and qualitative detection of target molecules. This ionic current-based transduction mechanism not only has the advantages of high sensitivity, fast response speed, and low detection cost but also establishes a solid theoretical foundation for the development of high-performance biomimetic sensing systems, providing a feasible technical path for the integration of molecular recognition and signal transduction. Developments are advancing in parallel across all fronts, including the design of recognition probes, the refinement of detection principles, the enhancement of mass transport, and the transduction and readout of sensing signals.
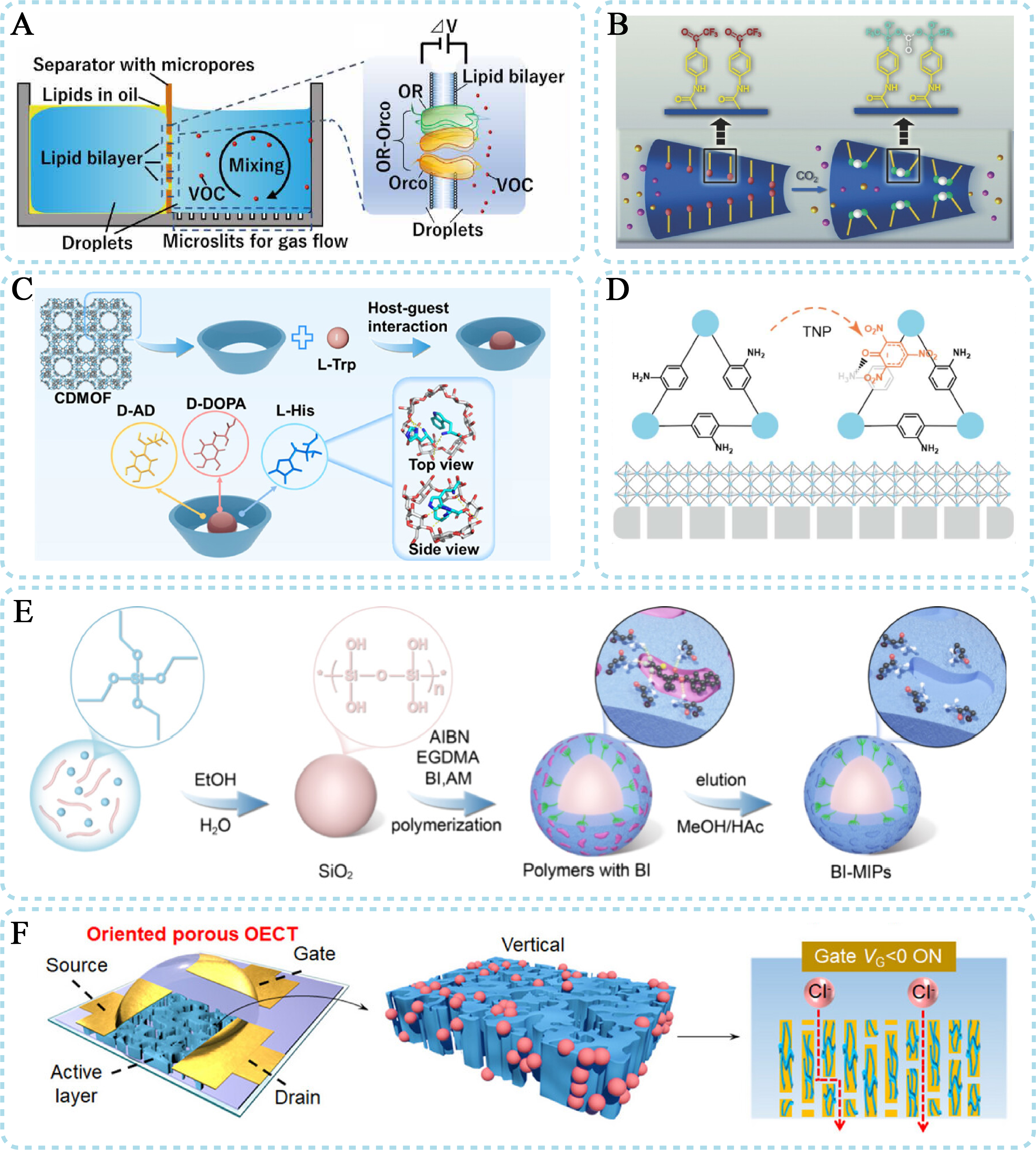
Research strategies have progressed from the use of biological materials to bioinspired materials. Yamada et al. report a volatile organic compound (VOC) sensor based on olfactory receptors, which, together with an olfactory receptor co-receptor, forms a ligand-gated ion channel and demonstrates the direct application of biological materials [Figure 4A][53]. The receptors serve as pivotal natural biological recognition elements after their reconstitution into lipid bilayers. Endowed with intrinsic specific binding affinity for target VOCs, these receptors trigger characteristic nanofluidic signal perturbations upon molecular interaction, which underpins the high-sensitivity and specific detection of VOCs. The rational utilization of insect olfactory receptors gives full play to their superior native recognition performance, furnishing a crucial and feasible strategy for the advancement of bionic olfactory nanofluidic sensing technology. A mosquito olfaction-inspired artificial ion gate abstracts the CO2-driven ionic gate into a functional gating mechanism, enabling intelligent gas detection [Figure 4B][54]. This engineered ionic gate features a rapid response time and an ultra-low detection limit for CO2; its specific molecular recognition of CO2 triggers dynamic modulation of nanofluidic ionic transport and generates distinct sensing signals, enabling high-precision, sensitive CO2 detection. It furnishes an innovative biomimetic design strategy and technical insight for constructing bionic olfactory nanofluidic sensing systems based on biomimetic recognition materials. Substantial research efforts are directed toward constructing high-performance synthetic recognition interfaces. For instance, the construction of chiral nanochannel membranes or cyclodextrin-based chiral microenvironments enables highly selective discrimination of protein adsorption behavior and enantiomeric molecules [Figure 4C][55]. Molecular probe-integrated nanofluidic sensors inspired by canine olfaction provide a stable and efficient solution for detecting small molecules such as trinitrophenol (TNP) [Figure 4D][56]. This study develops an ultrasensitive nanofluidic sensor for TNP, bioinspired by the olfactory sensory neurons of sniffer dogs, with bioengineered biomimetic materials as the core recognition elements for bionic olfactory nanofluidic sensing. The biomimetic elements realize specific molecular recognition of TNP, whose binding events trigger precise modulation of nanofluidic ionic transport and generate distinct, concentration-dependent sensing signals, thus enabling ultra-sensitive and selective detection of the target analyte. Furthermore, MIP ion channels that integrate dual-recognition mechanisms [Figure 4E][49], combining size sieving with specific binding, have been developed to simultaneously capture energy from ion-containing wastewater and monitor its composition, pointing toward a novel paradigm for sustainable environmental monitoring. The sensitivity of a sensor is strongly correlated with the efficiency of mass transport. Research demonstrates that employing highly aligned nanochannel aerogels as the transport matrix can drastically reduce ion transport resistance [Figure 4F], significantly enhancing device transconductance and signal amplification capability[57]. This advancement lays a critical material foundation for constructing ultrasensitive biosensors.
Figure 4. Olfactory-inspired nanofluidic sensors based on ionic current. Direct application of biomaterials in (A) Design of biomimetic materials in (B-E). Enhancement of mass transport in (F). (A) VOC sensor based on olfactory receptors. This figure is quoted with permission from The American Association for the Advancement of Science[53]; (B) Mosquito olfaction-inspired artificial ion gate. This figure is quoted with permission from John Wiley and Sons[54]; (C) The construction of chiral nanochannel. This figure is quoted with permission from The American Chemical Society[55]; (D) Molecular probe-integrated nanofluidic sensors. This figure is quoted with permission from The Royal Society of Chemistry[56]; (E) Molecularly imprinted polymer ion channels. This figure is quoted with permission from The American Chemical Society[49]; (F) Aligned nanochannel to reduce ion transport resistance. This figure is quoted with permission from The John Wiley and Sons[57]. HAc: Acetic acid; TNP: trinitrophenol; CDMOF: β-cyclodextrin metal−organic frameworks; D-AD: D-adrenaline; D-DOPA: D-dihydroxyphenylalanine; L-His: L-histidine; L-Trp: L-tryptophan; AIBN: 2,2'-azobis(2-methylpropionitrile; EGDMA: ethylene glycol dimethacrylate; BI: bifenthrin; MIP: molecularly imprinted polymer; VOC: volatile organic compound.
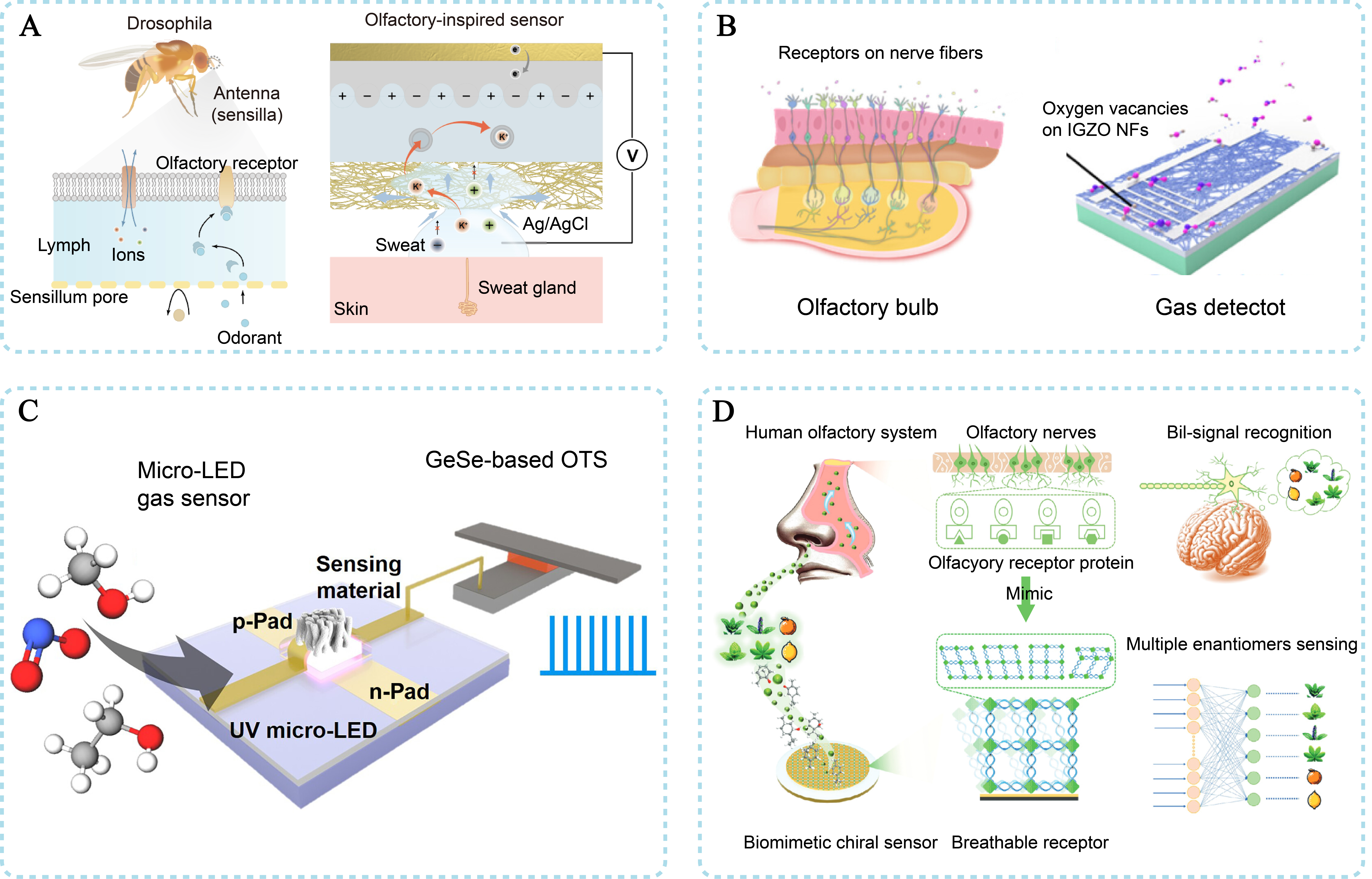
The ever-growing demands for enhanced stability, seamless integration and versatile functional performance across fields such as intelligent sensing, portable detection, micro-nano devices and on-site analysis are drastically accelerating the innovative development of diverse signal output forms in sensing technology. Research is dedicated to different types of signal outputs, which free sensor devices from bulky liquid-phase systems and pave the way for miniaturization and easier integration. Employing a variety of measurement methods enables the exploration of novel mechanisms and the discovery of new materials. Potential signals, owing to their inherent stability and low power consumption, represent an ideal choice for enabling continuous, wireless monitoring. Wu et al. report separation-sensing nanochannel-based electronics for sweat ion monitoring, converting ion recognition into a stable membrane potential output via an ion-selective electrode [Figure 5A][58]. This work broke the limitations of traditional fabrication by employing a biomimetic preparation strategy, significantly enhancing the performance of the sensitive membranes. Resistance signals, with their direct response and simple circuitry, dominate in environmental gas and breath biomarker sensing. Ma et al. present an autonomous mobile gas sensing system, based on high specific surface area indium gallium zinc oxide nanofibers (IGZO NFs), offering real-time monitoring and precise gas source localization without the need for human intervention. Room-temperature gas sensors exhibit low power consumption (∼ 0.5 mW), exceptional sensitivity (∼ 1,290% ppb-1), and a low detection limit of 20 ppb for toxic NO2 [Figure 5B][59]. A high-performance ammonia sensor inspired by the efficient mass transport and adsorption structures of a dog’s nasal cavity is also a typical example[62]. It utilizes biochar/SnO2 composite materials with a biomimetic hierarchical porous structure. The combination of ammonia molecules on the material surface directly modulates the bulk resistance of the semiconductor, enabling rapid, highly sensitive detection. Kang et al. report an artificial olfactory neuron module that generates spike signals with low operation voltage, demonstrating energy efficiency and advanced performance. A real-time gas classification based on the spiking neural network is feasibly conducted with an accuracy of 99.6% [Figure 5C][60]. Recordings of resistive current pulses during the translocation of single molecules or particles through electrolyte-filled nanopores enable researchers to understand properties at a more microscopic level, such as translocation time, volume, charge, shape and ligand affinity[63]. The recognition event does not cause a change in a single electrical parameter. Emerging non-electrochemical signals support both the development of sensitive materials and the convergence of multi-signal sensing paradigms [Figure 5D][61].
Figure 5. Different signal output forms. (A) Potentiometric measurement for continuous, wireless monitoring. This figure is quoted with permission from The American Chemical Society[58]; (B) Resistance measurement suited for gas molecules detection. This figure is quoted with permission from The American Chemical Society[59]; (C) Spike signals can classify different ingredients under humidity disturbance conditions. This figure is quoted with permission from The American Association for the Advancement of Science[60]; (D) Non-electrochemical signals contribute to the development of sensitive materials. This figure is quoted with permission from The John Wiley and Sons[61]. LED: Light-emitting diode; OTS: ovonic threshold switch.
Design strategies for improving sensing performance
The design of olfactory-inspired nanofluidic sensors requires performance enhancements at the technical level. To further advance the selectivity, sensitivity, and intelligence of bio-inspired sensors and bridge the gap between laboratory prototypes and practical industrial applications, targeted improvements can be systematically achieved through the organic integration of molecular design, channel engineering, interdisciplinary theory, and systematic array-based approaches, each of which plays a unique and irreplaceable role in optimizing sensor performance.
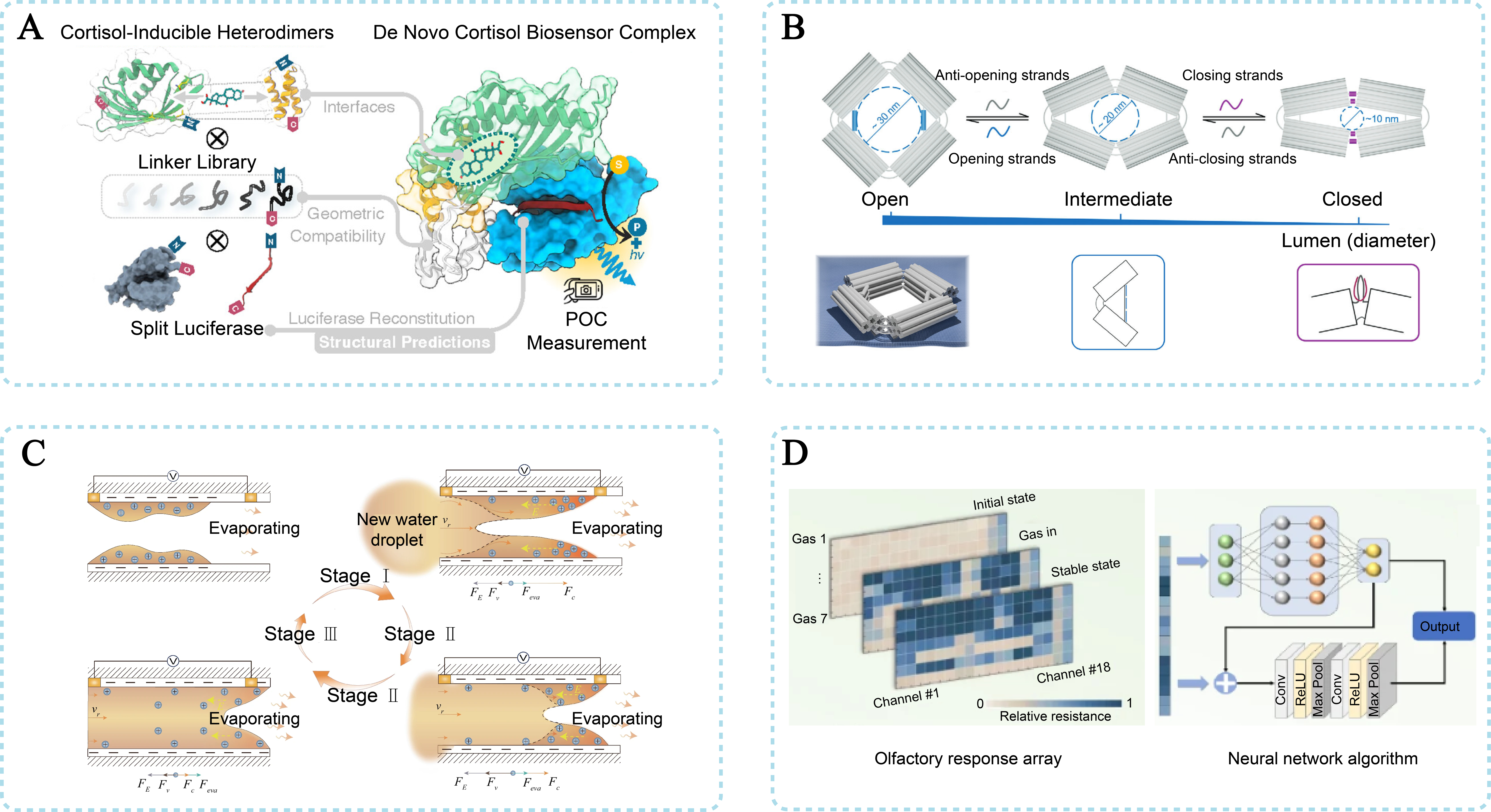
Molecular design serves as the fundamental core for enhancing the selectivity and specific recognition capability of bio-inspired sensors, as the interaction between the sensor’s recognition element and the target analyte is essentially determined by molecular-level structures and forces. Computational assistance materials design enables the precise regulation of the physicochemical properties of recognition units or sensing materials, providing the molecular foundation for high-performance sensing. Chen et al. de novo design cortisol-inducible dimerization modules [Figure 6A][64]. The resulting biosensor, designed straight from the computer, yields over 300-fold luminescent response with picomolar sensitivity and can be rapidly imaged by a standard camera or smartphone. Smart nanopores are endowed with dynamic adaptive capabilities, which can intelligently switch between distinct structural and functional states in response to subtle variations in the surrounding microenvironment. Such environmental triggers cover a diverse range of physical and chemical cues, including changes in pH values, ionic strength, temperature, redox potential, or the specific binding of target biomolecules such as proteins, nucleic acids and small metabolites. The state switching is realized through reversible conformational rearrangements of the nanopore’s structural moieties or stimulus-responsive interactions between its building blocks and environmental factors, leading to adjustable pore size, surface charge and molecular permeability. Yu et al.[65] present a structurally reconfigurable DNA origami MechanoPore that features a lumen that is tunable in size through molecular triggers, offering functional nanostructures that can be tuned on demand [Figure 6B]. Theory-guided material design stands as a pivotal strategy in advanced functional material development, breaking the limitations of traditional trial-and-error approaches and markedly enhancing the material’s performance in key physicochemical properties. This design paradigm integrates multidisciplinary theoretical frameworks - including computational simulation, thermodynamics, fluid dynamics and molecular modeling - to precisely predict the structure-performance correlations of materials. It enables rational optimization of critical structural parameters such as pore architecture, surface chemical properties, and hierarchical micro/nanostructures that govern mass transport processes, thereby accelerating the transfer rate of molecules/ions, improving transport selectivity and stability. Ge et al.[66] utilize the hydrovoltaic effect, which offers a promising route for ion sensing [Figure 6C]. The ultimate performance of bio-inspired olfactory sensing relies on large-scale, diversified sensor arrays and their advanced data processing methods, which emulate the high-throughput and high-discrimination capabilities of biological olfactory systems. This integrated design perfectly mimics the biological olfactory system’s neural encoding and signal processing mechanisms, breaking the performance bottleneck of single olfactory sensors and unlocking the full potential of bio-inspired olfactory sensing for high-precision applications in environmental monitoring, food quality detection, clinical disease diagnosis and environmental safety early warning. Yue et al. present a biomimetic intelligent olfactory sensing system based on the integration of an 18-channel microelectromechanical system (MEMS) sensor array (16 gas sensors, 1 humidity sensor and 1 temperature sensor), a complementary metal-oxide-semiconductor (CMOS) circuit and an olfactory lightweight machine-learning algorithm inspired by Drosophila [Figure 6D][67]. Wang et al. report biomimetic olfactory chips that integrate nanotube sensor arrays on nanoporous substrates with up to 10,000 individually addressable sensors per chip, and integrated with artificial intelligence[68]. The integration of the sensor array and artificial intelligence represents a core strategy to overcome the key challenges of selectivity in artificial smell.
Figure 6. Design Strategies for Improved Sensing Performance. (A) Computational assistance materials design. This figure is quoted with permission from The American Chemical Society[64]; (B) Smart Nanopores. This figure is quoted with permission from The John Wiley and Sons[65]; (C) Theory-guided material design. This figure is quoted with permission from Springer Nature[66]; (D) Olfactory sensor array. This figure is quoted with permission from Springer Nature[67]. POC: Point-of-care.
In addition, the complexity of the actual application environments for sensors is another issue that requires active consideration and resolution. In practical applications, high-precision sensors usually integrate sample preparation to remove interferents, and sensors developed in laboratories mostly adopt a two-stage process involving microfluidic separation, selective filtration, or pre-concentration units[69-71]. How to enable sensitive membranes to output signals while effectively shielding against interference is also a critical problem for the future development of biomimetic materials. This biomimetic strategy can actively exclude complex matrix interferences, significantly enhancing detection specificity and signal-to-noise ratio from the source. Through multi-signal coupling or logical processing, it can further improve detection accuracy and minimize the probability of false positives or false negatives. Breaking through the limitation of relying on a single electrical parameter, this method can simultaneously capture multi-dimensional signals triggered by target analytes, such as optical, electrical, acoustic, and thermal changes, with the help of customized material and device design[72]. After decoding by artificial intelligence, the ability to discriminate complex mixtures can be greatly enhanced. In addition, drawing on the efficient processing mechanism of the biological olfactory system, on-chip integration technology is used to fabricate biomimetic receptor arrays composed of sensing units, simulating its “combinatorial coding” principle. The massive and parallel data streams generated thereby lay the foundation for high-throughput identification. Meanwhile, inspired by the efficient perceptual logic of biological systems and to meet the demands for real-time operation, low power consumption, and privacy protection, the ultimate form of sensor performance improvement will be the deep hardware-level integration of sensing, storage, and computing functions. Relying on neuromorphic devices and in-memory computing architectures[73], localized real-time processing and decision-making of sensor signals can be achieved, which minimizes system latency and power consumption, thereby promoting the practical implementation of applications such as wearable diagnostics and embedded environmental monitoring.
CONCLUSION AND OUTLOOK
Olfactory-inspired nanofluidic sensors have emerged as an important component of chemical sensing. By learning from biological perception mechanisms and drawing inspiration from biological olfactory signal amplification strategies, a comprehensive theoretical and operational framework has been established, encompassing material design, specific binding, structural regulation, mass transport, signal transduction, and amplification. These sensors achieve control at the microscopic nanoscale, including fabrication methods, surface functionalization strategies, and the decoupling of ion transport processes, demonstrating exceptional sensitivity and selectivity. With applications spanning health monitoring, environmental detection, food safety, and public security, the development of their integrated capabilities holds promise for enabling real-time, in-situ intelligent monitoring across various fields. Furthermore, their deployment on intelligent robotic platforms can meet the detection demands in hazardous environments.
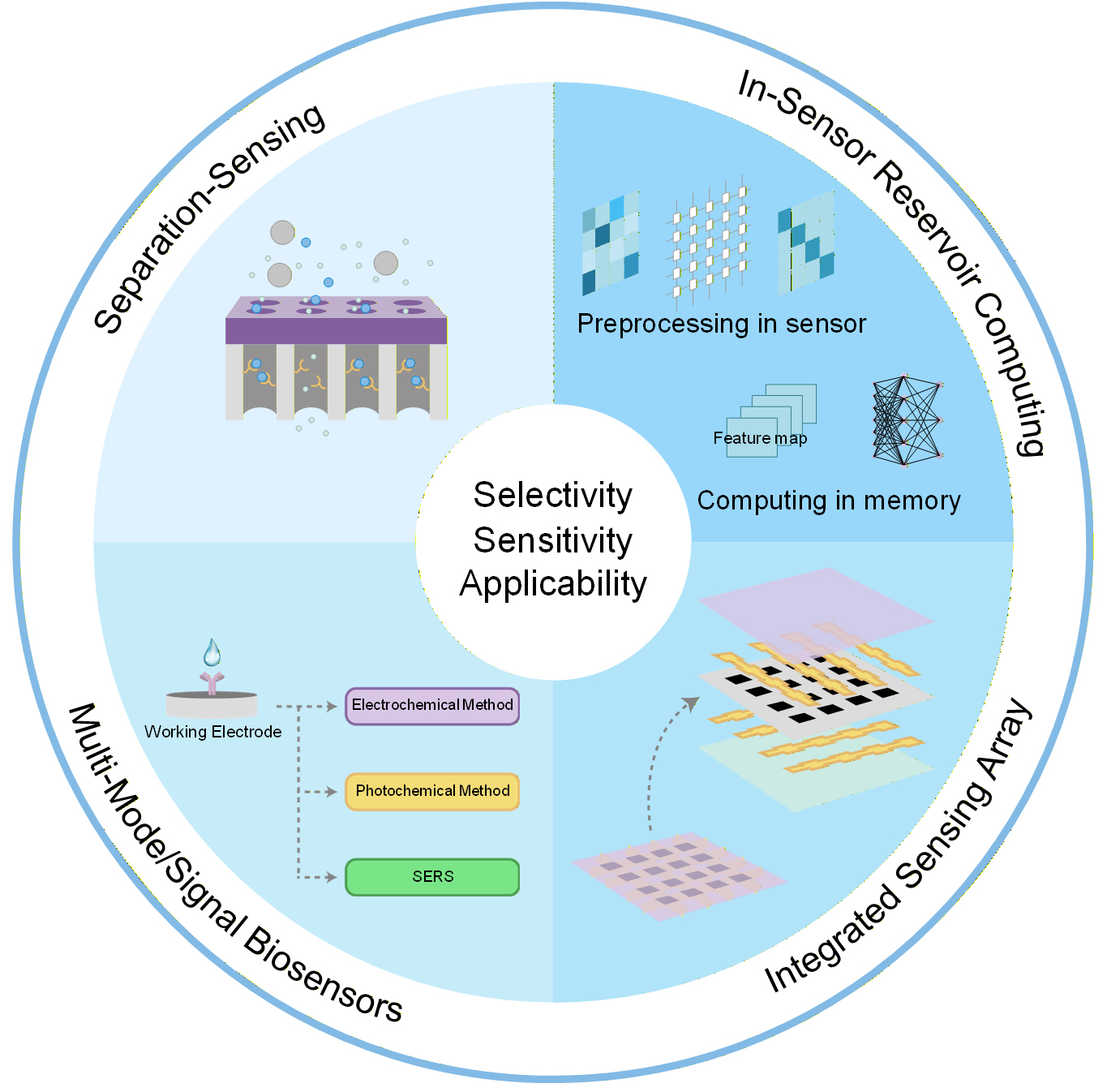
Improving the selectivity, sensitivity, and practical usability of nanofluidic sensors requires progress along four interconnected paths: refined design strategies, multi-modal signal integration, scalable sensor array architectures, and the co-development of sensing, storage, and computing functions[Figure 7].
Separation-sensing
In separation-sensing integration strategies, the use of heterogeneous materials is inevitable, making interface compatibility a primary consideration. Furthermore, addressing the critical issue of the material compatibility between interference shielding and signal output efficiency requires overcoming the challenges of precise sieving, analyte enrichment, and mass transport.
Multi-mode/signal biosensors
For multimodal signal integration, the primary breakthroughs needed lie in reducing cross-interference among signals and achieving effective data fusion and accurate interpretation. This requires sensor designs that comprehensively account for the physicochemical characteristics of various detection modalities, along with the establishment of robust data processing frameworks and models.
Integrated sensing array
For sensing arrays integration, the key obstacles lie in the consistency and reliability of large-scale manufacturing, the complexity of multi-channel interconnection, the uniform distribution of fluid samples, as well as issues related to system integration and miniaturization. This demands new breakthroughs in nanofluidic fabrication technologies, optimized signal-circuit synergy, and the development of plug-and-play sensor array modules to reduce maintenance costs.
In-sensor reservoir computing
The goal of in-sensor reservoir computing is to enable in situ, real-time, and intelligent signal processing, offering a promising pathway to overcome core bottlenecks in nanofluidic sensing, such as power consumption, latency, and intelligent analysis. Realizing this requires reliable on-chip integration of micro/nanofluidic systems with solid-state memristor arrays, a complex interdisciplinary fabrication challenge. Additionally, material design must ensure consistency and reliability for large-scale manufacturing.
DECLARATIONS
Authors’ contributions
Conceptualized the idea and led the project: Wu, Y.; Zhu, C.; Kong, X. Y.; Wen, L.
Made substantial contributions to writing the paper: Wu, Y.; Liu, Y.; Zhu, C.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool DeepSeek was used exclusively for language polishing. The tool did not contribute to the scientific content of the study. All authors accept full responsibility for the integrity of the research and the final text.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant Nos. 22304172 and 22122207), the Natural Science Foundation of Jiangxi Province (Grant No. 20252BAC200220), and the Director’s Fund of the Technical Institute of Physics and Chemistry, Chinese Academy of Sciences (Grant No. E5A9Q10201).
Conflicts of interest
Wen, L. serves as an Associate Editor of the journal Iontronics, and Kong, X. Y. is a member of the Editorial Board of the journal Iontronics. They were not involved in any steps of the editorial process, notably including reviewers’ selection, manuscript handling, or decision-making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Auer, T. O.; Khallaf, M. A.; Silbering, A. F.; et al. Olfactory receptor and circuit evolution promote host specialization. Nature 2020, 579, 402-8.
2. Korsching, S. I. Evolution of vertebrate olfactory receptor repertoires and their function. Curr. Opin. Behav. Sci. 2025, 61, 101483.
3. Shepherd, G. M. Smell images and the flavour system in the human brain. Nature 2006, 444, 316-21.
4. Guerrini, L.; Garcia-Rico, E.; Pazos-Perez, N.; Alvarez-Puebla, R. A. Smelling, seeing, tasting-old senses for new sensing. ACS. Nano. 2017, 11, 5217-22.
6. Buzek, A.; Serwańska-Leja, K.; Zaworska-Zakrzewska, A.; Kasprowicz-Potocka, M. The shape of the nasal cavity and adaptations to sniffing in the dog (canis familiaris) compared to other domesticated mammals: a review article. Animals. (Basel). 2022, 12, 517.
7. Kokocińska-Kusiak, A.; Woszczyło, M.; Zybala, M.; Maciocha, J.; Barłowska, K.; Dzięcioł, M. Canine olfaction: physiology, behavior, and possibilities for practical applications. Animals. (Basel). 2021, 11, 2463.
8. Zhao, Z.; Yang, Q.; Li, R.; et al. A comprehensive review on the evolution of bio-inspired sensors from aquatic creatures. Cell. Rep. Phys. Sci. 2024, 5, 102064.
9. Abel, R. L.; Maclaine, J. S.; Cotton, R.; et al. Functional morphology of the nasal region of a hammerhead shark. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2010, 155, 464-75.
10. Rygg, A. D.; van Duin, A. C.; Craven, B. A. Molecular dynamics simulations of water/mucus partition coefficients for feeding stimulants in fish and the implications for olfaction. PLoS. ONE. 2013, 8, e72271.
11. Kaczmarek, P.; Hermyt, M.; Rupik, W. Embryology of the VNO and associated structures in the grass snake Natrix natrix (Squamata: Naticinae): a 3D perspective. Front. Zool. 2017, 14, 1.
12. Holtzman, D. A.; Halpern, M. Embryonic and neonatal development of the vomeronasal and olfactory systems in garter snakes (Thamnophis spp.). J. Morphol. 1990, 203, 123-40.
13. Kaupp, U. B. Olfactory signalling in vertebrates and insects: differences and commonalities. Nat. Rev. Neurosci. 2010, 11, 188-200.
14. Ando, T.; Sekine, S.; Inagaki, S.; et al. Nanopore formation in the cuticle of an insect olfactory sensillum. Curr. Biol. 2019, 29, 1512-1520.e6.
15. Chu, W.; Yang, M.; Shang, Z.; et al. Machine learning assisted nanofluidic array for multiprotein detection. ACS. Nano. 2025, 19, 8539-51.
16. Emmerich, T.; Ronceray, N.; Agrawal, K. V.; et al. Nanofluidics. Nat. Rev. Methods. Primers. 2024, 4, 69.
18. Ling, H.; Kong, X.; Wen, L. Ionic superfluidics: a perspective on emerging frameworks for ion transport in confined channels. Iontronics 2025, 1, 6.
19. Brunet, T.; King, N. The origin of animal multicellularity and cell differentiation. Dev. Cell. 2017, 43, 124-40.
20. Fulton, K. A.; Zimmerman, D.; Samuel, A.; Vogt, K.; Datta, S. R. Common principles for odour coding across vertebrates and invertebrates. Nat. Rev. Neurosci. 2024, 25, 453-72.
21. Bhandawat, V.; Reisert, J.; Yau, K. W. Elementary response of olfactory receptor neurons to odorants. Science 2005, 308, 1931-4.
22. Wicher, D.; Schäfer, R.; Bauernfeind, R.; et al. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 2008, 452, 1007-11.
23. Li, Z.; Huang, L.; Wang, K.; Xia, X. Developing solid-state single-, arrayed-, and composite-nanopore sensors for biochemical sensing applications. Acc. Mater. Res. 2024, 5, 761-71.
24. Cai, J.; Zhang, W.; Xu, L.; et al. Polarization-sensitive optoionic membranes from chiral plasmonic nanoparticles. Nat. Nanotechnol. 2022, 17, 408-16.
25. Li, X.; Zhu, C.; Wu, Y.; Kong, X. Y.; Wen, L. Bioinspired solid-state nanochannels for molecular analysis. Nanoscale 2025, 17, 1225-37.
26. Lee, S.; Cha, J.; Shin, S.; et al. Corona dynamics of nanoparticles and their functional design space in molecular sensing. ACS. Nano. 2025, 19, 24653-68.
27. Park, Y.; Kim, H.; Ranjith, K. S.; et al. Facile and tunable ligand engineering of nanofiber-embedded perovskite quantum dots for ammonia sensing. Adv. Fiber. Mater. 2026, 668.
28. Ge, C.; Wang, Y.; Wang, M.; et al. Silk fibroin-regulated nanochannels for flexible hydrovoltaic ion sensing. Adv. Mater. 2024, 36, e2310260.
29. Liu, P.; Zheng, M.; Wang, Y.; et al. Photoactivated ion transport: role of intrinsic defects and plasmonics for efficient ionic power harvesting. J. Am. Chem. Soc. 2025, 147, 26944-54.
30. Hu, X.; Xu, H.; Lu, J.; et al. Selective ion transport of nonlinear resistive switching by hierarchical nanometer-to-angstrom channels for nanofluidic transistors. Sci. Adv. 2025, 11, eadw7882.
31. Garaj, S.; Hubbard, W.; Reina, A.; Kong, J.; Branton, D.; Golovchenko, J. A. Graphene as a subnanometre trans-electrode membrane. Nature 2010, 467, 190-3.
32. Xiao, K.; Xie, G.; Li, P.; et al. A biomimetic multi-stimuli-response ionic gate using a hydroxypyrene derivation-functionalized asymmetric single nanochannel. Adv. Mater. 2014, 26, 6560-5.
33. Lee, W.; Park, S. J. Porous anodic aluminum oxide: anodization and templated synthesis of functional nanostructures. Chem. Rev. 2014, 114, 7487-556.
34. Meng, D.; Li, C.; Hao, C.; et al. Interfacial self‐assembly of chiral selenide nanomembrane for enantiospecific recognition. Angew. Chem. 2023, 135, e202311416.
35. Zhu, C.; Wu, Y.; Li, X.; et al. Engineered nanofluidics for molecular recognition and physical perception. Angew. Chem. Int. Ed. Engl. 2025, 64, e202506689.
36. Li, X.; Wang, Z. L.; Wei, D. Iontronic logic control driven by dynamic electrical double layer regulation. Iontronics 2025, 1, 2.
37. Li, X.; Cheng, T.; Wang, Z. L.; Wei, D. Neuromimetic circuits enabled by dynamic regulation of the electrical double layer. npj. Flex. Electron. 2025, 9, 66.
38. Zhang, H.; Li, X.; Hou, J.; Jiang, L.; Wang, H. Angstrom-scale ion channels towards single-ion selectivity. Chem. Soc. Rev. 2022, 51, 2224-54.
39. Mayer, S. F.; Mitsioni, M. F.; Robin, P.; et al. Lumen charge governs gated ion transport in β-barrel nanopores. Nat. Nanotechnol. 2026, 21, 116-24.
40. Russell, W. S.; Lin, C. Y.; Siwy, Z. S. Gating with charge inversion to control ionic transport in nanopores. ACS. Appl. Nano. Mater. 2022, 5, 17682-92.
41. Yang, L.; Qian, Y.; Kong, X. Y.; et al. Specific recognition of uranyl ion employing a functionalized nanochannel platform for dealing with radioactive contamination. ACS. Appl. Mater. Interfaces. 2020, 12, 3854-61.
42. Chen, Y.; Yue, X.; Yu, W.; et al. Breaking the sensitivity-specificity trade-off: a nanoconfined dual-DNA aptamer synergy strategy. Anal. Chem. 2025, 97, 11874-82.
43. Ryu, J. S.; Lee, S. H.; Kim, H.; et al. Advancing SARS‐CoV‐2 variant detection with high affinity monoclonal antibodies and plasmonic nanostructure. Adv. Funct. Mater. 2024, 34, 2405340.
44. Tang, W.; Yin, L.; Sempionatto, J. R.; Moon, J. M.; Teymourian, H.; Wang, J. Touch-based stressless cortisol sensing. Adv. Mater. 2021, 33, e2008465.
45. Hu, J. J.; Lin, N.; Yuan, L.; Lou, X.; Xia, F. Detection of analytes with the outer surface of solid-state nanochannels: from pm to μm. Acc. Chem. Res. 2025, 58, 834-46.
46. Mintz Hemed, N.; Leal-Ortiz, S.; Zhao, E. T.; Melosh, N. A. On-demand, reversible, ultrasensitive polymer membrane based on molecular imprinting polymer. ACS. Nano. 2023, 17, 5632-43.
47. Jin, H.; Yang, H.; Huang, W.; et al. protein-templated ultrasmall Pt nanocluster in adenosine ATP-responsive MOF nanoarchitectonics for ultrasensitive point-of-care detection. Anal. Chem. 2025, 97, 19982-91.
48. Liu, Y.; Jia, S.; Yiu, C. K.; et al. Intelligent wearable olfactory interface for latency-free mixed reality and fast olfactory enhancement. Nat. Commun. 2024, 15, 4474.
49. Xu, Y.; Awati, A.; Zhou, S.; et al. Concentration energy ion channels with molecular-structure dual recognition for sustainable environmental monitoring. J. Am. Chem. Soc. 2025, 147, 18910-22.
50. Lee, D. H.; Lee, W. Y.; Kim, J. Introducing nanoscale electrochemistry in small-molecule detection for tackling existing limitations of affinity-based label-free biosensing applications. J. Am. Chem. Soc. 2023, 145, 17767-78.
51. Gao, W.; Emaminejad, S.; Nyein, H. Y. Y.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509-14.
52. Gao, P.; Ma, Q.; Ding, D.; et al. Distinct functional elements for outer-surface anti-interference and inner-wall ion gating of nanochannels. Nat. Commun. 2018, 9, 4557.
53. Yamada, T.; Sugiura, H.; Mimura, H.; Kamiya, K.; Osaki, T.; Takeuchi, S. Highly sensitive VOC detectors using insect olfactory receptors reconstituted into lipid bilayers. Sci. Adv. 2021, 7, eabd2013.
54. Shang, X.; Xie, G.; Kong, X. Y.; et al. An artificial CO2 -driven ionic gate inspired by olfactory sensory neurons in mosquitoes. Adv. Mater. 2017, 29, 1603884.
55. Su, T.; Yang, M.; Wang, S.; Song, Y. Y.; Gao, Z.; Zhao, C. Electrochemical platform regulated by cyclodextrin chiral microenvironments for enhanced enantioselective recognition. Anal. Chem. 2025, 97, 21660-7.
56. Li, X.; Liu, Z.; Yang, L.; et al. An ultrasensitive 2,4,6-trinitrophenol nanofluidic sensor inspired by olfactory sensory neurons in sniffer dogs. Chem. Sci. 2024, 15, 19504-12.
57. Sun, Z.; Hu, Z.; Liu, X.; et al. Oriented nanochannel aerogels for high-transconductance organic transistors and ultrasensitive biosensors. Adv. Mater. 2025, 37, e09290.
58. Wu, Y.; Wang, Q.; Li, X.; et al. Olfactory-inspired separation-sensing nanochannel-based electronics for wireless sweat monitoring. ACS. Nano. 2025, 19, 3781-90.
59. Ma, Q.; Cao, S.; Wang, H.; et al. Autonomous and ultrasensitive low-power metal oxide nanofiber gas sensor for source tracking and localization. ACS. Sens. 2025, 10, 2938-47.
60. Kang, M.; Han, J. K.; Lee, K.; et al. Neuromorphic olfaction with ultralow-power gas sensors and ovonic threshold switch. Sci. Adv. 2025, 11, eadv9222.
61. Li, N.; Zhang, J. B.; Wöll, C.; Gu, Z. G.; Zhang, J. Breathable biomimetic chiral porous MOF thin films for multiple enantiomers sensing. Adv. Funct. Mater. 2025, 35, 2422860.
62. Qin, Z.; Wu, Z.; Sun, Q.; et al. Dog nose-inspired high-performance ammonia sensor based on biochar/SnO2 composite. Carbon 2023, 213, 118297.
63. Yusko, E. C.; Johnson, J. M.; Majd, S.; et al. Controlling protein translocation through nanopores with bio-inspired fluid walls. Nat. Nanotechnol. 2011, 6, 253-60.
64. Chen, J. Y.; Peng, X.; Xi, C.; Lee, G. R.; Baker, D.; Yeh, A. H. De novo design of high-performance cortisol luminescent biosensors. J. Am. Chem. Soc. 2025, 147, 27494-505.
65. Yu, Z.; Baptist, A. V.; Reinhardt, S. C. M.; et al. Compliant DNA origami nanoactuators as size-selective nanopores. Adv. Mater. 2024, 36, e2405104.
66. Ge, C.; Wang, M.; Zhou, Y.; et al. Ion transport-triggered rapid flexible hydrovoltaic sensing. Nat. Commun. 2025, 16, 8110.
67. Yue, X.; Wang, J.; Yang, H.; et al. A Drosophila-inspired intelligent olfactory biomimetic sensing system for gas recognition in complex environments. Microsyst. Nanoeng. 2024, 10, 153.
68. Wang, C.; Chen, Z.; Chan, C. L. J.; et al. Biomimetic olfactory chips based on large-scale monolithically integrated nanotube sensor arrays. Nat. Electron. 2024, 7, 157-67.
69. Zhao, W.; Li, J.; Xue, Z.; et al. A separation-sensing platform performing accurate diagnosis of jaundice in complex biological tear fluids. Angew. Chem. Int. Ed. Engl. 2022, 61, e202205628.
70. Lee, J. C.; Kim, S. Y.; Song, J.; et al. Micrometer-thick and porous nanocomposite coating for electrochemical sensors with exceptional antifouling and electroconducting properties. Nat. Commun. 2024, 15, 711.
71. Zhou, L.; Hou, H.; Wei, H.; et al. In Vivo Monitoring of oxygen in rat brain by carbon fiber microelectrode modified with antifouling nanoporous membrane. Anal. Chem. 2019, 91, 3645-51.
72. Han, Q.; Wang, H.; Wang, J. Multi-mode/signal biosensors: electrochemical integrated sensing techniques. Adv. Funct. Mater. 2024, 34, 2403122.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].