Thermal stability and safety challenges of all-solid-state batteries
Abstract
All-solid-state batteries (ASSBs) are widely regarded as promising candidates for next-generation energy storage owing to their high energy density and intrinsic safety advantages. However, growing evidence indicates that ASSBs are not inherently safe. The thermal instability of key materials and interfaces can still trigger severe thermal hazards under abusive conditions. In this review, we systematically examine the thermal stability of critical components in ASSBs and elucidate the underlying safety challenges and thermal runaway mechanisms from the materials level to the full-cell and system levels. Current strategies for improving thermal stability and safety, including materials design, interface engineering, and cell-level protection, are summarized. In addition, emerging approaches based on advanced thermal characterization techniques and data-driven modeling are highlighted. This review provides a unified framework for understanding the thermal failure behavior of ASSBs and offers practical guidance for the rational design of future energy storage technologies.
Keywords
INTRODUCTION
The increasing demand for high-energy-density and high-safety energy storage systems in electric vehicles, portable electronics, and grid applications has driven intensive research into all-solid-state batteries (ASSBs)[1-4]. By replacing flammable liquid electrolytes (LEs) with solid-state electrolytes (SSEs), ASSBs offer the potential for higher energy density, wider operating temperature windows, and improved safety[5-7]. The ability to pair high-voltage cathodes with lithium metal anodes further ranks these systems among leading candidates for next-generation batteries[8-10]. Significant progress has been achieved in the development of various SSEs with high ionic conductivity, accelerating the practical deployment of ASSB technologies[11-14].
Despite these advances, growing evidence indicates that ASSBs are not intrinsically safe and can still experience severe thermal hazards[7,15,16]. Under abusive conditions such as internal short circuits, overcharging, and mechanical damage, substantial heat generation, exothermic decomposition, oxygen or sulfur release, and thermally accelerated interfacial reactions may occur[17-20]. The rigid nature of SSEs and the prevalence of solid-solid interfaces introduce additional complexity, promoting localized heat accumulation and triggering cascading degradation[21]. These characteristics result in unique thermal runaway mechanisms that are distinct from those in conventional liquid electrolyte-based lithium-ion batteries (LELIBs)[22].
Accordingly, a comprehensive understanding of the thermal stability and safety of ASSBs across materials, interfaces, and cell architectures is urgently needed. In this review, we systematically summarize the thermal stability of key components and the critical safety challenges of ASSBs. Current strategies for improving thermal stability and safety through materials design, interface engineering, and cell-level protection are further examined. Finally, future research directions for the realization of intrinsically safe, high-energy ASSBs are highlighted. By integrating recent experimental observations, theoretical insights, and engineering perspectives, this review provides a unified framework for understanding the thermal failure mechanisms of ASSBs and establishes practical guidelines for the rational design of safer solid-state energy storage systems.
FUNDAMENTALS OF THERMAL STABILITY IN ASSBS
Thermal stability in ASSBs is governed by a complex interplay of material properties, interfacial chemistry, and cell-level architecture[7,21]. Unlike conventional LELIBs, in which the presence of flammable LEs dominates thermal behavior, ASSBs rely on SSEs, whose intrinsic thermochemical, mechanical, and electrochemical characteristics dictate their response to heat generation and external thermal stimuli. Moreover, owing to diverse material selections and cell structural designs, the thermal stability of different ASSB systems usually varies greatly. Understanding these fundamentals is essential for evaluating how ASSBs behave under normal operation, fast charging, high-power demand, or abuse conditions.
Key components in ASSBs
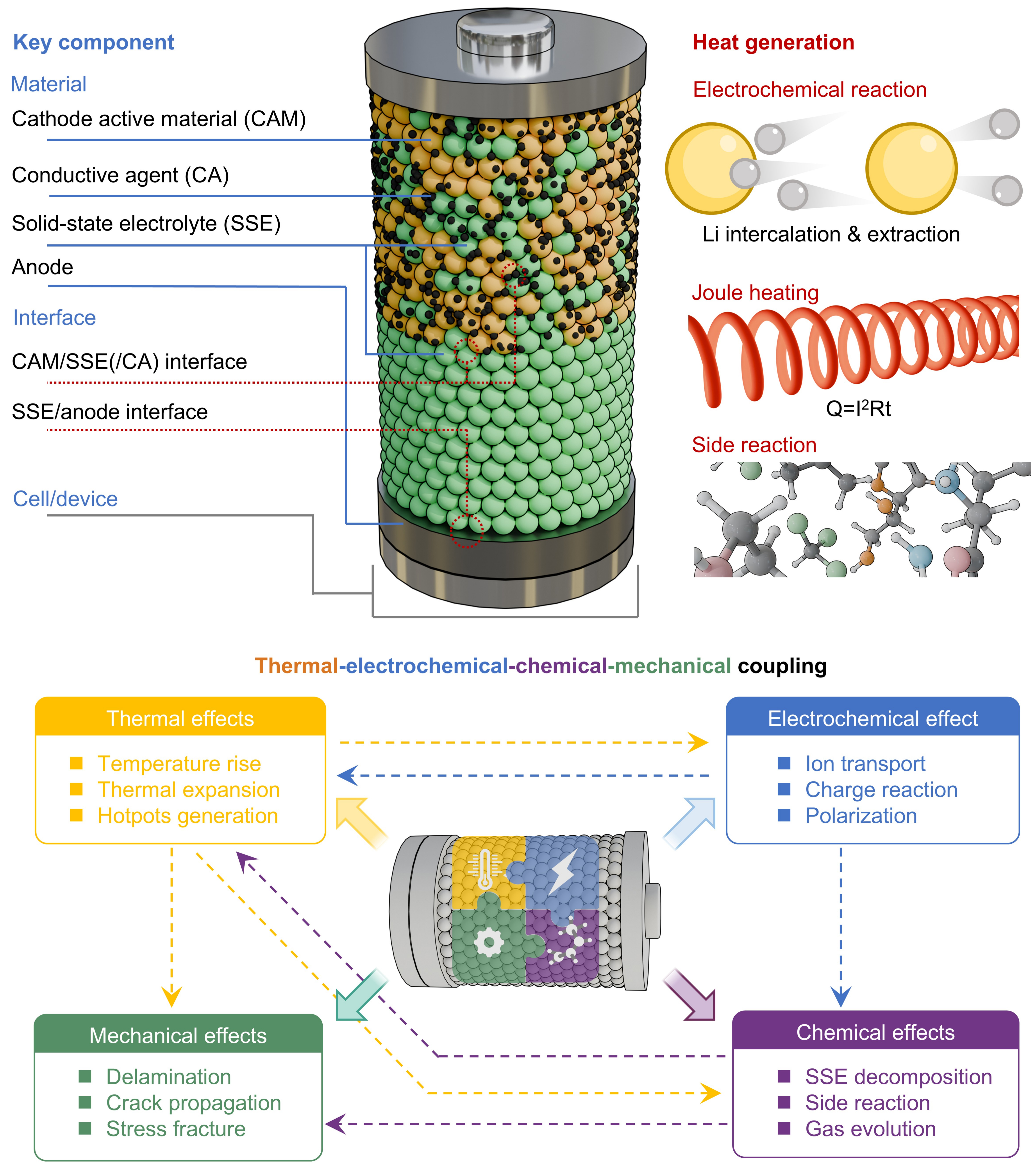
The configuration and key components of a typical ASSB are first briefly introduced prior to discussing thermal stability issues. As illustrated in Figure 1, a prototypical ASSB consists of a multilayer architecture in which a solid electrolyte layer is sandwiched between a cathode composite and an anode[23]. The cathode composite generally includes active material particles, an SSE (denoted as a catholyte) for ionic percolation, and in certain cases a conductive agent (carbonaceous materials). The SSE separator forms a dense, ion-conducting barrier that prevents electronic shorting while enabling Li-ion transport. The selection of the anode prioritizes lithium metal or alloys and Si-based materials due to their high capacity. This solid-state stacked configuration defines the electrochemical, mechanical, and thermal environment of the ASSB and underpins its overall performance and safety. Accordingly, the thermal stability of ASSBs can be analyzed at multiple hierarchical levels, including the material, interface, and cell/system levels, as discussed in the following sections.
Heat generation in ASSBs
Heat generation in ASSBs arises from a combination of electrochemical, resistive, and parasitic processes, and its magnitude directly influences thermal stability, cycling performance, and overall safety[24]. Unlike liquid-based systems, where convection and solvent evaporation contribute to heat dissipation, ASSBs typically employ dense ceramic or composite architectures with limited thermal conductivity, rendering the management of internally generated heat particularly critical. Understanding the sources, mechanisms, and temperature dependence of heat generation is therefore essential for predicting thermal behavior and designing stable ASSB systems.
Heat generation in ASSBs can be broadly categorized into reversible and irreversible contributions. The reversible component, often referred to as entropic heat, originates from entropy changes associated with electrochemical reactions (i.e., lithium intercalation and extraction)[25,26]. This term can be endothermic or exothermic, depending on the electrode chemistry and state of charge (SoC). Although reversible heat generally contributes modestly to the overall temperature rise under moderate cycling rates, its effect becomes non-negligible in high-energy cathodes exhibiting large entropy variations or at elevated operating temperatures.
Irreversible heat generation is typically the dominant contributor and stems from several mechanisms. Joule (ohmic) heating, arising from ionic and electronic resistances within electrodes, solid electrolytes, and interfaces, increases quadratically with current and is strongly influenced by the higher interfacial resistances in ASSBs compared to liquid systems[27,28]. In addition, parasitic reactions, including thermally activated electrolyte decomposition, interphase formation, and side reactions, produce exothermic heat that can accelerate degradation or initiate thermal runaway under abusive conditions.
Thermal-electrochemical-chemical-mechanical coupling
Temperature plays a central role in controlling the coupled electrochemical, chemical, and mechanical processes in ASSBs[29]. Variations in temperature strongly influence electrochemical transport properties, interfacial reactions, and structural integrity, leading to complex multiphysics interactions during battery operation, as illustrated in Figure 1. Understanding these coupled effects is therefore essential for evaluating the thermal stability and safety of ASSBs.
Electrochemically, temperature strongly influences ion transport and reaction kinetics in ASSBs. The ionic conductivity of most SSEs follows Arrhenius-type behavior, increasing exponentially with temperature[30]. Moderate temperature elevation can improve ionic transport and reduce interfacial resistance, thereby enhancing electrochemical performance. However, excessive temperatures may accelerate undesirable processes such as interfacial reactions, phase transitions, and electrolyte decomposition. Charge-transfer reactions at electrode-electrolyte interfaces are also highly temperature-dependent; although higher temperatures reduce activation barriers, they can promote the formation of unstable interphases, particularly at lithium metal/SSE and high-voltage cathode/SSE interfaces. Meanwhile, electrochemical processes generate heat through resistive losses, which increase with current density and internal resistance. This thermal-electrochemical interplay can create positive feedback loops, where local hotspots accelerate degradation and generate additional heat, eventually leading to localized failure. Such effects are especially critical in sulfide-based systems, where thermally activated decomposition reactions may release heat and gases, further worsening thermal instability.
Temperature also significantly affects chemical stability within ASSBs. Elevated temperatures can accelerate parasitic reactions between electrodes and SSEs, leading to electrolyte decomposition, gas evolution, and the formation of resistive interphases[31]. In sulfide-based systems, for example, thermally activated reactions between sulfide electrolytes and oxide cathodes may release heat and gaseous products, further destabilizing the system[32]. These chemical reactions not only degrade electrochemical performance but also contribute additional heat, thereby reinforcing thermal-chemical feedback processes.
Beyond electrochemical and chemical effects, mechanical processes are also tightly coupled with thermal behavior. Increasing temperature can induce thermal expansion of materials, leading to mechanical stresses at electrode-electrolyte interfaces[33,34]. Due to the rigid nature of SSEs and electrodes, mismatches in thermal expansion coefficients among different components may result in microcracks, interfacial delamination, or loss of interfacial contact. Such structural degradation can increase local resistance, generate non-uniform current distributions, and expose fresh reactive surfaces, thereby accelerating both electrochemical degradation and chemical side reactions. Furthermore, side reactions and material decomposition at interfaces can induce non-uniform distributions of interfacial stress and finally contact loss[35].
Therefore, the thermal behavior of ASSBs is governed by a complex thermal-electrochemical-chemical-mechanical coupling mechanism. Interactions among these processes can lead to nonlinear responses in cell impedance, interfacial stability, and heat generation. A comprehensive understanding of these coupled phenomena is essential for predicting battery behavior under practical operating conditions and for designing ASSB systems with improved thermal stability and safety.
THERMAL STABILITY OF KEY COMPONENTS
The thermal stability of key components in ASSBs is central to determining their safety and operational limits. Due to the chemical diversity of materials in ASSBs, including SSEs, electrode active materials, and interfacial layers, their thermal behaviors vary widely in both mechanism and severity. These differences influence heat generation, interfacial reactions, and mechanical integrity under thermal stress. The overall thermal stability of an ASSB can be assessed based on the decomposition temperatures and phase transition stability of key materials, as well as the exothermic intensity and onset temperature of interfacial reactions. A clear understanding of how each component responds to temperature is therefore essential for evaluating failure modes and guiding the design of safer ASSB architectures.
SSE
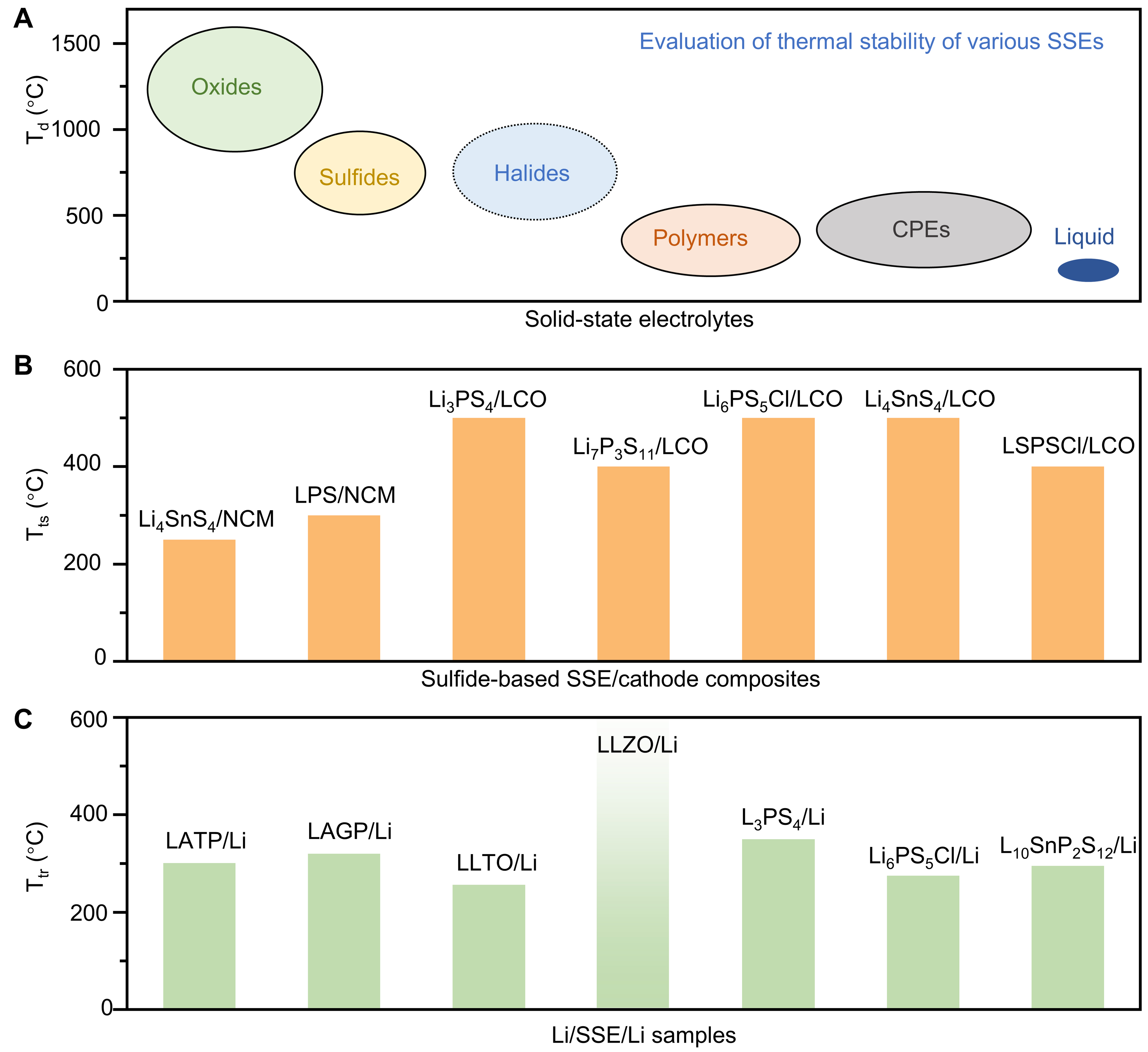
In principle, the thermal stability of SSEs largely determines the safe operating temperature window of ASSBs. The thermal stability of mainstream SSEs, including oxides, sulfides, halides, polymers, and composite polymers, is compared in Figure 2A, based on their decomposition temperatures Td (or high-impedance phase transition temperatures and melting points). Generally, inorganic SSEs exhibit superior thermal stability compared to their polymer-based counterparts. For instance, oxide-based SSEs such as Li6.6La3Zr1.6Ta0.4O12 (LLZO) have theoretical decomposition temperatures exceeding 1,500 °C[36]. Sulfide-based SSEs are also highly stable, with decomposition temperatures typically ranging from approximately 500 to 900 °C[37]. To date, the thermal stability of halide-based SSEs has been rarely studied. Based on existing publications, most halide SSEs exhibit high ionic conductivity and maintain structural integrity above 400 °C. It can therefore be inferred that their actual decomposition temperatures are likely higher than the tested ranges[38]. In Figure 2A, this estimated temperature range for halides is indicated by dashed circles. Polymer-based SSEs are intrinsically limited, with decomposition temperatures typically ranging from 200 to 300 °C. While the addition of inorganic fillers to form composite polymer electrolytes (CPEs) can improve thermal stability to a degree, the fundamental thermal limitations of the polymer matrix remain.
Figure 2. (A) Comparison of thermal stability among mainstream SSEs and LEs, in which Td refers to the decomposition temperature of the material based on experimental or calculated values. The Td values of halide-based SSEs are estimated from studies on their high-temperature performance and are therefore indicated by dashed circles. Adapted from Ref.[38] under a CC BY 4.0 license; (B) Summary of the thermal stability of representative sulfide-based SSE/CAM composites, indicated by the thermally stable temperature (Tts). Reproduced with permission based on data from Ref.[32,42,43]; (C) Summary of the thermal stability of representative Li/SSE/Li composites, indicated by the critical temperature of thermal runaway (Ttr; identified as T2 in ARC tests). Reproduced with permission based on data from Ref.[45,46]. SSEs: Solid-state electrolytes; LEs: liquid electrolytes; CAM: cathode active material; ARC: accelerating rate calorimeter; CPEs: composite polymer electrolytes; NCM: LiNixCoyMn1-x-yO2; LPS: 75Li2S·25P2S5; LCO: LiCoO2; LSPSCl: Li9.54Si1.74P1.44S11.7Cl0.3; LATP: Li1.4Al0.4Ti1.6(PO4)3; LAGP: Li1.5Al0.5Ge1.5(PO4)3; LLTO: Li3xLa2/3-xTiO3; LLZO: Li6.4La3Zr1.4Ta0.6O12.
Composite and interface
Composite structures and interfaces constitute some of the most thermally sensitive regions in ASSBs. Their stability depends not only on the intrinsic properties of the constituent materials but also on the quality of physical contact, chemical compatibility across phase boundaries, and the evolution of these factors under thermal stress. In particular, because the composite cathode provides the largest interfacial contact area in ASSBs, its thermal stability strongly influences the overall thermal stability of the cell. Compared to oxide-based SSEs, sulfide-based SSEs provide high ionic conductivity and excellent deformability, which enable intimate mixing with cathode particles. However, sulfide-based composite cathodes usually experience interfacial incompatibility due to the intrinsic chemical reactivity of sulfide electrolytes with cathode materials, especially at elevated temperatures. Figure 2B presents representative thermal stability data for composite cathodes composed of various sulfide-based SSEs and cathode active materials
The SSE/lithium metal interface at the anode side is another critical region where thermal instability can dictate overall safety. Elevated temperatures accelerate the chemical decomposition of the electrolyte in contact with lithium metal, which can thicken the interphase and increase interfacial resistance. Heat can also enhance lithium diffusion into the SSE, leading to interfacial void formation, uneven deposition, and eventual short circuiting. Figure 2C summarizes the thermal stability of various SSE/lithium composites, expressed as the critical temperature of thermal runaway (Ttr), defined as the onset temperature associated with rapid and substantial heat release, as measured by accelerating rate calorimeter (ARC) tests[45,46]. Most SSE/lithium composites exhibit signs of thermal runaway within a narrow temperature window of roughly 270 to 350 °C. In contrast, Li6.4La3Zr1.4Ta0.6O12 (LLZO) demonstrates exceptionally high thermal stability against lithium metal, with no thermal runaway observed in ARC tests. According to thermodynamic calculations, the extremely high thermal stability of the LLZO/lithium interface can be attributed to limited oxygen release at elevated temperatures and low chemical reactivity with lithium metal[47]. Compared with the cathode side, the lithium metal anode side of ASSBs presents more serious challenges in thermal stability and risks of thermal runaway, primarily due to the low melting point of lithium metal and the potential oxygen release from SSEs at elevated temperatures[48,49].
SAFETY CHALLENGES OF ASSBS
The elimination of flammable LEs significantly reduces certain hazards, but new safety challenges emerge from the use of SSEs, solid-solid interfaces, and high-energy-density cell designs. From a thermal management perspective, the safety of ASSBs is ultimately determined by the balance between internal heat generation and heat dissipation[50,51]. Therefore, cell design plays a critical role in battery safety, since it controls the generation, accumulation, and propagation of heat within the cell, as well as the efficiency of heat dissipation to the surrounding environment[52]. More importantly, thermal runaway in ASSBs can be triggered by various abuse conditions, including mechanical, electrical, and thermal stresses. Consequently, developing targeted mitigation strategies to address these external abuse scenarios is essential for improving the safety of ASSBs. In this section, a systematic analysis of ASSB safety is provided to clarify the underlying safety challenges in ASSBs, which typically arise from complex processes involving multiple coupled factors.
Liquid vs. solid
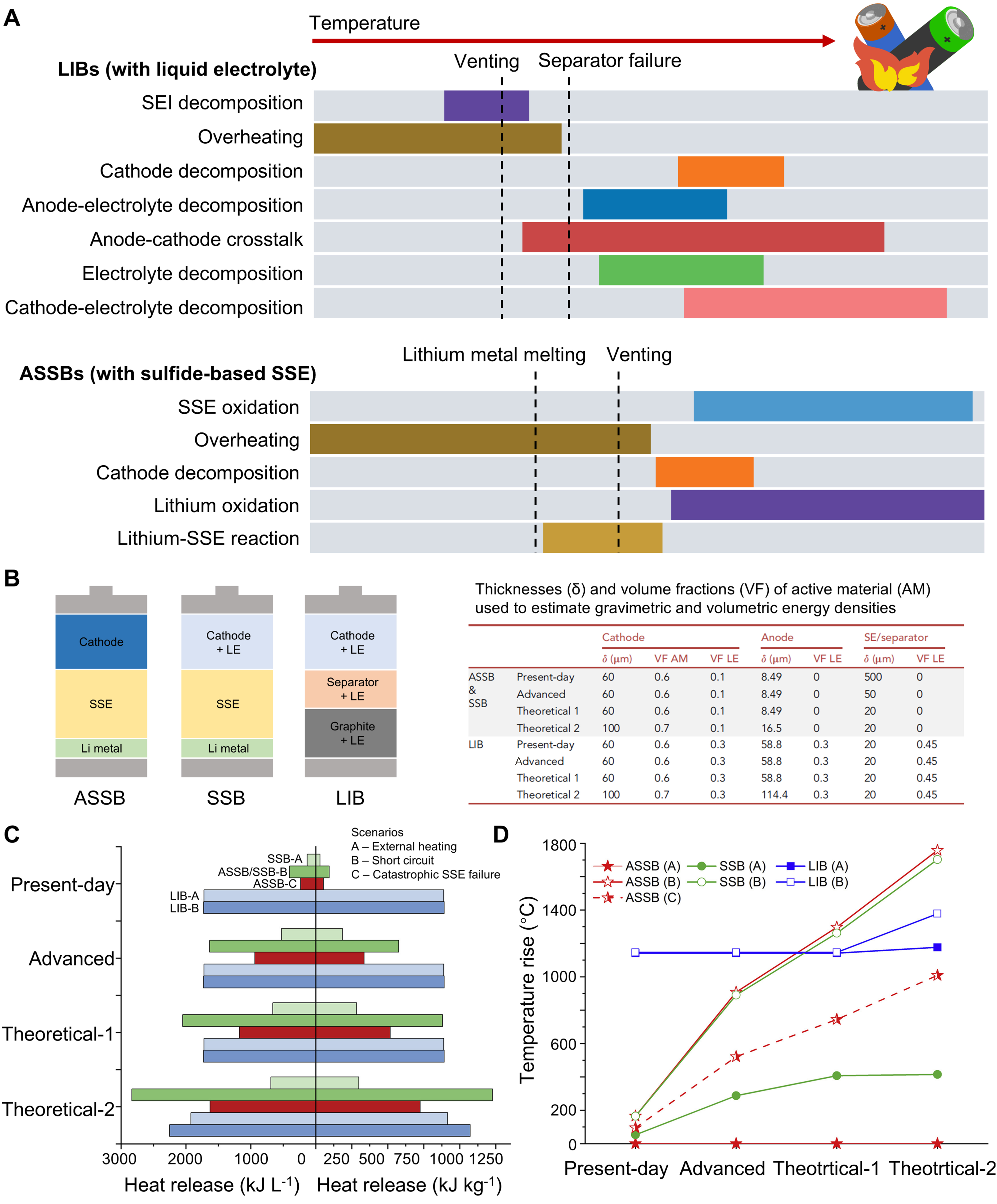
The replacement of flammable LEs with SSEs is widely regarded as a major safety advantage of ASSBs compared with conventional LELIBs. However, ASSBs do not fully eliminate the inherent safety risks associated with batteries as energy-dense and highly chemically active systems. Instead, the application of higher-energy-density electrode materials in ASSBs may introduce additional safety concerns. Figure 3A compares the temperature-dependent thermal failure sequences of LELIBs and ASSBs employing sulfide-based SSEs[53]. In LE systems, increasing temperature triggers a cascade of exothermic processes, including decomposition of the solid electrolyte interphase (SEI), degradation of the electrolyte and electrodes, separator failure, and eventual venting and combustion. These overlapping reactions occur over a broad temperature range and are strongly amplified by the presence of flammable solvents. In contrast, ASSBs eliminate solvent combustion and separator melting, but introduce distinct thermally driven failure modes, such as lithium metal melting, solid electrolyte oxidation, and reactions between lithium and the solid electrolyte. As a result, thermal instability in ASSBs is governed by interfacial chemical reactions and lithium metal behavior, rather than solvent ignition, leading to delayed but still significant heat release at elevated temperatures.
Figure 3. (A) Stages of thermal runaway for conventional LIBs and SSBs under a thermal abuse condition. Adapted from Ref.[53] under a CC BY 4.0 license; (B) Configurations of ASSB, SSB, and LIB systems, along with key parameters of different cell formats with varied component thicknesses and volume fractions; (C) Gravimetric and volumetric potential heat release as a function of cell format; (D) Potential temperature rise as a function of cell format and increasing energy density. (B-D) are adapted from Ref.[54], Copyright © 2022 Elsevier Inc. LIBs: Lithium-ion batteries; SSBs: solid-state batteries; ASSB: all-solid-state battery; SEI: solid electrolyte interphase; LE: liquid electrolyte.
For a quantitative evaluation and comparison of the safety of LELIBs and ASSBs, Bates et al. applied thermodynamic models to assess heat release and temperature rise under different failure scenarios[54]. Three battery configurations were designed for safety assessment, as illustrated in Figure 3B. In these configurations, the ASSB contains no LE, the SSB includes LE within the cathode layer, and the conventional LIB contains LE in the anode, separator, and cathode layers. In addition, multiple cell formats were examined for each battery type by varying the thicknesses of the cathode, anode, and SSE/separator layers. Figure 3C compares the gravimetric and volumetric heat release of various cell formats under different failure scenarios, based on variations in SSE thickness and cathode volume fraction or thickness, as illustrated in Figure 3B. The results show that in high-energy-density systems, represented by the Theoretical 1 and Theoretical 2 formats, ASSBs and SSBs with LE can release more heat than conventional LIBs under extreme conditions, such as internal short circuits. The corresponding potential temperature rise for different cell formats is presented in Figure 3D. In these high-energy-density designs, ultra-high temperatures exceeding 1,600 °C are predicted for ASSBs and SSBs during short-circuit failures, suggesting that, under such conditions, these systems may exhibit lower thermal safety margins than conventional LIBs. Although this thermodynamic model was initially developed for an ASSB configuration consisting of a lithium metal anode, LLZO electrolyte, and NCM cathode, the framework can be extended to other battery systems with different SSEs and electrode chemistries by incorporating the relevant reaction pathways and associated heat-release processes.
Thermal runaway mechanisms of ASSBs
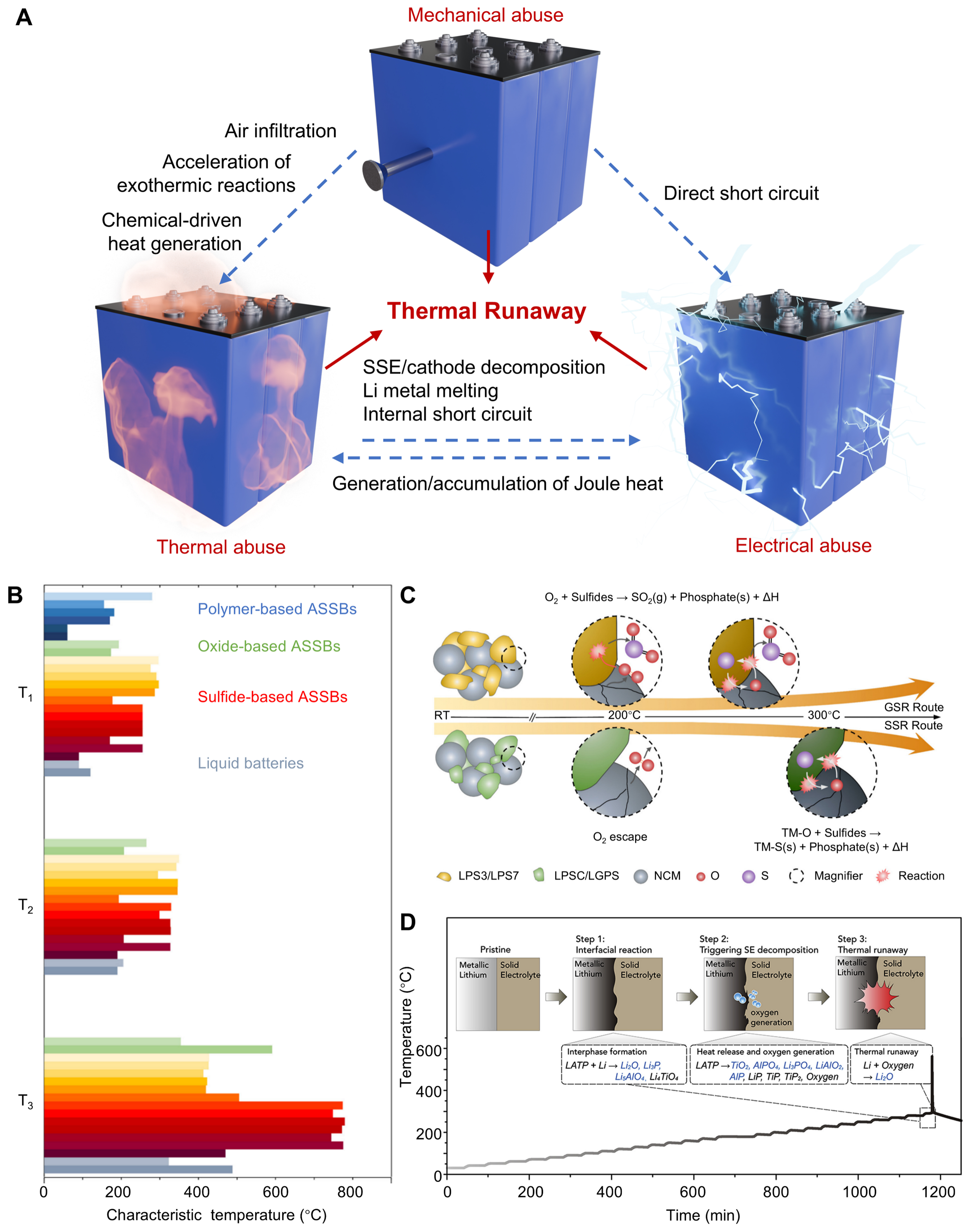
As discussed above, ASSBs cannot currently be regarded as intrinsically safe systems. Accordingly, a thorough investigation and understanding of their thermal runaway mechanisms are urgently needed. In practice, a battery or battery pack is a highly complex system that integrates diverse materials and electrode architectures across multiple length scales. Consequently, thermal runaway in ASSBs is inherently a complex, multistep process. Drawing on experience from LELIBs, the thermal behavior of a single battery is generally influenced by external operating conditions, including thermal, mechanical, and electrical abuse, as illustrated in Figure 4A[55]. These three types of abuse are not independent but are frequently strongly coupled and interconvertible[56]. In particular, mechanical failure may allow air ingress into the battery, thereby accelerating exothermic interfacial reactions or promoting direct cathode-anode interactions, both of which increase heat generation through chemical pathways. Severe mechanical damage, such as nail penetration, can also directly induce internal short circuits, leading to electrical abuse. The resulting short circuit generates a large amount of Joule heat within a short time, which can rapidly trigger thermal runaway[57,58]. Conversely, even under relatively mild conditions, accumulated heat may cause solid electrolyte decomposition, cathode degradation, and lithium metal melting, ultimately inducing internal short circuits. Overall, despite the strong coupling among thermal, mechanical, and electrical abuse, the fundamental origin of battery thermal runaway lies in the intrinsic thermal responses of materials and interfaces, unexpected heat generation, and uncontrolled heat accumulation.
Figure 4. (A) Schematic diagram of thermal runaway triggers of ASSBs at the cell level. Adapted from Ref.[55] under a CC BY 4.0 license; (B) Comparison of characteristic temperatures of ASSBs and liquid batteries during ARC tests. Adapted from Ref.[59] under a CC BY 4.0 license; (C) Schematic diagram of two distinct failure routes of different sulfide SSEs with NCM. Adapted from Ref.[60], Copyright © The Royal Society of Chemistry 2023; (D) Schematic illustration of multistep thermal runaway reactions between oxide SSEs and metallic lithium. Adapted from Ref.[45], Copyright © 2020 Elsevier Inc. ASSBs: All-solid-state batteries; ARC: accelerating rate calorimeter; SSEs: solid-state electrolytes; NCM: LiNixCoyMn1-x-yO2; LPS: 75Li2S·25P2S5; LPSC: Li6PS5Cl; LGPS: Li10GeP2S12; LATP: Li1.4Al0.4Ti1.6(PO4)3.
Figure 4B summarizes key thermal parameters T1, T2, and T3 obtained from ARC tests for various ASSBs with different configurations[59]. These parameters provide important indicators of thermal behavior. Specifically, T1 corresponds to the onset temperature of detectable self-heating, T2 represents the temperature at which rapid self-heating begins, and T3 reflects the maximum temperature reached during the runaway process, which is related to the overall severity of heat release. Although ARC measurements are conducted under controlled conditions, these parameters serve as useful indicators for practical abuse scenarios, where T1/T2 relate to the initiation of thermally activated reactions and thermal runaway, and T3 reflects the severity of heat release once runaway occurs.
Relative to other systems, sulfide-based ASSBs exhibit noticeably higher T2 and T3 values, indicating more severe thermal runaway behavior once abuse occurs. In response to this concern, sulfide-based ASSBs have attracted significant research attention in recent years, particularly regarding their thermal runaway mechanisms[16,60-64]. Specifically, Rui et al. revealed two distinct thermal runaway mechanisms in sulfide-based ASSBs, namely gas-solid and solid-solid reactions[60]. As shown in Figure 4C, for LPS3 (Li3PS4) and LPS7 (Li7P3S11), oxygen released from fully charged NCM cathodes at about 200 °C oxidizes the sulfide-based electrolytes, triggering gas-phase reactions that generate SO2 and phosphates with substantial heat release, followed by further redox reactions at higher temperatures. In contrast, LPSC (Li6PS5Cl) and LGPS (Li10GeP2S12) are initially stable against oxygen release, but undergo solid-state reactions with NCM decomposition products at around 300 °C, producing large amounts of heat without noticeable SO2 evolution.
Oxide-based SSEs are generally considered to have better thermal stability than sulfide-based counterparts; however, the thermal runaway behavior of oxide-based ASSBs cannot be neglected. Chen et al. elucidated the thermal runaway mechanism of ASSBs employing LATP SSE, as illustrated in Figure 4D[45]. Interphases initially form upon contact with lithium metal, and elevated temperatures accelerate interfacial reactions, especially after lithium melting, which promotes electrolyte decomposition and increases oxygen activity. Lithium melting can further accelerate interfacial reactions between lithium and LATP by inducing mechanical stress and interfacial cracking that expose fresh electrolyte surfaces[15,65]. Meanwhile, the increased mobility of molten lithium enlarges the effective contact area with the electrolyte, further promoting thermally activated reactions. The subsequent highly exothermic reaction between released oxygen and metallic lithium generates substantial heat and ultimately triggers thermal runaway.
STRATEGIES TO IMPROVE THERMAL STABILITY AND SAFETY
Enhancing the thermal stability and safety of ASSBs requires a comprehensive approach covering materials selection, interface regulation, and cell-level protection. Because thermal runaway often originates from coupled chemical reactions and heat accumulation at interfaces, effective mitigation strategies must suppress thermally activated reactions, limit heat generation, and prevent the propagation of localized thermal events. Recent advances demonstrate that improvements at multiple levels can synergistically enhance the overall thermal safety of ASSBs.
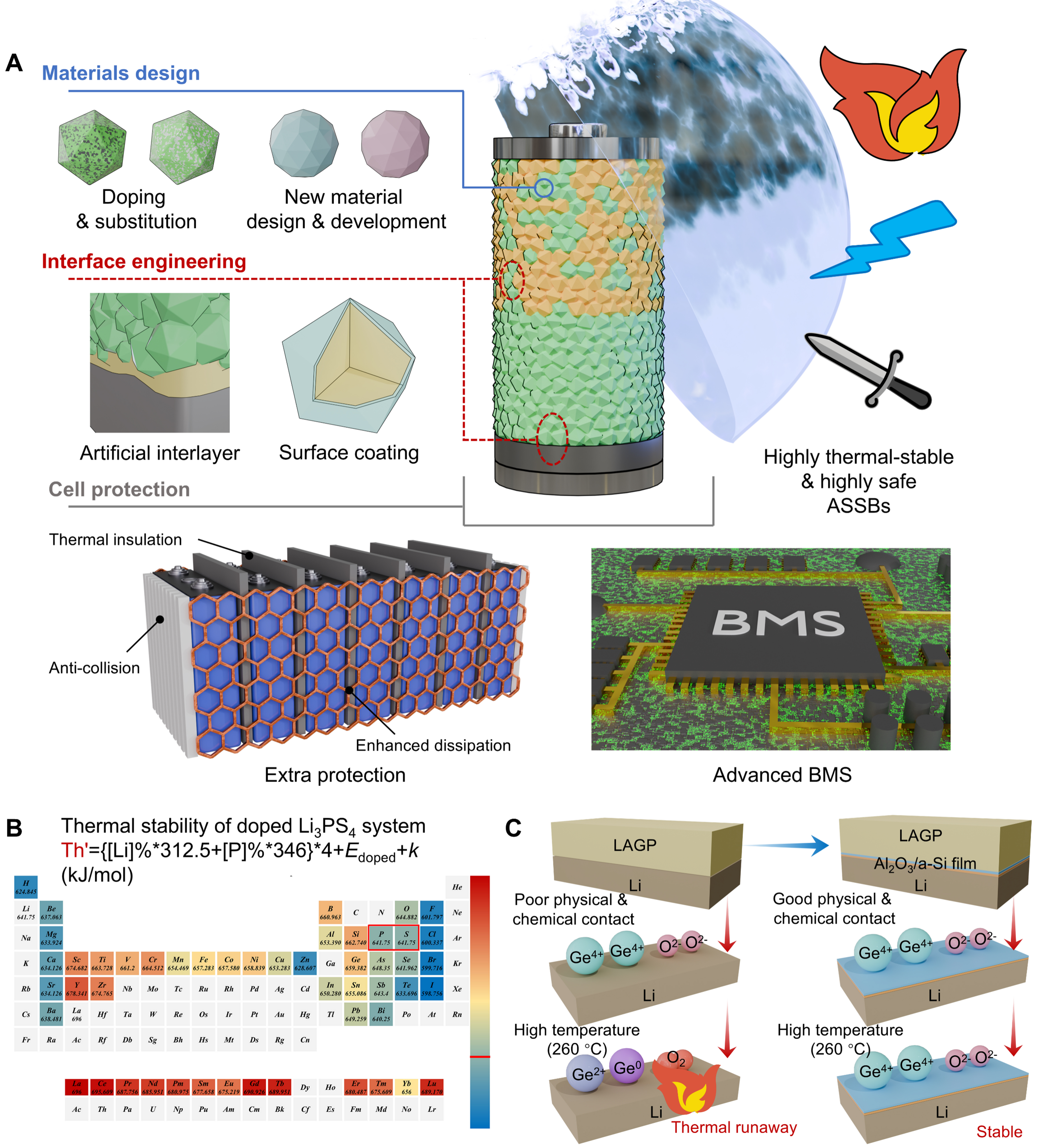
Material design
Material design provides the most fundamental pathway to improving the thermal stability of batteries. For SSEs, compositional tuning through elemental substitution or doping is regarded as a highly effective strategy to enhance thermal robustness and suppress oxygen or sulfur release at elevated temperatures, as shown in Figure 5A. As a representative example, Wang et al. proposed a conceptual parameter (Th′) to quantify the thermal stability of Li3PS4 doped with hetero-elements[37]. The calculated Th′ values for Li3PS4 doped with various elements are summarized in Figure 5B, where larger Th′ values, indicated by redder colors, correspond to enhanced thermal stability. Furthermore, the reliability of this theoretical prediction was validated through experimental evaluations of Li3PS4 doped with Cu, Si, Sn, and O.
Figure 5. (A) Strategies to improve the thermal stability and safety of ASSBs through material design, interface engineering, and cell protection. The subfigure illustrating additional protection is adapted from Ref.[55] under a CC BY 4.0 license; (B) Thermal stability parameter (Th′) values of Li3PS4 modified by doping, where the values are marked below each element symbol across the periodic table. Adapted from Ref.[37] under a CC BY 4.0 license; (C) Schematic diagram of the artificial Al2O3/a-Si interlayer at the LAGP/lithium metal interface for suppressing thermal runaway. Adapted from Ref.[76], Copyright © 2025 Wiley-VCH GmbH. ASSBs: All-solid-state batteries; a-Si: amorphous Si; LAGP: Li1.5Al0.5Ge1.5(PO4)3; BMS: battery management system.
Developing new types of SSEs represents a more fundamental approach to achieving intrinsically safe ASSBs. In recent years, halide-based SSEs have attracted significant interest owing to their inherent nonflammability originating from halogen chemistry[66,67]. Moreover, halide-based SSEs offer advantages of high-voltage stability, excellent deformability, and desirable dry-air stability[13,68]. State-of-the-art studies have further demonstrated that the poor lithium metal compatibility of common halide-based SSEs (e.g., Li3InCl6) can be effectively addressed through targeted compositional design (e.g., Li3YCl4Br2, Li2ZrCl5.5F0.5)[69-72]. These advances move all-halide-based ASSBs with ultra-high thermal stability and enhanced safety toward practical realization.
Interface engineering
Interface engineering is also essential for improving the thermal stability and safety of ASSBs, since many thermally induced failures originate from chemically and mechanically unstable interfaces [Figure 5A]. Artificial interlayers introduced at electrode-SSE interfaces can effectively regulate interfacial reactions by acting as physical and chemical barriers that suppress direct contact between highly reactive components[73-75]. As shown in Figure 5C, Lin et al. introduced a stable heterogeneous interface layer consisting of Al2O3 and amorphous Si (a-Si) into the Li1.5Al0.5Ge1.5(PO4)3 (LAGP)/lithium interface[76]. This artificial interlayer effectively enhanced the thermal stability of the battery by suppressing the reduction of Ge and O2 release. Surface coatings on CAMs provide another effective strategy to enhance thermal stability[77,78]. Thin layers of oxides or phosphates can isolate sulfide-based solid electrolytes from oxygen-releasing cathodes, thereby suppressing interfacial redox reactions and associated heat release. In addition to chemical passivation, these coatings can improve interfacial mechanical integrity and mitigate thermal expansion mismatch during temperature fluctuations. Together, artificial interlayers and surface coatings are critical for reducing interfacial heat generation and interrupting the positive feedback between temperature rise and interfacial degradation in ASSBs.
Cell protection
Cell-level protection strategies play a crucial role in preventing thermal runaway and limiting its consequences. Drawing on strategies from conventional LIBs, external protection measures are also necessary for ASSBs to mitigate the severity of thermal abuse, such as thermal insulation, anti-collision components, and enhanced cooling systems [Figure 5A][55]. At the pack level, an advanced battery management system (BMS), including effective thermal management and early fault detection, is essential for preventing the propagation of thermal events between adjacent cells. Serving as both the first line of monitoring and the final layer of defense, these protection strategies enable safer operation of ASSBs under practical conditions. Recently, Wang et al. developed a machine learning-based intelligent system that integrates state perception and intelligent cycling with a battery testing station to monitor ASSB states in real time and adaptively optimize subsequent cycling procedures based on operational data from previous cycles[79]. Such an artificial intelligence (AI)-driven framework holds strong potential for safety monitoring of ASSBs, as it enables early detection of abnormal operating behavior, dynamic regulation of cycling conditions, and proactive mitigation of thermal and electrochemical instabilities before critical failure occurs.
CONCLUSION AND OUTLOOK
This review comprehensively examines the thermal stability and safety challenges of ASSBs, with a particular focus on the roles of materials chemistry, interfacial reactions, and multiscale heat generation processes. Although ASSBs eliminate flammable LEs, they are not intrinsically safe. Instead, their thermal behavior is governed by complex interactions among SSEs, electrode materials, and solid-solid interfaces, which collectively determine heat generation, accumulation, and the onset of thermal runaway. At the materials level, the intrinsic thermal stability of SSEs critically influences battery safety, while at the interface level, thermally activated reactions, oxygen or other gas release, and interphase evolution dominate heat generation. At the cell and system levels, the accumulation and propagation of localized thermal events under certain abusive conditions ultimately determine the severity of thermal failure. Correspondingly, strategies to enhance the thermal stability and safety of ASSBs have been developed across material design, interface engineering, and cell protection.
Despite significant progress, major challenges remain in understanding thermal behavior under coupled electrochemical/chemical, mechanical, and thermal fields, especially at solid-solid interfaces. The dynamic evolution of interfacial chemistry and structure at elevated temperatures, as well as the quantitative relationship between interfacial reactions and thermal runaway thresholds, remains poorly understood. Moreover, at the cell level, the detailed transformation mechanisms among the three types of abuse conditions and the quantitative driving forces governing their interconversion remain insufficiently explored. To effectively prevent thermal runaway, the abuse tolerance of individual materials, such as electrodes and solid electrolytes, as well as that of integrated battery systems, must be systematically identified and evaluated. In the near future, several research directions are particularly worthy of further exploration to enable more stable and safer ASSBs.
(i) Achieving high ionic conductivity should not be the sole objective in the rational design of SSEs; thermal and chemical stability must be prioritized. Emerging approaches based on AI and digital twin technology are expected to further accelerate the rational design of ideal SSEs. AI-driven materials discovery and data-driven modeling can rapidly screen compositions and predict key properties, such as ionic conductivity, electrochemical stability, and thermal behavior[80]. Meanwhile, digital twin frameworks that integrate multiphysics models with real-time data offer the potential to simulate and optimize the coupled thermal-electrochemical-chemical-mechanical processes in ASSBs under realistic operating conditions. The combination of these advanced tools with experimental validation may provide a powerful pathway for designing next-generation SSEs with simultaneously improved performance, thermal stability, and safety.
(ii) Synergistic strategies combining material design and interface engineering are expected to play a key role in improving the thermal stability of ASSBs. Optimizing the composition and crystal structure of SSEs and electrode materials can enhance their intrinsic thermochemical stability, while interface modification strategies can effectively suppress undesirable interfacial reactions and heat generation. The combination of these approaches enables the simultaneous stabilization of both bulk materials and interfaces, thereby providing a promising pathway toward thermally robust and safer ASSB systems. Further systematic investigations into the coupling effects between bulk material properties and interfacial stability will be essential for the rational design of high-safety ASSBs. In particular, future research should consider coupled thermal-electrochemical-chemical-mechanical processes, as synergistic optimization of bulk materials and interfaces may effectively regulate these interactions and mitigate thermal instability.
(iii) Interfacial engineering has been extensively developed to suppress chemical and electrochemical side reactions at electrode-electrolyte interfaces, thereby improving the thermal stability of ASSBs. Beyond these advances, enhancing the mechanical compatibility of interfaces deserves greater attention. In this context, integrating negative thermal expansion (NTE) and positive thermal expansion (PTE) materials offers a promising strategy to construct near-zero thermal expansion interfaces in ASSBs[81,82]. Such designs can effectively mitigate thermomechanical stress mismatch, maintain interfacial integrity, and thereby enhance the thermal stability and safety of ASSBs.
(iv) Heat release rate and heat dissipation rate are critical parameters governing the thermal safety of ASSBs. However, corresponding quantitative data remain limited due to the complexity of solid-state systems and the lack of standardized measurement methods. Current studies suggest that ASSBs generally exhibit lower and more gradual heat release compared to conventional LELIBs[83,84]. Nevertheless, their overall thermal behavior is determined by the balance between heat generation and heat dissipation, which strongly depends on material chemistry, interface stability, and cell design. Therefore, systematic quantification of both heat release and dissipation characteristics, as well as their coupling with multiphysics processes, is essential for the rational design of safer ASSBs.
(v) Future advances will strongly benefit from the integration of advanced characterization, modeling, and monitoring strategies to achieve a comprehensive understanding and control of thermal behavior in ASSBs. Emerging in situ and operando techniques, such as advanced calorimetry, synchrotron-based spectroscopy, temperature-resolved microscopy, and spatially resolved thermal imaging, enable direct observation of thermal responses, phase transitions, gas evolution, and reaction pathways under heating and abuse conditions, providing unprecedented insight into the origin and propagation of thermal events[52,85,86]. In parallel, computational and data-driven approaches, including first-principles calculations, thermodynamic modeling, AI, and machine learning, offer powerful tools for predicting material stability, screening candidate systems, and identifying key descriptors governing thermal safety[87-89]. Complementarily, advanced monitoring strategies, including temperature, strain, and gas or byproduct detection, are essential for the early identification of thermal instability and failure processes. Compared with conventional LIBs, the development of dedicated risk monitoring and early warning systems for ASSBs remains both essential and urgent. The integration of these approaches with battery design and management systems will be critical for achieving safer and more reliable ASSBs.
In summary, the realization of intrinsically safe ASSBs demands a paradigm shift from passive protection to proactive thermal stability design. This includes the development of novel materials with minimal reactive species release and flame-retardant properties, interfaces with long-term chemical and mechanical robustness across wide temperature ranges, and cell architectures that inherently suppress heat accumulation and thermal propagation. Through the coordinated advancement of materials design, interface engineering, thermal diagnostics, and predictive modeling, ASSBs with both high energy density and robust thermal safety are expected to become a cornerstone technology for next-generation energy storage systems.
DECLARATIONS
Authors’ contributions
Manuscript preparation and visualization: Liang, S.; Heng, Y.
Supervision: Wang, C.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version GPT 5.3, released 2026-03-03) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22409103, 92572112, W2441017), the National Key Research and Development Program of China (2025YFF0523000), the Commanding Heights of Science and Technology of the Chinese Academy of Sciences (LDES15 0000), the Zhejiang Provincial Natural Science Foundation of China (LZ26E020006), and the “Innovation Yongjiang 2035” Key R&D Program (2025Z063, 2024Z040).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Schmaltz, T.; Hartmann, F.; Wicke, T.; Weymann, L.; Neef, C.; Janek, J. A roadmap for solid-state batteries. Adv. Energy. Mater. 2023, 13, 2301886.
3. Grey, C. P.; Hall, D. S. Prospects for lithium-ion batteries and beyond-a 2030 vision. Nat. Commun. 2020, 11, 6279.
4. Liang, S.; Sun, Y.; Cheng, Y. J.; et al. Probing all-solid-state batteries with real-time synchrotron and neutron techniques. Adv. Energy. Mater. 2025, 16, e04045.
5. Zhao, Q.; Stalin, S.; Zhao, C.; Archer, L. A. Designing solid-state electrolytes for safe, energy-dense batteries. Nat. Rev. Mater. 2020, 5, 229-52.
6. Huang, Y.; Cao, B.; Geng, Z.; Li, H. Advanced electrolytes for rechargeable lithium metal batteries with high safety and cycling stability. Acc. Mater. Res. 2024, 5, 184-93.
7. Luo, Y.; Rao, Z.; Yang, X.; Wang, C.; Sun, X.; Li, X. Safety concerns in solid-state lithium batteries: from materials to devices. Energy. Environ. Sci. 2024, 17, 7543-65.
8. Yao, Y.; Wei, Z.; Wang, H.; et al. Toward high energy density all solid-state sodium batteries with excellent flexibility. Adv. Energy. Mater. 2020, 10, 1903698.
9. Tan, D. H.; Meng, Y. S.; Jang, J. Scaling up high-energy-density sulfidic solid-state batteries: a lab-to-pilot perspective. Joule 2022, 6, 1755-69.
10. Kong, W. J.; Zhao, C. Z.; Sun, S.; et al. From liquid to solid-state batteries: Li-rich Mn-based layered oxides as emerging cathodes with high energy density. Adv. Mater. 2024, 36, e2310738.
11. Park, K. H.; Bai, Q.; Kim, D. H.; et al. Design strategies, practical considerations, and new solution processes of sulfide solid electrolytes for all-solid-state batteries. Adv. Energy. Mater. 2018, 8, 1800035.
12. Wang, C.; Adair, K.; Sun, X. All-solid-state lithium metal batteries with sulfide electrolytes: understanding interfacial ion and electron transport. Acc. Mater. Res. 2021, 3, 21-32.
13. Wang, C.; Liang, J.; Kim, J. T.; Sun, X. Prospects of halide-based all-solid-state batteries: from material design to practical application. Sci. Adv. 2022, 8, eadc9516.
14. Kim, K. J.; Balaish, M.; Wadaguchi, M.; Kong, L.; Rupp, J. L. M. Solid-state Li–metal batteries: challenges and horizons of oxide and sulfide solid electrolytes and their interfaces. Adv. Energy. Mater. 2020, 11, 2002689.
15. Kausthubharam; Vishnugopi, B. S.; Alujjage, A. S. J.; et al. Mechanistic understanding of thermal stability and safety in lithium metal batteries. Chem. Rev. 2026, 126, 404-47.
16. Kim, J. S.; Bong, S.; Kim, J. S.; et al. Thermal runaway in sulfide-based all-solid-state batteries: risk landscape, diagnostic gaps, and strategic directions. Adv. Energy. Mater. 2025, 15, e03593.
17. Chen, C.; Jiang, M.; Zhou, T.; et al. Interface aspects in all-solid-state Li-based batteries reviewed. Adv. Energy. Mater. 2021, 11, 2003939.
18. Ren, D.; Feng, X.; Liu, L.; et al. Investigating the relationship between internal short circuit and thermal runaway of lithium-ion batteries under thermal abuse condition. Energy. Stor. Mater. 2021, 34, 563-73.
19. Zhang, G.; Wei, X.; Chen, S.; Zhu, J.; Han, G.; Dai, H. Revealing the impact of slight electrical abuse on the thermal safety characteristics for lithium-ion batteries. ACS. Appl. Energy. Mater. 2021, 4, 12858-70.
20. Zhang, Y.; Li, Y.; Jia, Z.; et al. Investigating the safety of solid/liquid hybrid electrolyte lithium-ion battery: a comparative study with traditional LIBs under abuse condition. J. Power. Sources. 2024, 620, 235261.
21. Banerjee, A.; Wang, X.; Fang, C.; Wu, E. A.; Meng, Y. S. Interfaces and interphases in all-solid-state batteries with inorganic solid electrolytes. Chem. Rev. 2020, 120, 6878-933.
22. Rao, Z.; Lyu, P.; Li, M.; Liu, X.; Feng, X. A thermal perspective on battery safety. Nat. Rev. Clean. Technol. 2025, 1, 511-24.
23. Janek, J.; Zeier, W. G. Challenges in speeding up solid-state battery development. Nat. Energy. 2023, 8, 230-40.
24. Ye, Y.; Huang, W.; Xu, R.; et al. Cold-starting all-solid-state batteries from room temperature by thermally modulated current collector in sub-minute. Adv. Mater. 2022, 34, e2202848.
25. Chun, H.; Choi, H.; Jun, Y.; Lee, H. Comprehensive study on thermal characteristics of lithium-ion battery with entropic heat. Int. J. Energy. Res. 2024, 2024, 8815580.
26. Han, U.; Choi, H.; Lee, H.; Lee, H. Inverse heat transfer analysis method to determine the entropic coefficient of reversible heat in lithium-ion battery. Int. J. Energy. Res. 2023, 2023, 1-18.
27. Yu, H.; Huang, M.; Li, Y.; et al. Toward Joule heating recycling of spent lithium-ion batteries: a rising direct regeneration method. J. Energy. Chem. 2025, 105, 501-13.
28. Maher, K.; Boumaiza, A.; Amin, R. Understanding the heat generation mechanisms and the interplay between joule heat and entropy effects as a function of state of charge in lithium-ion batteries. J. Power. Sources. 2024, 623, 235504.
29. Song, G.; Lee, S.; Lee, M.; Park, J.; Lee, K. T. Electro-chemo-mechanical coupling in composite cathodes of sulfide-based all-solid-state batteries: pathways, degradation, and design rules. Adv. Sci. 2026, 13, e24187.
30. Li, X.; Liu, H.; Zhao, C.; et al. Hopping rate and migration entropy as the origin of superionic conduction within solid-state electrolytes. J. Am. Chem. Soc. 2023, 145, 11701-9.
31. Zhu, Y.; Li, W.; Zhang, L.; et al. Electrode/electrolyte interphases in high-temperature batteries: a review. Energy. Environ. Sci. 2023, 16, 2825-55.
32. Wang, S.; Wu, Y.; Ma, T.; Chen, L.; Li, H.; Wu, F. Thermal stability between sulfide solid electrolytes and oxide cathode. ACS. Nano. 2022, 16, 16158-76.
33. Hubaud, A. A.; Schroeder, D. J.; Ingram, B. J.; Okasinski, J. S.; Vaughey, J. T. Thermal expansion in the garnet-type solid electrolyte (Li7-xAlx/3)La3Zr2O12 as a function of Al content. J. Alloys. Compd. 2015, 644, 804-7.
34. Zhang, W.; Schröder, D.; Arlt, T.; et al. (Electro)chemical expansion during cycling: monitoring the pressure changes in operating solid-state lithium batteries. J. Mater. Chem. A. 2017, 5, 9929-36.
35. Koerver, R.; Zhang, W.; De Biasi, L.; et al. Chemo-mechanical expansion of lithium electrode materials - on the route to mechanically optimized all-solid-state batteries. Energy. Environ. Sci. 2018, 11, 2142-58.
36. Miara, L.; Windmüller, A.; Tsai, C. L.; et al. About the compatibility between high voltage spinel cathode materials and solid oxide electrolytes as a function of temperature. ACS. Appl. Mater. Interfaces. 2016, 8, 26842-50.
37. Wang, S.; Wu, Y.; Li, H.; Chen, L.; Wu, F. Improving thermal stability of sulfide solid electrolytes: an intrinsic theoretical paradigm. InfoMat 2022, 4, e12316.
38. Wu, Y.; Wang, S.; Li, H.; Chen, L.; Wu, F. Progress in thermal stability of all-solid-state-Li-ion-batteries. InfoMat 2021, 3, 827-53.
39. Tsukasaki, H.; Otoyama, M.; Mori, Y.; et al. Analysis of structural and thermal stability in the positive electrode for sulfide-based all-solid-state lithium batteries. J. Power. Sources. 2017, 367, 42-8.
40. Tsukasaki, H.; Mori, Y.; Otoyama, M.; et al. Crystallization behavior of the Li2S-P2S5 glass electrolyte in the LiNi1/3Mn1/3Co1/3O2 positive electrode layer. Sci. Rep. 2018, 8, 6214.
41. Atarashi, A.; Tsukasaki, H.; Otoyama, M.; et al. Ex situ investigation of exothermal behavior and structural changes of the Li3PS4-LiNi1/3Mn1/3Co1/3O2 electrode composites. Solid. State. Ionics. 2019, 342, 115046.
42. Tsukasaki, H.; Uchiyama, T.; Yamamoto, K.; et al. Exothermal mechanisms in the charged LiNi1/3Mn1/3Co1/3O2 electrode layers for sulfide-based all-solid-state lithium batteries. J. Power. Sources. 2019, 434, 226714.
43. Tsukasaki, H.; Otoyama, M.; Kimura, T.; et al. Exothermal behavior and microstructure of a LiNi1/3Mn1/3Co1/3O2 electrode layer using a Li4SnS4 solid electrolyte. J. Power. Sources. 2020, 479, 228827.
44. Kim, T.; Kim, K.; Lee, S.; Song, G.; Jung, M. S.; Lee, K. T. Thermal runaway behavior of Li6PS5Cl solid electrolytes for LiNi0.8Co0.1Mn0.1O2 and LiFePO4 in all-solid-state batteries. Chem. Mater. 2022, 34, 9159-71.
45. Chen, R.; Nolan, A. M.; Lu, J.; et al. The thermal stability of lithium solid electrolytes with metallic lithium. Joule 2020, 4, 812-21.
46. Vishnugopi, B. S.; Hasan, M. T.; Zhou, H.; Mukherjee, P. P. Interphases and electrode crosstalk dictate the thermal stability of solid-state batteries. ACS. Energy. Lett. 2022, 8, 398-407.
47. Wolfenstine, J.; Allen, J. L.; Read, J.; Sakamoto, J. Chemical stability of cubic Li7La3Zr2O12 with molten lithium at elevated temperature. J. Mater. Sci. 2013, 48, 5846-51.
48. Han, Y.; Liu, B.; Xiao, Z.; et al. Interface issues of lithium metal anode for high-energy batteries: challenges, strategies, and perspectives. InfoMat 2021, 3, 155-74.
49. Heubner, C.; Maletti, S.; Auer, H.; et al. From lithium-metal toward anode-free solid-state batteries: current developments, issues, and challenges. Adv. Funct. Mater. 2021, 31, 2106608.
50. Longchamps, R. S.; Yang, X. G.; Wang, C. Y. Fundamental insights into battery thermal management and safety. ACS. Energy. Lett. 2022, 7, 1103-11.
51. Yin, Z.; Zhu, J.; Yan, A.; et al. Battery management system towards solid-state batteries. Chain 2024, 1, 319-53.
52. Wang, J.; Yang, K.; Sun, S.; et al. Advances in thermal-related analysis techniques for solid-state lithium batteries. InfoMat 2023, 5, e12401.
53. Yang, C.; Singh, A.; Pu, X.; et al. Addressing the safety of next-generation batteries. Nature 2025, 645, 603-13.
54. Bates, A. M.; Preger, Y.; Torres-Castro, L.; Harrison, K. L.; Harris, S. J.; Hewson, J. Are solid-state batteries safer than lithium-ion batteries? Joule 2022, 6, 742-55.
55. Yang, S. J.; Hu, J. K.; Jiang, F. N.; Yuan, H.; Park, H. S.; Huang, J. Q. Safer solid-state lithium metal batteries: mechanisms and strategies. InfoMat 2023, 6, e12512.
56. Feng, X.; Ren, D.; He, X.; Ouyang, M. Mitigating thermal runaway of lithium-ion batteries. Joule 2020, 4, 743-70.
57. Mao, B.; Chen, H.; Cui, Z.; Wu, T.; Wang, Q. Failure mechanism of the lithium ion battery during nail penetration. Int. J. Heat. Mass. Transfer. 2018, 122, 1103-15.
58. Xu, J.; Mei, W.; Zhao, C.; Liu, Y.; Zhang, L.; Wang, Q. Study on thermal runaway mechanism of 1000 mAh lithium ion pouch cell during nail penetration. J. Therm. Anal. Calorim. 2020, 144, 273-84.
59. He, Y.; Wang, J.; Rong, C.; et al. Status of cell-level thermal safety assessments toward optimization of all-solid-state batteries. Cell. Rep. Phys. Sci. 2024, 5, 102056.
60. Rui, X.; Ren, D.; Liu, X.; et al. Distinct thermal runaway mechanisms of sulfide-based all-solid-state batteries. Energy. Environ. Sci. 2023, 16, 3552-63.
61. Yang, S.; Hu, J.; Jiang, F.; et al. Oxygen-induced thermal runaway mechanisms of Ah-level solid-state lithium metal pouch cells. eTransportation 2023, 18, 100279.
62. Kim, T.; Chang, H.; Song, G.; et al. Critical factors contributing to the thermal runaway of thiophosphate solid electrolytes for all-solid-state batteries. Adv. Funct. Mater. 2024, 34, 2404806.
63. Bertrand, M.; Johnson, N. B.; Jin, L.; et al. Unveiling the thermite-driven lithium fire ignition in solid-state batteries. Joule 2025, 9, 101953.
64. Wu, Y.; Zhang, W.; Rui, X.; et al. Thermal runaway mechanism of composite cathodes for all-solid-state batteries. Adv. Energy. Mater. 2025, 15, 2405183.
65. Kaboli, S.; Girard, G.; Zhu, W.; et al. Thermal evolution of NASICON type solid-state electrolytes with lithium at high temperature via in situ scanning electron microscopy. Chem. Commun. 2021, 57, 11076-9.
66. Li, X.; Liang, J.; Luo, J.; et al. Air-stable Li3InCl6 electrolyte with high voltage compatibility for all-solid-state batteries. Energy. Environ. Sci. 2019, 12, 2665-71.
67. Li, X.; Liang, J.; Yang, X.; et al. Progress and perspectives on halide lithium conductors for all-solid-state lithium batteries. Energy. Environ. Sci. 2020, 13, 1429-61.
68. Wang, C.; Liang, J.; Luo, J.; et al. A universal wet-chemistry synthesis of solid-state halide electrolytes for all-solid-state lithium-metal batteries. Sci. Adv. 2021, 7, eabh1896.
69. Ganesan, P.; Soans, M.; Cambaz, M. A.; et al. Fluorine-substituted halide solid electrolytes with enhanced stability toward the lithium metal. ACS. Appl. Mater. Interfaces. 2023, 15, 38391-402.
70. Lannelongue, P.; Lindberg, S.; Gonzalo, E.; et al. Stable cycling of halide solid state electrolyte enabled by a dynamic layered solid electrolyte interphase between Li metal and Li3YCl4Br2. Energy. Stor. Mater. 2024, 72, 103733.
71. Wang, Q.; Zhou, Y.; Wang, X.; et al. Designing lithium halide solid electrolytes. Nat. Commun. 2024, 15, 1050.
72. Cheng, Z.; Zhao, W.; Wang, Q.; et al. Beneficial redox activity of halide solid electrolytes empowering high-performance anodes in all-solid-state batteries. Nat. Mater. 2025, 24, 1763-72.
73. Lu, J.; Zhou, J.; Chen, R.; et al. 4.2V poly(ethylene oxide)-based all-solid-state lithium batteries with superior cycle and safety performance. Energy. Stor. Mater. 2020, 32, 191-8.
74. Chen, R.; Yao, C.; Yang, Q.; et al. Enhancing the thermal stability of NASICON solid electrolyte pellets against metallic lithium by defect modification. ACS. Appl. Mater. Interfaces. 2021, 13, 18743-9.
75. Lee, M. J.; Han, J.; Lee, K.; et al. Elastomeric electrolytes for high-energy solid-state lithium batteries. Nature 2022, 601, 217-22.
76. Lin, Z.; Yao, Q.; Yang, S.; et al. Highly safe all-solid-state lithium metal battery enabled by interface thermal runaway regulation between lithium metal and solid-state electrolyte. Adv. Funct. Mater. 2025, 35, 2424110.
77. Zhao Y, ; Zhang L, ; Liu J, ; et al. Atomic/molecular layer deposition for energy storage and conversion. Chem. Soc. Rev. 2021, 50, 3889-956.
78. Sun, Y.; Ma, J.; Wu, D.; et al. A breathable inorganic–organic interface for fabricating a crack-free nickel-rich cathode with long-term stability. Energy. Environ. Sci. 2024, 17, 5124-36.
79. Wang, M.; Wu, Y.; Cao, Y.; Li, G.; Miao, X.; Li, X. Real-time artificial intelligence for solid-state lithium metal batteries. Nat. Commun. 2025, 16, 11160.
80. Dutra, A. C. C.; Goldmann, B. A.; Islam, M. S.; Dawson, J. A. Understanding solid-state battery electrolytes using atomistic modelling and machine learning. Nat. Rev. Mater. 2025, 10, 566-83.
81. Qiu, B.; Zhou, Y.; Liang, H.; et al. Negative thermal expansion and oxygen-redox electrochemistry. Nature 2025, 640, 941-6.
82. Li, Q.; Yang, L.; Liang, G.; et al. Negative thermal expansion behavior enabling good electrochemical-energy-storage performance at low temperatures. Angew. Chem. Int. Ed. Engl. 2025, 64, e202419300.
83. Charbonnel, J.; Darmet, N.; Deilhes, C.; et al. Safety evaluation of all-solid-state batteries: an innovative methodology using in situ synchrotron X-ray radiography. ACS. Appl. Energy. Mater. 2022, 5, 10862-71.
84. Liu, T.; Kum, L. W.; Singh, D. K.; Kumar, J. Thermal, electrical, and environmental safeties of sulfide electrolyte-based all-solid-state Li-ion batteries. ACS. Omega. 2023, 8, 12411-7.
85. Wei, Y.; Wang, M.; Zhang, M.; Cai, T.; Huang, Y.; Xu, M. Advancements, challenges, and future trajectories in advanced battery safety detection. Electrochem. Energy. Rev. 2025, 8, 10.
86. Kong, D.; Lv, H.; Ping, P.; Wang, G. A review of early warning methods of thermal runaway of lithium ion batteries. J. Energy. Storage. 2023, 64, 107073.
87. Choi, H. J.; Kim, S. A.; Kim, C. H.; Shin, B. S. Machine learning - based on analysis of EV battery thermal runaway simulation. J. Mech. Sci. Technol. 2025, 39, 3667-77.
88. Wang, Y.; Feng, X.; Guo, D.; et al. Temperature excavation to boost machine learning battery thermochemical predictions. Joule 2024, 8, 2639-51.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].