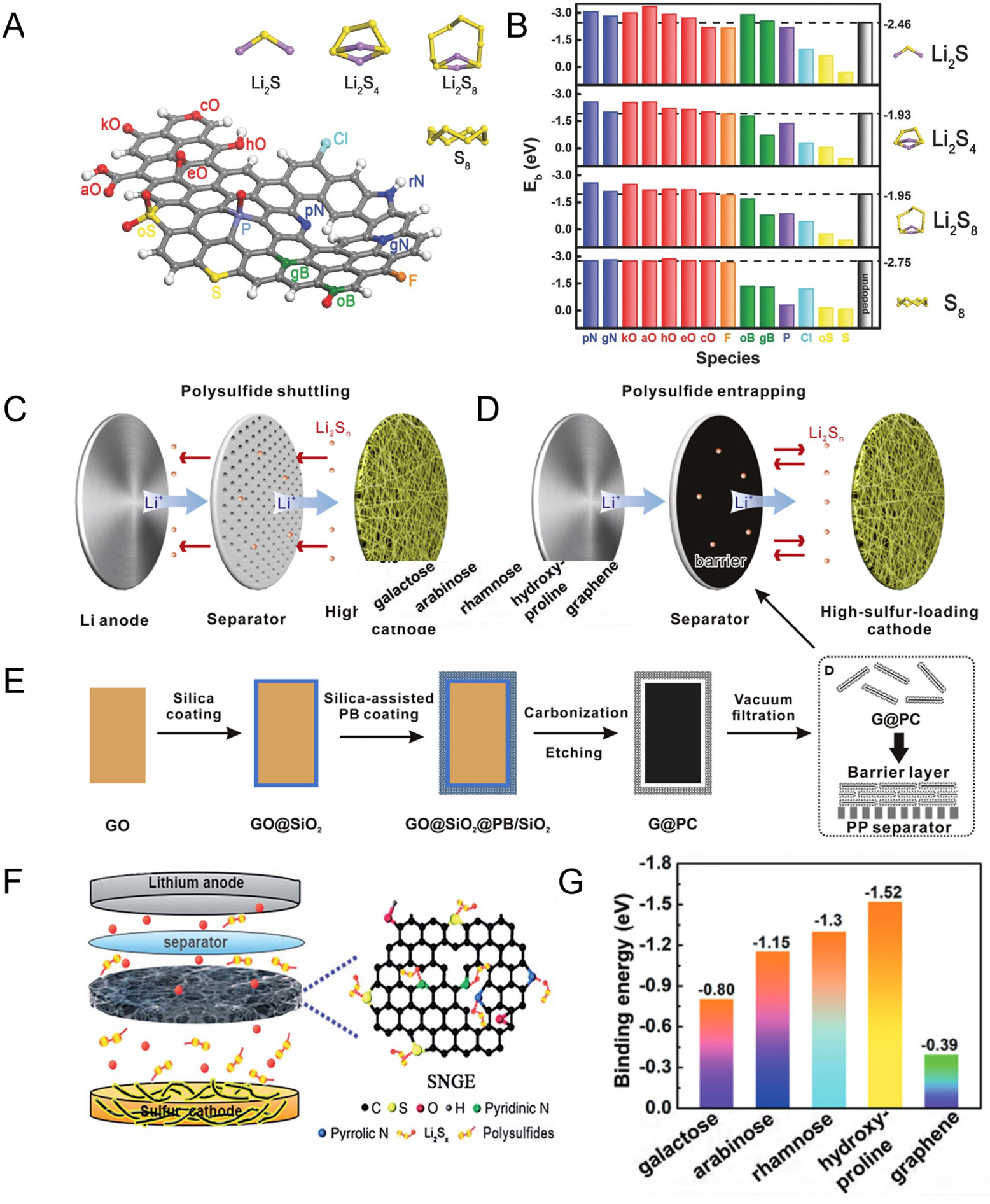
fig9
Figure 9. (A) The schematic diagram of X-doped nanocarbon materials (X = N, O, F, B, P, S, Cl) and PBE level optimized structure of Li2S, Li2S4, Li2S8, S8 molecules; (B) The binding energies of Li2S, Li2S4, Li2S8, and S8 interacting with X-doped GNRs and pristine (undoped) GN. Reproduced with permission; (A and B) are reprinted with permission from Ref[247], Copyright © 2016 Wiley; (C) Schematic illustration of shuttling effect; (D) The separator with a barrier layer for suppressing the polysulfide migration; (E) The synthetic procedures for the G@PC nanosheets. Reproduced with permission; (C-E) are reprinted with permission from Ref[249], Copyright © 2018 Elsevier; (F) Schematic of the electrode configuration for the LSB with the SNGE interlayer. Reproduced with permission; (F) are reprinted with permission from Ref[250], Copyright © 2025 Royal Society of Chemistry; (G) The summary of binding energies between Li2S4 and GA/graphene; (G) are reprinted with permission from Ref[250], Copyright © 2018 Wiley. GO: Graphene oxide; PC: porous carbon nanosheet; PB: Prussian blue; PP: polypropylene; SNGE: sulfur-nitrogen dual-doped graphene; GNR: graphene nanoribbon; GN: graphene nanoplatelet; PBE: Perdew-Burke-Ernzerhof; GA: graphene aerogel; LSB: lithium-sulfur battery.