fig4
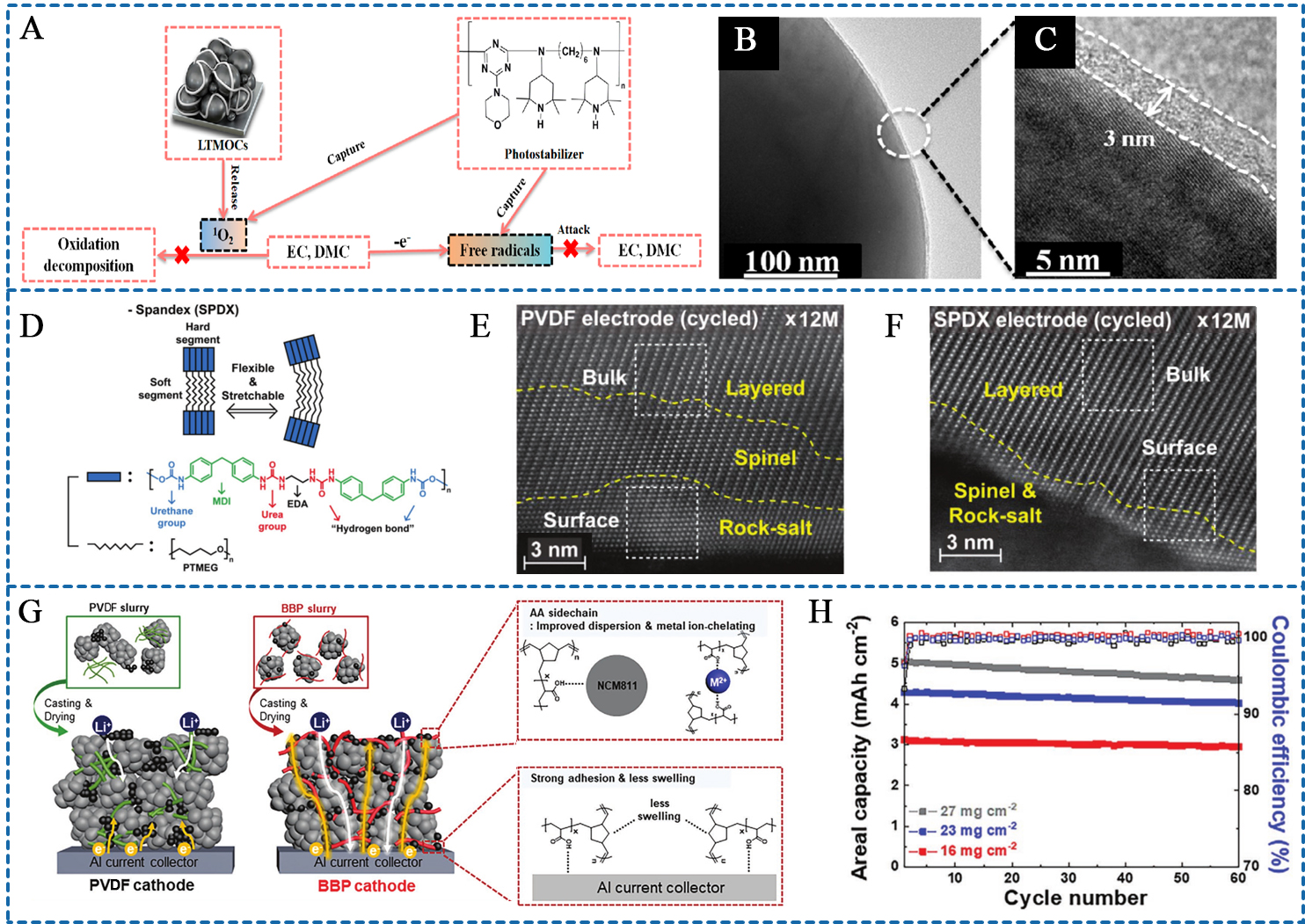
Figure 4. (A) Schematic illustration of the singlet oxygen and free radicals scavenging ability of PS binder; (B and C) TEM images of PS binders-based cathode material surface after 400 cycles; (A-C) are reprinted with permission from Ref.[65], Copyright © 2021, American Chemical Society; (D) Binding scheme of SPDX binder with NCM particle via hydrogen bonding interaction; (E and F) HAADF-STEM images of the PVDF and SPDX-based cathodes after 100 cycles; (D-F) are reprinted with permission from Ref.[57], Copyright © 2020 Wiley; (G) Schematic representation of the BBP and PVDF cathodes; (H) Cycling performance of BBP cathodes as a function of areal mass loading. (G and H) are reprinted with permission from Ref.[92], Copyright © 2021 Wiley. LTMOC: Layered oxides-based cathodes; EC: ethylene carbonate; EMC: ethyl methyl carbonate; SPDX: spandex; PTMEG: poly(tetramethylene ether glycol); MDI: methylene diphenyl diisocyanate; EDA: ethylenediamine; PVDF: poly(vinylidene fluoride); BBP: bottlebrush polymer; NCM: LiNixCoyMn1-x-yO2, (x ≥ 0.8); PS: photostabilizer; TEM: transmission electron microscopy; HAADF-STEM: high-angle annular dark-field scanning transmission electron microscopy.