fig3
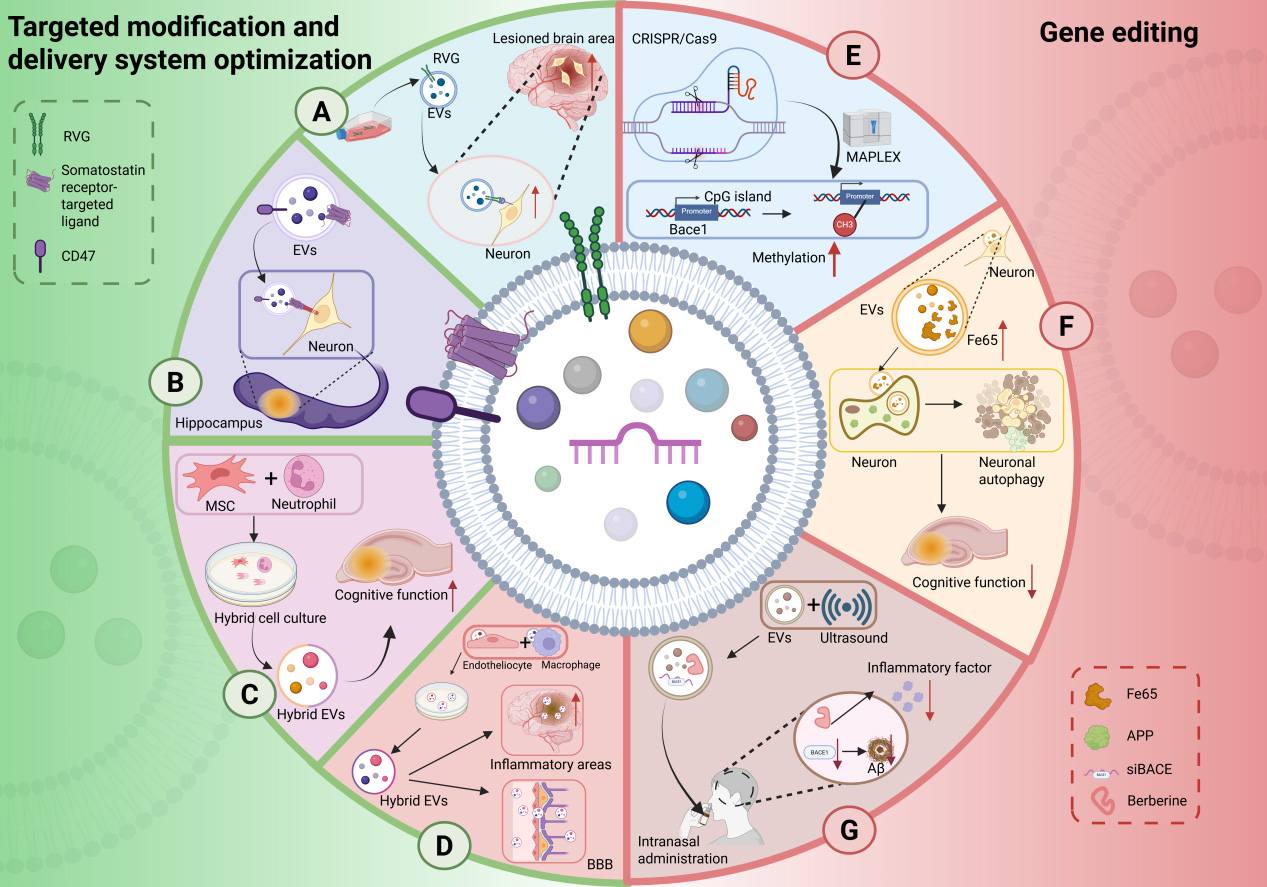
Figure 3. EV engineering strategies: targeted modification and gene editing. (A) Surface modification with RVG peptide enhances neuron-specific binding of EVs; (B) Expression of somatostatin receptor ligands enables hippocampus-specific EV delivery, while loading of CD47 ligands reduces their immune clearance and prolongs circulatory half-life; (C) EVs derived from mesenchymal stem cell-neutrophil hybrid cells target brain regions affected by AD; (D) Hybrid EVs generated by fusing membranes from brain microvascular endothelial cells and macrophage-derived EVs exhibit dual capabilities, which can efficiently cross the blood-brain barrier and accumulate in the inflamed areas of the brain, promoting the clearance of Aβ; (E) The CRISPR/Cas9 system is utilized to epigenetically target and methylate the BACE1 promoter, thereby reducing its expression; (F) Engineered hippocampal neuron-derived EVs overexpressing the Fe65 protein induce autophagy in APP-expressing neurons; (G) Ultrasound-mediated preparation generates EVs co-loaded with BACE1 siRNA and the anti-inflammatory drug berberine, enabling synergistic therapy upon intranasal administration. RVG: Rabies virus glycoprotein; EV: extracellular vesicle; MSC: mesenchymal stem cell; BBB: blood-brain barrier; MAPLEX: mMaple3-mediated protein loading into and release from exosomes; APP: amyloid precursor protein; Aβ: amyloid-beta; BACE1: beta-site amyloid precursor protein cleaving enzyme 1; CRISPR: clustered regularly interspaced short palindromic repeats; Cas9: CRISPR-associated protein 9; siRNA: small interfering RNA. Created in BioRender. yinghan, h. (2025) https://BioRender.com/br23jn3