CuO foam with in-situ grown polyaniline for high sensitivity room temperature NO2 gas sensors
Abstract
The rapid and accurate detection of NO2 gas at ppm level is of great value in human health and environmental protection. However, the most widely used metal-oxide semiconductor (MOS)-based NO2 sensors usually require high operating temperatures (> 200 °C) to perform sensing functions. The preparation of these sensors possessing excellent sensing performance at room temperature (RT) remains a challenge. In this paper, copper oxide (CuO)-polyaniline (PANI) composites were prepared by heat treatment of copper foam and in situ growth of PANI. These composites exhibited a response value of 7.63 to
Keywords
INTRODUCTION
Nitrogen dioxide (NO2) is a serious air pollutant harmful to human health and the environment[1]. It is mainly generated from fossil fuel combustion, industrial production, automobile exhaust and agricultural processes[2-4]. Its environmental effects are wide-ranging, including impacts on wetlands, changes in competition and composition among terrestrial plant species[5], reduced atmospheric visibility, acidification of surface waters, eutrophication (a lack of oxygen due to algal blooms in nutrient-rich water, such as nitrogen and phosphorus)[6], and an increase in levels of toxins in the water column that are harmful to fish and other aquatic life[7]. Moreover, NO2 can inflict harm on the human respiratory system and induce respiratory illnesses, such as asthma[8]. Therefore, it is crucial to create stable, high-selectivity, and high-sensitivity gas sensors to more effectively detect and regulate the concentration of the toxic NO2[9].
Resistive metal-oxide semiconductor (MOS) gas sensors are the most commonly used because of their high sensitivity, fast response speed, and low cost for efficient target gas detection[10]. Based on previous research, n-type MOSs, including Ag2Te[5], SnO2[3], ZnO[11], and Ga2O3[4], have been used for NO2 sensors. Due to its low humidity dependence and strong thermal stability, copper oxide (CuO), a typical p-type MOS material with a bandgap of 1.2-1.9 eV, has emerged as a research hotspot in gas sensors. However, CuO-based gas sensors must function at high temperatures (200-500 °C). High operating temperatures will increase energy consumption and cause CuO nanomaterials to sinter and aggregate, altering their gas-sensitive characteristics. This seriously hinders the practical application of CuO-based sensors. Therefore, it is crucial to develop CuO-based gas sensors with high sensitivity and good stability at room temperature (RT)[12].
In order to improve the sensing ability of CuO at lower temperatures, considerable efforts have been made to combine CuO with various nanomaterials[13]. Hmeed et al. synthesized In2O3-CuO composite thin films using the chemical spray pyrolysis technique[14]. The sensitivity reached 89.77% toward 193 ppm NO2 at 200 °C with an equal response time of 24 s. Wang et al. prepared a 3D structure consisting of Cu2O-CuO nanorods with the hydrothermal method[15]. The materials exhibited high sensitivity to NO2 at 187 °C, and the response/recovery times were 35/47 s, respectively. Ambedkar et al. reported the Pd-CuO composite nanoparticle sensor to enhance NO2 gas sensing performance[16]. The results yielded a Pd-CuO response of 817% toward 100 ppm NO2 at 125 °C. Hsueh et al. reported that a CuO nanowire (NW)/micro-electro-mechanical system (MEMS) nanocomposite-based NO2 gas sensor demonstrated a response of 50.1% toward 500 ppb NO2 at 119 °C[17]. Govind et al. prepared the CuO/ZnO composite with a NO2 detection limit of 155 ppb at 30 °C, and the response/recovery times for 5 ppm NO2 were 18 and 32 s, respectively[18]. However, these sensors are still unable to exhibit good sensing performance at RT. Extensive unresolved characteristics remain to be explored in the development of CuO-based gas sensors at RT[19].
Conductive polymers are promising candidates to composite with CuO[20]. Polythiophene (PTh), polypyrrole (PPy), polyaniline (PANI), poly(3,4-ethylenedioxythiophene) (PEDOT), polyacetylene (PA) and their derivatives are the most studied conducting polymers[21]. PANI has gained a lot of attention due to its good RT gas-sensitive properties[22], unusual electrical properties, and ease of production[23,24]. In 1993, Wu and Hirata synthesized PANI powders and prepared the first PANI gas sensor. The response was 5% at 20 °C for 10 ppm NH3, which laid a solid foundation for the study of PANI gas sensors[10]. However, PANI has low sensitivity and a slow recovery rate in the air[25]. Various PANI-based composites for NO2 sensors have been investigated[26]. Nguyet et al. prepared the PANI/ZnO nanohybrid nanoplates using the hydrothermal method for the low-temperature NO2 sensor[27]. The response and recovery times were 31 and 401 s, respectively. Bora Patowary et al. reported the application of Ta2O5/PANI composite for NO2 gas detection[28]. It was found that the response time was reduced to 55 s and the recovery time was 160 s (S = 83% to 500 ppm NO2).
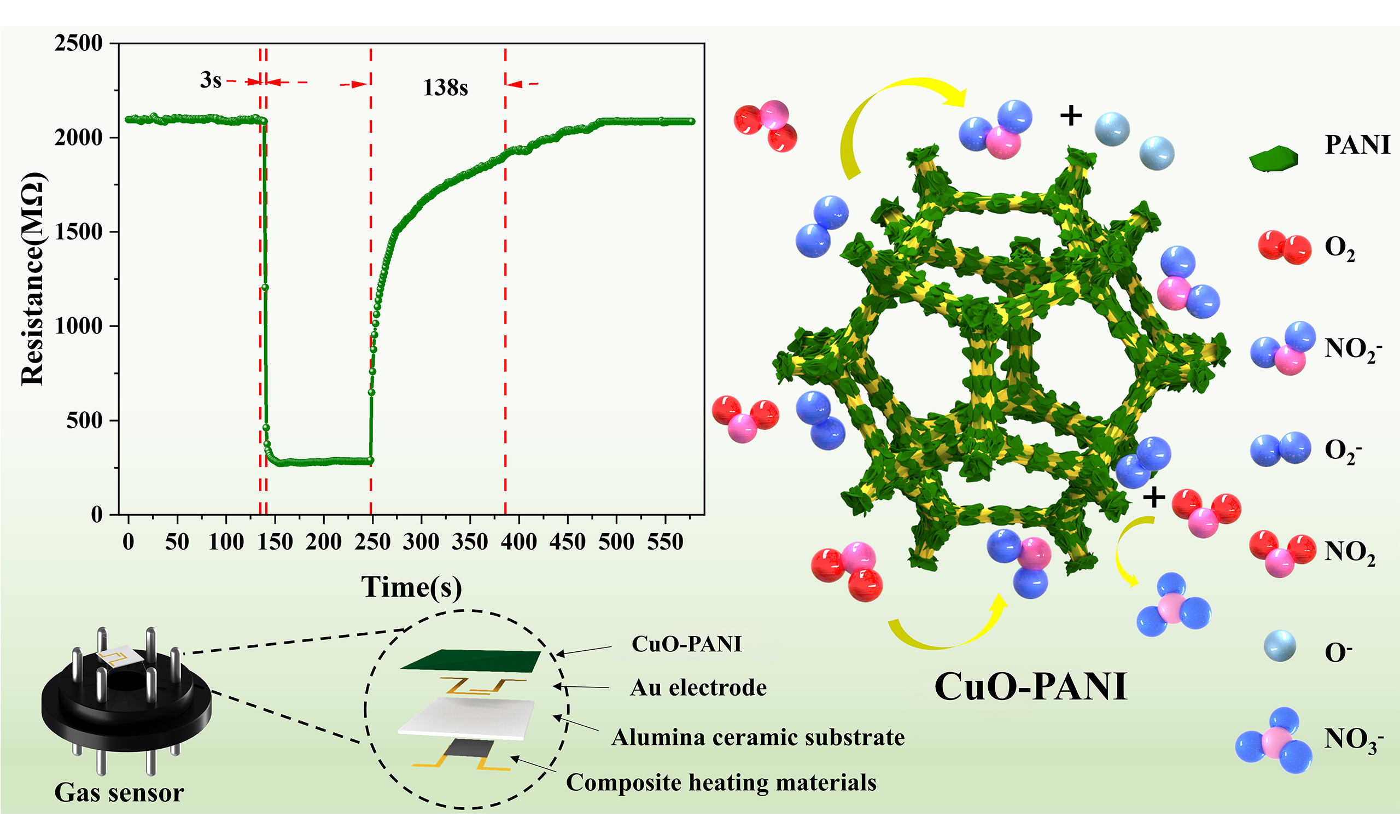
In this study, NO2 sensors with high sensitivity at RT based on the CuO-PANI composite were prepared by in-situ growth of PANI on CuO foam. Due to good matching and extensive heterogeneous interfaces[29], the composites combine the advantages of both MOSs and PANI[30]. The sensing properties of CuO-PANI were adjusted by varying the mass ratio of aniline to CuO. The test results revealed that when the mass ratio of CuO to aniline was 1:1, the composite material exhibited a response value of 7.63 to 100 ppm NO2 at RT, which was 4.8 times higher than that of pure PANI and CuO. Moreover, the RT response time (3 s) was dramatically shortened compared to that of PANI (27 s). In addition, the prepared sensors exhibited excellent stability and high NO2 selectivity. The mechanism underlying the significant change in the gas-sensing characteristics due to the PANI-coated CuO foam was investigated. The CuO-PANI sensor exhibits a higher NO2 sensing response and faster response/recovery time compared with pristine PANI and CuO sensors, which provides a new solution for the preparation of high-performance RT NO2 sensors for air-quality testing applications.
EXPERIMENTAL
In this paper, all the reagents used are of analytical grade and are used directly without further purification.
Synthesis of pristine PANI and CuO foam
The synthesis of pristine PANI was performed according to the literature[31]. Typical, 0.57 g of ammonium persulfate (APS) and 230 μL of aniline monomer (Sinopharm Chemical Reagent Co., Ltd.) were dissolved in two vials containing 20 mL HCl (1 mol/L), respectively. APS solution was pre-cooled at 4 °C for 30 min, and was added dropwise to the aniline solution. The reaction temperature is maintained at approximately 4 °C during the polymerization process, as the aniline polymerization is a vigorous exothermic reaction. After
The purchased copper foam (100 × 100 × 0.5 mm) underwent a pretreatment with HCl (1 mol/L). Then, it was washed in acetone, anhydrous ethanol, and deionized water and dried at 60 °C for 30 min to remove the organic molecules and oxide layer. The pretreated copper foam was then sintered in a muffle furnace at
Preparation of CuO-PANI sensors and response tests
The CuO-PANI composites were prepared using in situ polymerization. Typically, 0.608 g, 0.76 g, and
The synthesized materials were mixed with deionized water to form slurries. The slurries were uniformly coated on the interdigitated gold electrodes. Then, the sensors were further dried at 60 °C for 1 h. The gas sensor tester system (WS-30A, Zhengzhou Winsen Electronics Technology Co., Ltd., China) was used to test the sensor response. The platinum wires were welded to the sensor’s pedestal before the gas-detecting test.
NO2 standard gas (99.99%) was purchased from Henan Yuanzheng Science and Technology Development Co., Ltd., China. In the process of sensing measurements, the sensors were exposed to 100 ppm NO2
Material characterization
The surface morphology of materials was observed using focused ion beam scanning electron microscopy (SEM, Zeiss/Merlin Compact, Germany), and the surface element distribution was measured with an energy-dispersive X-ray spectrometer (EDS) attachment on the SEM instrument. The material’s crystal structure was examined using Cu-Kα radiation with a wavelength of 0.154056 nm on a standard X-ray diffractometer (XRD, D8-Advance, Germany). Transmission electron microscopy (TEM, FEI TalosF200X, American) was employed to assess the morphology and lattice of the samples. The thermogravimetric analyzer (TG, DSC Mattler Switzerland) was used to determine the thermal stability of the samples from 30 to 800 °C. The test was conducted in the presence of air and at a heating rate of 5 °C·min-1. The elements and their valences on the material surface were analyzed using X-ray photoelectron spectroscopy with Al Kα radiation (XPS, Thermo Fischer ESCALAB 250Xi, American).
RESULTS AND DISCUSSION
Characterization of sensing materials
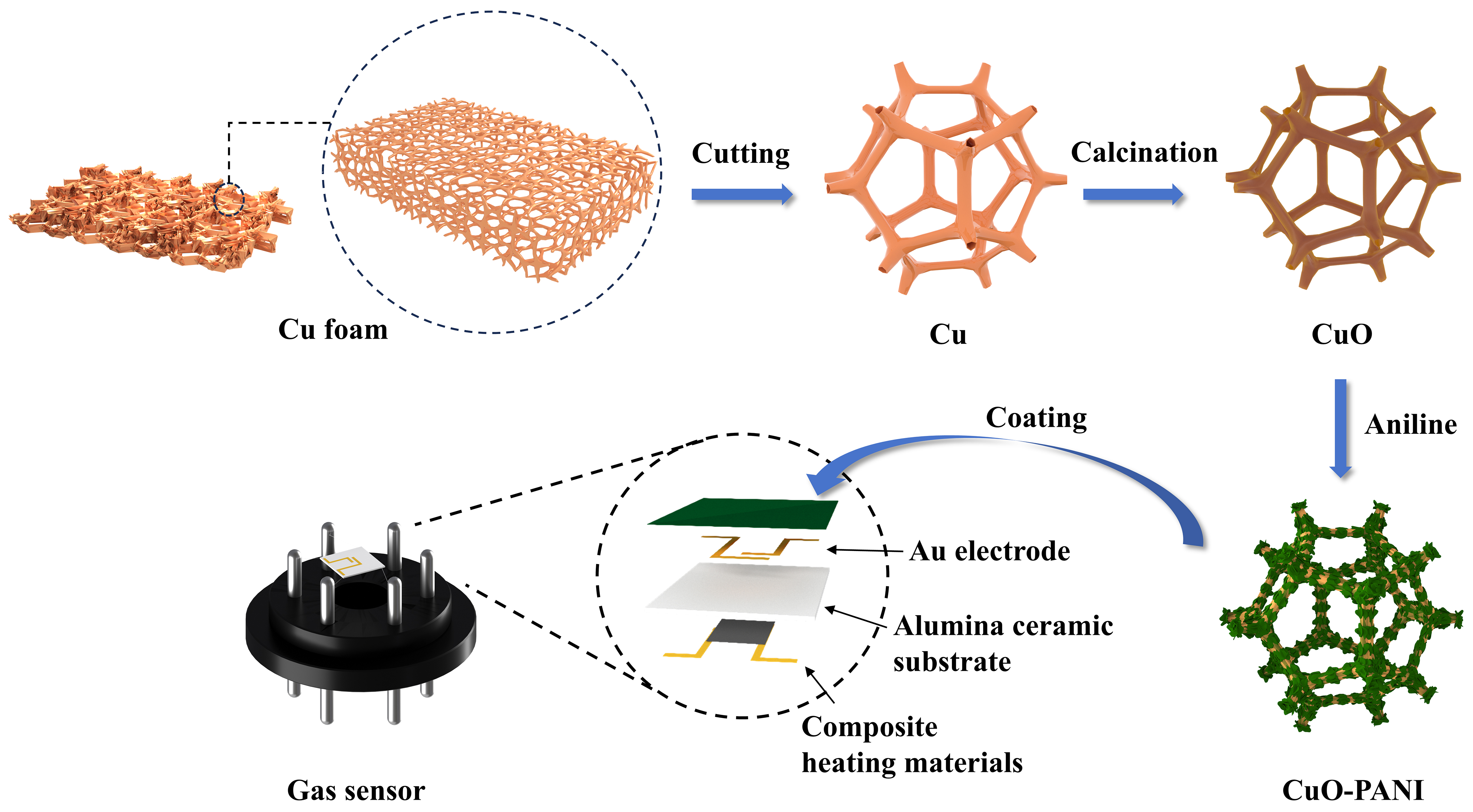
The preparation process of the CuO-PANI complex is shown in Figure 1. Firstly, copper foam with a macroporous structure was calcined in air to obtain CuO foam. Then, PANI was grown in situ on the surface of CuO foam by aniline oxidative polymerization. The in-situ composite of PANI allows it to be uniformly coated on the surface of the CuO foam, thus providing a more stable heterogeneous interface.
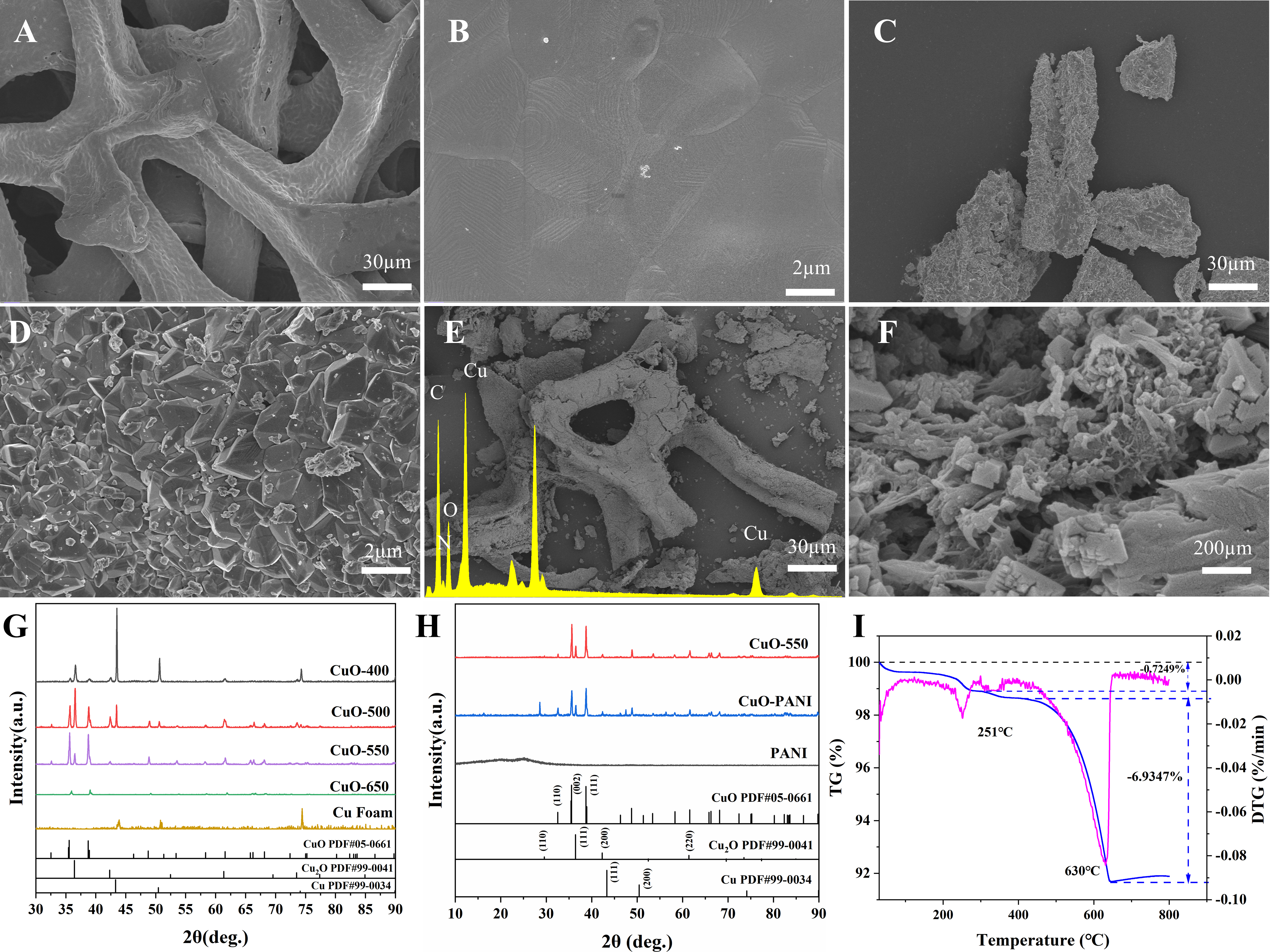
Supplementary Figure 1 shows that CuO-PANI was uniformly coated on the planar electrode. As shown in Figure 2A, the Cu foam is shown to have a three-dimensional (3D) porous skeleton. This unique structure allows for rapid gas circulation and can provide ample space for the growth of PANI. The surface of the Cu foam is smooth and flat, as depicted in Figure 2B. Figure 2C displays the surface morphology of CuO foam after calcination at 550 °C. The 3D structure of CuO foam is disrupted by grinding, but its fundamental prismatic skeleton remains, which is favorable for the coating of PANI. The surface of the material becomes roughened due to the growth of the oxidized layer of CuO, as evidenced in Figure 2D. CuO foam demonstrates an irregular cubic morphology, and this nanoscale nanoporous structure with a large specific surface area is conducive to the coating of PANI and favorable for gas diffusion. Figure 2E illustrates the surface morphology of the CuO-PANI-2, which maintains the basic 3D skeleton structure after grinding. Figure 2F shows that the fibrous PANI can be seen to be encapsulated on the surface of CuO foam, and the densely folded morphology can significantly increase the specific surface area, providing substantial active sites for the reaction of target gases. The EDS pattern of the CuO-PANI-2 also confirms the successful coating of PANI.
Figure 2. SEM images of (A) and (B) Cu foam, (C) and (D) CuO foam, (E) and (F) CuO-PANI-2; (G) and (H) XRD patterns of the as-synthesized samples; (I) TG analysis curves of CuO-PANI-2. SEM: Scanning electron microscopy; PANI: polyaniline; XRD: X-ray diffraction; TG: thermogravimetric.
X-ray diffraction (XRD) was used to examine the crystal structures of CuO, PANI, and CuO-PANI-2. Copper foams were calcined at various temperatures to obtain CuO foams. Figure 2G reveals distinct crystalline phases in the various CuO samples. The diffraction peaks at 35.553° and 38.730° are assigned to the (-111) and (111) planes of CuO (PDF#05-0661), respectively. The peaks at 36.423°, 42.307°, and 61.374° are caused by the (111), (200), and (220) crystal planes of Cu2O (PDF#99-0041), respectively. The (111) and (200) crystal planes of Cu (PDF#99-0034) are responsible for the diffraction peaks at 43.316° and 50.448°, respectively. The characteristic peak intensity of the Cu phase decreases as the calcination temperature increases. While below 500 °C, the peak intensity of the Cu2O phase continues to increase with temperature, and the peak of Cu remains intense. Above 500 °C, the peak of CuO gradually exceeds that of Cu2O, and the peak of Cu disappears. Only the peak of CuO exists at 650 °C, implying that the copper foam has been completely oxidized to CuO. Due to the good electrical conductivity of Cu, CuO foams containing Cu composite with PANI may result in a short circuit. When the calcination temperature is too high, the copper foam is completely oxidized to CuO, and the great resistance at RT is detrimental to the gas-sensing properties of the composite. Therefore, we select a calcination temperature of 550 °C for the CuO and PANI composite. At this point, the calcined product consists mainly of CuO and a small amount of Cu2O. The presence of Cu2O can lead to lattice defects and heterojunctions in CuO, which can promote gas-sensitive properties. There is no notable distinctive peak for PANI, as shown in Figure 2H. The peaks at the (110) crystal plane of Cu2O were significantly enhanced after PANI coating, possibly due to the partial reduction of CuO to Cu2O during the oxidative polymerization of aniline.
The TG curves of copper foam, CuO foam, and CuO-PANI-2 are illustrated in Figure 2I and Supplementary Figure 2. The mass of copper and CuO foams decreases as the temperature increases from 30 to 90 °C. This is mainly due to the volatilization of physically adsorbed water and organic matter on the surface of the material[32]. It can be seen that as the temperature increases from 100 to 800 °C, the mass of the copper foam and CuO foam gradually grows due to oxidation by oxygen in the air. In contrast, CuO-PANI-2 exhibits clear weight loss intervals at 230-250 °C and 500-620 °C, with weight losses of 0.72% and 6.93%, respectively. Due to the volatilization of adsorbed water in the composite and the complete degradation of the PANI, there are two distinct exothermic maxima at 251 and 630 °C, respectively[33]. The results further show that PANI is successfully composited with CuO foam.
The structure and morphology of CuO foam and CuO-PANI-2 were further analyzed using TEM. It can be seen that the bulk CuO has an inhomogeneous thickness [Figure 3A], corresponding to the rough surface in the SEM image. The distinct stripes with lattice spacings of 0.19592 and 0.2687 nm in CuO foam [Figure 3B] correspond to the (-112) and (110) planes of CuO, respectively. After PANI coating, amorphous regions appear at the edges of the CuO [Figure 3C], which is consistent with the XRD results. The streak with a lattice spacing of 0.2368 nm in CuO-PANI-2 [Figure 3D] corresponds to the (111) plane of the CuO phase. The energy-dispersive X-ray spectroscopy (EDX) spectra [Figure 3E] show the presence of Cu, N, and O elements in CuO-PANI-2. The homogeneous distribution of N elements further confirms the uniform coating of PANI.
Figure 3. TEM and HRTEM images of (A) and (B) CuO-550, (C) and (D) CuO-PANI-2; (E) TEM-EDS element mappings images of CuO-PANI-2. TEM: Transmission electron microscopy; HRTEM: high resolution transmission electron microscopy; PANI: polyaniline; EDS: energy-dispersive X-ray spectrometer.
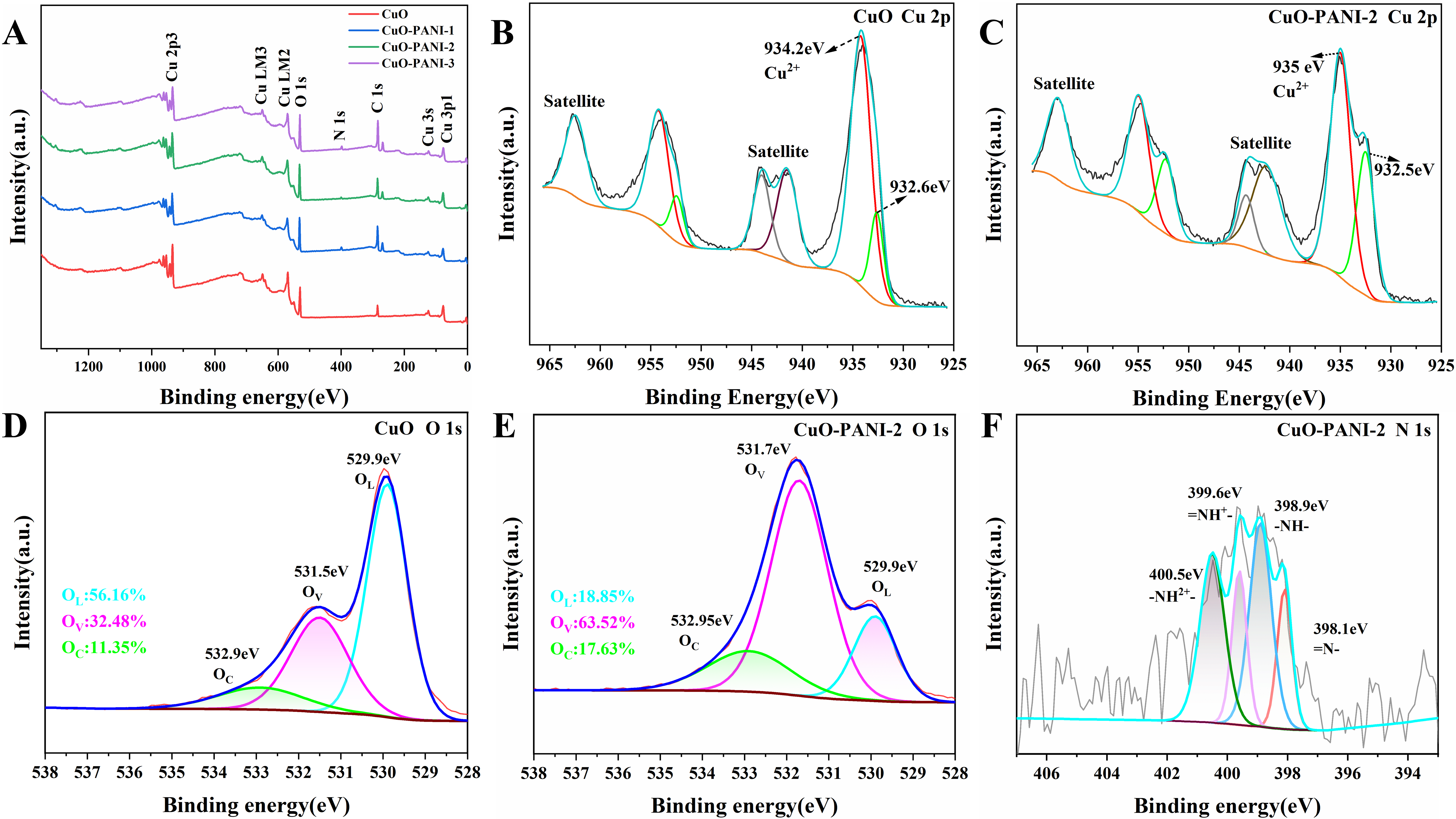
X-ray photoelectron spectrometer (XPS) measurement was used to analyze the surface elemental compositions and electronic valence states of the samples. The XPS survey spectra [Figure 4A] show the presence of C, O, and Cu elements in CuO, whereas a weak N 1s peak can also be observed in the CuO-PANI samples in addition to these elements. It confirms that the PANI is coated on the surface of the CuO foam. Furthermore, high-resolution elemental spectra of Cu, O, and N were examined [Figure 4B-D]. CuO foam mainly exhibits characteristic peaks of 2p3/2 and 2p1/2 located at binding energies of 932.6 and
Figure 4. (A) XPS spectra of the as-synthesized samples; High-resolution energy spectra of Cu 2p of (B) CuO and (C) CuO-PANI-2; O 1s spectrum of (D) CuO and (E) CuO-PANI-2; (F) N 1s spectrum of CuO-PANI-2. XPS: X-ray photoelectron spectrometer; PANI: polyaniline.
The sensing performance of the sensor is significantly influenced by the oxygen species of the gas-sensitive material[34]. The O 1s spectra of CuO foam and CuO-PANI-2 can be deconvoluted into three major peaks, nearly at 532.9, 531.5, and 529.9 eV [Figure 4D and E], assigned to the surface adsorbed oxygen (OC), oxygen vacancies (OV) and lattice oxygen (OL), respectively[35]. The content of OV in CuO-PANI-2 increases by nearly twofold, from 32.48% to 63.52%, as compared with CuO foam. The significant increase may be related to the reduction of Cu2+ to Cu+. Ov can modulate the electronic structure of semiconductor gas materials to facilitate charge transfer while altering the surface atomic coordination environments, hence acting as preferential adsorption sites and further improving gas sensor performance[36]. Moreover, the OC content in CuO-PANI-2 also increases from 11.35% to 17.63%, which can also promote the gas sensing performance.
The N 1s spectrum of CuO-PANI-2 can be deconvoluted into four characteristic peaks, which are attributed to =NH-, -NH2+-, =NH+-, and -NH2+-, respectively [Figure 4F]. These four characteristic peaks are located at 398.1, 398.9, 399.6, and 400.5 eV, respectively. Protonation is defined as the proportion of protonated nitrogen atoms (the sum of characteristic peaks area ratios corresponding to =NH+- and -NH2+-)[37]. The proportions of the area under the peaks of =NH+- and -NH2+- are 17.54% and 32.03%, which indicates that its protonation degree is 49.57%.
Gas-sensing characteristics
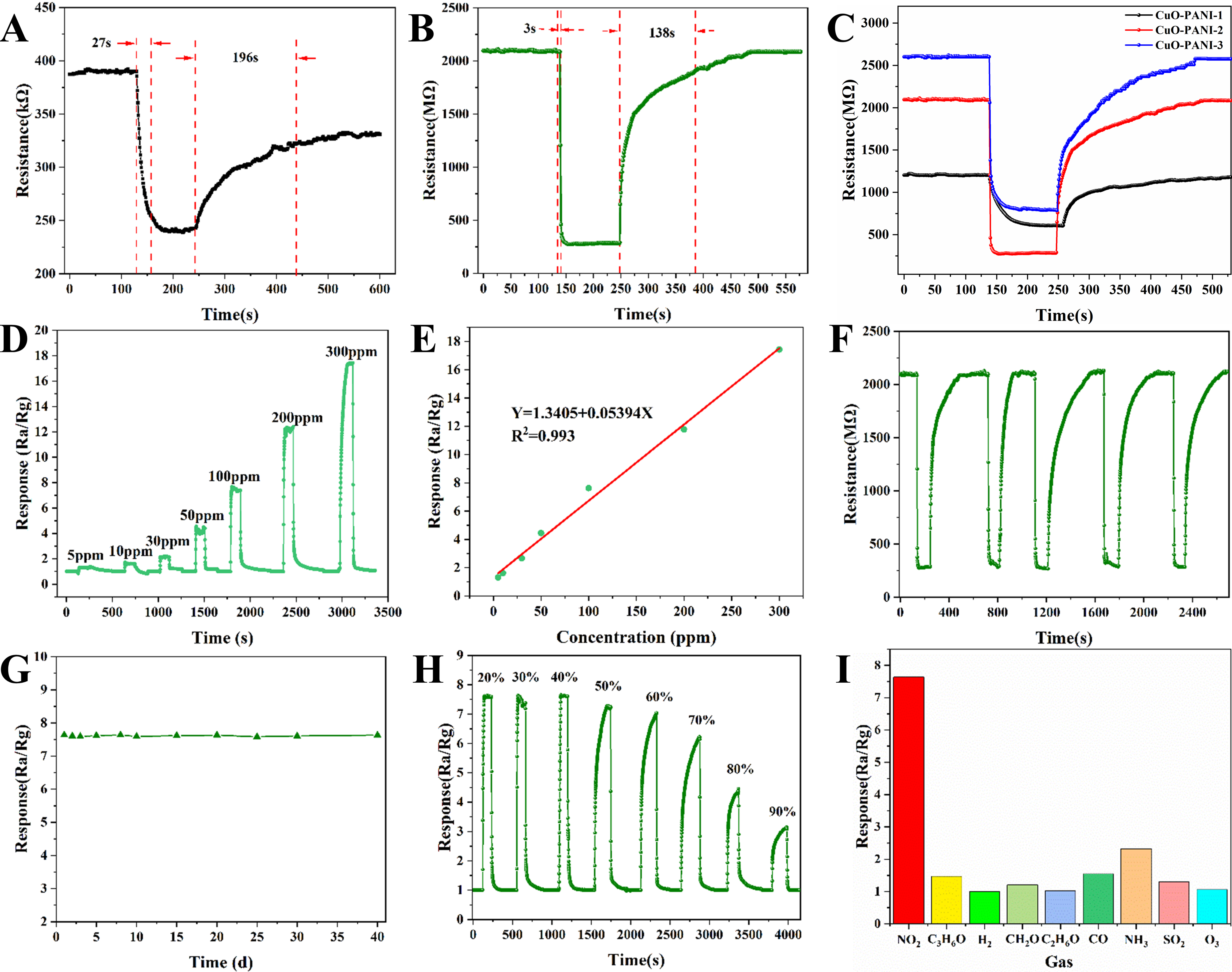
The gas sensing performance of CuO foam, PANI and various CuO-PANI samples for NO2 was measured at RT. The CuO foam and PANI show typical p-type responses to 100 ppm NO2 [Figure 5A and Supplementary Figure 3]. The response values and response/recovery times are 1.55 and 7/21s for the CuO foam and 1.62 and 27/196s for the PANI, respectively. CuO foam exhibits a low response and relatively fast response/recovery to NO2 at RT. PANI has a slightly higher response to NO2 compared with CuO, but the response/recovery is very slow. The different response/recovery rates are due to differences in the gas sensing mechanisms of PANI and CuO. In addition, the resistance of PANI is unable to return to its initial state after reacting with NO2. Therefore, neither CuO nor PANI can meet the requirements of NO2 sensing for practical applications. The response curve of CuO-PANI-2 to 100 ppm NO2 at RT is shown in Figure 5B, which also displays a p-type response. The response value and response/recovery time are 7.63 and 3/138s, respectively. The response of CuO-PANI-2 is about 4.8 times that of CuO and PANI, which is a dramatic improvement. Also, benefiting from the sensing properties of CuO, CuO-PANI shows a faster response/recovery rate than PANI. The resistance in the air (Ra) of CuO, PANI, and CuO-PANI-2 are 16 MΩ,
Figure 5. Response and recovery curve towards 100 ppm NO2 at RT for (A) PANI and (B) CuO-PANI-2; (C) Dynamic NO2 sensing response-recovery curve to 100 ppm NO2 at RT of CuO-PANI-1, CuO-PANI-2 and CuO-PANI-3; (D) The response and (E) fitting function curves for CuO-PANI-2 at RT to various concentrations of NO2; (F) Reproducibility and (G) long-term stability of CuO-PANI-2 at RT to 100 ppm NO2; (H) Effect of RH on the gas-sensing responses of CuO-PANI-2 to 100 ppm NO2 gas at RT; (I) Selectivity of CuO-PANI-2 sensor to 100 ppm of various interfering gases at RT. RT: Room temperature; PANI: polyaniline; RH: relative humidity.
The gas sensing performance of composites with different CuO contents has been tested, as shown in Figure 5C. It can be seen that Ra of the composites increases with CuO content. The responses of CuO-PANI-1, CuO-PANI-2 and CuO-PANI-3 to 100 ppm NO2 at RT are 1.97, 7.63 and 3.25, respectively. CuO-PANI-2 shows a higher response and faster response/recovery process. For CuO-PANI-1, the relatively high content of PANI may affect the amount of chemisorbed oxygen on the CuO surface, thus reducing the active sites for reaction with NO2 and decreasing the response of the sensor. With the decrease of PANI, CuO can react more effectively with oxygen in the air to form chemisorbed oxygen. The synergistic effect of surface adsorbed oxygen and the p-p heterojunction between PANI and CuO significantly improves the sensing performance of the composites. For CuO-PANI-3, the large reduction in p-p heterojunctions due to the low PANI content may degrade the gas-sensing properties of the composite. Because of its higher response and faster response/recovery time, CuO-PANI-2 is chosen for further testing of sensing performance.
The dynamic response curve of CuO-PANI-2 to various NO2 concentrations is displayed in Figure 5D. It can be observed that the response of the sensor increases significantly as the NO2 concentration rises from 5 to 300 ppm. The sensor presents a recognizable signal with a response of 1.35 for NO2 as low as 5 ppm. The low detection limit allows the sensor to accurately recognize low concentrations of NO2, which is of greater practical value. The relationship between response and NO2 concentration in the range of 5-300 ppm can be fitted as Y = 1.3405 + 0.05394X [Figure 5E]. The high correlation coefficient (R2 = 0.993) indicates their good linear relationship, endowing the sensor with the ability to quantify the NO2 concentration based on the signal.
Figure 5F shows the cyclic sensing test results of the CuO-PANI-2 sensor. In each test, the resistance of the sensor exhibits a rapid decrease following NO2 injection and subsequently returns to its initial state upon
At RT, ambient humidity has a great impact on the performance of the sensor[38]. Figure 5H gives the response of CuO-PANI to 100 ppm NO2 under varying RH. The response is essentially unchanged when the RH is below 60%. The response undergoes a substantial decrease when the RH exceeds 70%. As the RH rises to 90%, the response drops to 3.15, which suggests that high humidity has a great effect on the gas-sensing properties of the CuO-PANI composites. Water molecules can interact with ionized oxygen species to form hydroxyl groups, occupying active sites that can react with NO2 molecules, thus reducing sensing performance[39]. The CuO-PANI-2 sensor was exposed to various interfering gases at a concentration of
Additionally, Table 1 presents a comparison of the sensing performance of different kinds of CuO- and PANI-based NO2 sensors in recent years. The results show that the CuO-PANI gas sensor in this paper has the outstanding advantages of fast response and short recovery time for NO2 at RT.
Comparison of sensing performance of CuO-PANI-based NO2 gas sensors in literature
| Materials | Temp. (°C) | Conc. (ppm) | Response | tres/trec (s) | Ref. |
| In2O3/PANI | 250 | 30 | 341.5b | 24/53 | [2] |
| ZnO-PANI | 300 | 10 | 40b | 130/140 | [7] |
| Cu2O-CuO | 187 | 1 | 10.2a | 35/47 | [15] |
| CuO/ZnO | 140 | 50 | 21b | 91/116 | [41] |
| CuO/SnO2 | 150 | 1 | 56a | 65/165 | [42] |
| PANI/Ag2O/GO | 100 | 25 | 5.85b | 100/140 | [43] |
| PANI-WO3 | 50 | 10 | 2.39b | - | [44] |
| Ni@ZnO/PANI | RT | 100 | 75c | 82/399 | [45] |
| CuO-PANI | RT | 100 | 7.63a | 3/138 | This work |
Gas-sensing mechanism
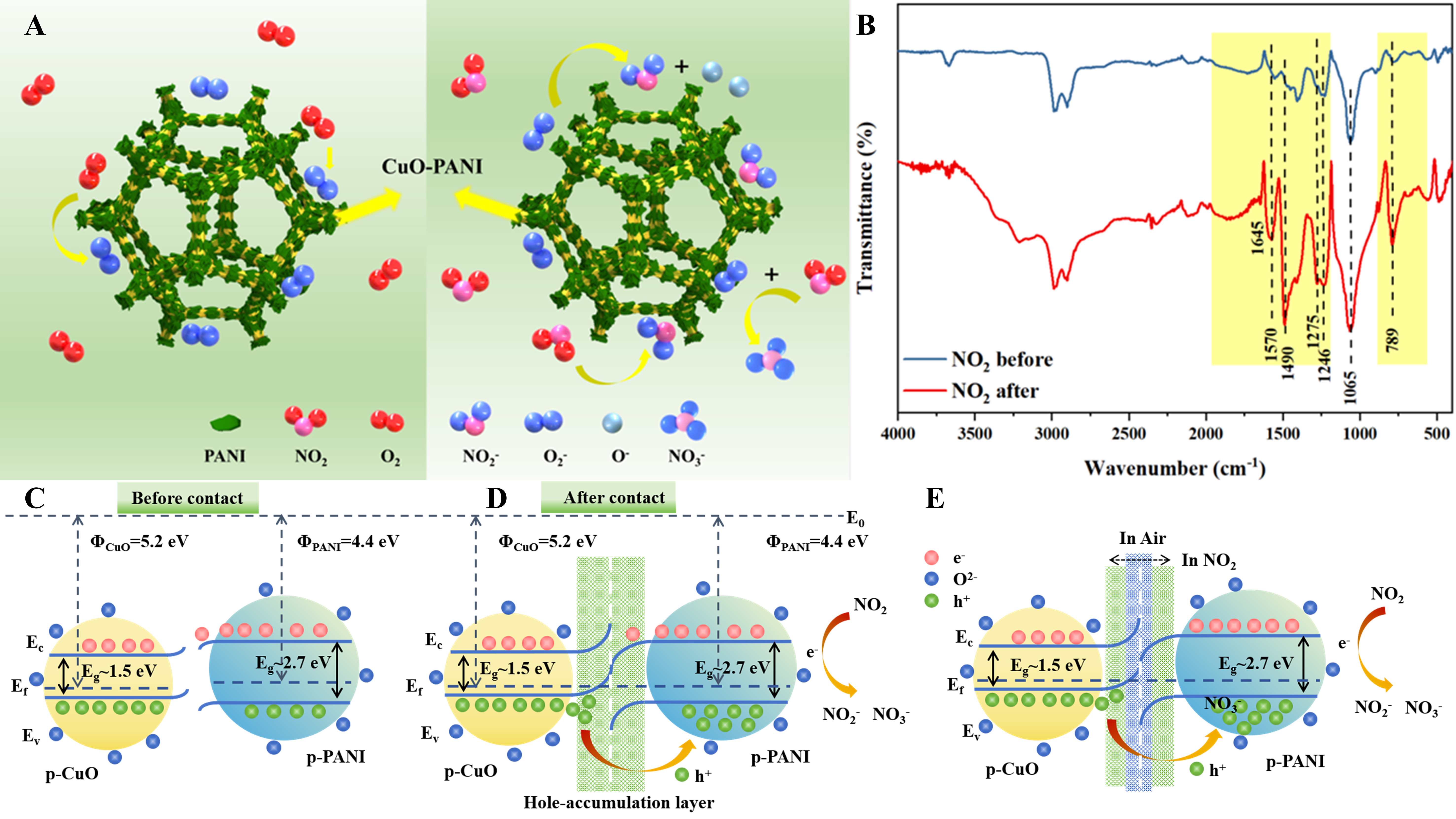
The sensing performance of MOS-based gas sensors depends on the difference in resistance of the sensing material in air and the target gas[46]. However, in the internal reaction with/without the target gas, the resistance is closely related to the concentration of charge carriers at the surface of the material[47]. Therefore, analyzing the relationship between the target gas molecules and the charge carriers is very important for elucidating the gas induction mechanism. The gas sensing mechanism of the CuO-PANI-2 sensor is shown schematically in Figure 6A. A complete gas sensing process mainly consists of three stages: gas chemisorption, change transfer and gas desorption[18]. Both PANI and CuO are typical p-type semiconductors in which holes are the dominant carriers. The adsorption and desorption processes of NO2 mainly occur on the CuO-PANI-2 surface. When the sensor is exposed to air, oxygen molecules will capture electrons from the composite conduction band and form chemisorbed oxygen ions O2- on the surface of CuO-PANI-2, as given in[48]:
Figure 6. (A) Schematic for the NO2 sensing mechanism of CuO-PANI; (B) FT-IR spectra of CuO-PANI before and after exposure to
The depletion of electrons in the conduction band will increase the hole concentration of CuO, which can lead to the formation of a hole accumulation layer (HAL). When the sensor is exposed to NO2, NO2 molecules adsorbed on the surface will capture electrons from the conduction band of CuO, forming NO2- and NO3- [Equations (3-6)]. Fourier transform infrared spectroscopy (FT-IR) tracking of the reaction intermediates was used to analyze the NO2 adsorption process [Figure 6B]. The signal at 789 cm-1 corresponds to the N-O stretching vibration[49]. The discernible peaks 1,065 and 1,246 cm-1 can be seen after NO2 adsorption and assigned to nitrite species (NO2-)[50,51]. The enhanced signal after NO2 adsorption results from the superposition of the nitrite peak with the C-H plane vibration absorption peak. Obviously, new absorption bands at 1,278, 1,490, 1,570 and 1,645 cm-1 are detected, suggesting the generation of monodentate and bridging nitrate[52-54]. The presence of NO2- and NO3- proves the chemisorption of NO2 on the CuO-PANI surface. The above reaction will further deplete the electrons in CuO and increase the thickness of the HAL. The resistance decreases further due to the increase in hole concentration, which is consistent with the dynamic response and recovery behavior. The above reactions are expressed as follows:
The CuO-PANI-based sensors show superior sensing performance for NO2 at RT than CuO. Therefore, there is a high potential that the heterogeneous interface of PANI-modified CuO will play a prominent role in gas sensing performance. During the in-situ growth process, PANI can form p-p heterojunctions with CuO foam at the interface. Figure 6C shows the energy band structures of PANI and CuO before contact. The work functions of CuO and PANI are around 5.2 and 4.4 eV, respectively. Driven by the difference in work functions, the interaction between CuO and PANI promotes the migration of holes from CuO to PANI and the reverse migration of electrons until the equilibrium is established at both Fermi levels [Figure 6D]. It may eventually result in the formation of a hole depletion layer (HDL) on the CuO side and a HAL on the PANI side. As a result, the amount of change in the electrical signal generated by the gas reaction is amplified. As shown in Figure 6E, the formation of p-p heterojunctions and the change of HAL thickness at the heterojunction boundary[55] greatly enhance the sensor performance. The inherent electric field propels the charge transfer across the p-p junction to generate a depletion layer, which will increase the resistance of the sensor and establish a potential barrier. In the presence of air, electrons trapped by adsorbed oxygen augment the availability of holes near the surface. When encountering NO2 gas, the chemisorbed NO2 escalates the number of available holes near the material surface, thereby thickening the HAL and further reducing resistance, as elucidated in Figure 6D[40]. In addition, according to the XPS results, the small amount of Cu2O in CuO can provide a large number of OV and lattice defects, which can also promote the adsorption of NO2 gas[56], thus enhancing the sensing ability. Furthermore, the 3D macroporous structure of CuO-PANI can accelerate the diffusion of NO2 gas, thus promoting the gas sensing performance. We have added a detailed analysis of the mechanism for the excellent sensing performance of CuO-PANI-based gas sensors at RT. From the performance tests, it is observed that the CuO-PANI composites show good response values at RT. This is mainly attributed to two factors: Firstly, the excellent electron transport ability of PANI enables the electron migration induced by NO2 adsorption to be realized. Secondly, the heterogeneous interface formed by PANI and CuO exhibits high activity, providing active sites for NO2 adsorption on the material. The conductivity of PANI is influenced by its dual capability to transit the charge carriers through the polymer backbone and to promote carrier hopping between polymer chains. As a result, PANI molecules readily react with substances that add or remove protons or electrons, such as protonating, deprotonating, reducing, and oxidizing agents, leading to changes in their conductivity even at RT[57]. When PANI is exposed to oxidizing NO2 gas, the oxidization degree of PANI will increase and its conductivity will decrease. On the other hand, since NO2 is a highly oxidizing species, its contact with the electron network of PANI is likely to result in the transfer of an electron from the polymer to the gas. When this occurs, the polymer becomes positively charged, increasing its conductivity. In addition, integrating p-type PANI with p-type CuO can provide important electronic properties that are expected to improve gas sensing performance through synergistic effects. Specifically, the formation of p-p heterojunctions in CuO-PANI composites will significantly improve the sensor sensitivity. This composite structure leads to Fermi alignment at the interface, which promotes the development of extensive charge regions and facilitates efficient charge transfer[58].
CONCLUSIONS
In summary, we designed a unique CuO-PANI sensor capable of detecting NO2 at RT. CuO-PANI composites were prepared by thermal oxidation of copper foam and in situ polymerization of aniline. Characterization results showed that PANI was uniformly distributed on the surface of the CuO. Gas sensing test results indicated that the composite exhibited a response value of 7.63 to 100 ppm NO2 at RT with a response time of 3 s and a recovery time of 138 s. The response of CuO-PANI-2 is about 4.8 times that of CuO and PANI. Additionally, it exhibited excellent reproducibility, gas selectivity, and a lower limit of detection. The primary factor contributing to the improved gas-sensitive performance of the CuO-PANI-2 composite was the formation of a p-p heterojunction between the PANI and CuO. The CuO-PANI room-temperature sensor offers a plausible solution to the constraints of PANI and metal oxide gas-sensitive materials in practical applications.
DECLARATIONS
Authors’ contributions
Experiment design, data acquisition, writing - original draft preparation, formal analysis: Zhang, Y., Zhang, Z., Li, S., Li, F.
Supervision, investigation, visualization, writing - review and editing: Zhan, Z., Fang, F., Wang, P.
Validation, formal analysis: Fang, F., Zhang, H., Liu, S.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22008223, 22208325), the National Key R&D Program of China (2023YFB3809400), and the State Key Laboratory of Fine Chemicals, Dalian University of Technology (KF2206).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Bai, H.; Guo, H.; Feng, C.; et al. A room-temperature chemiresistive NO2 sensor based on one-step synthesized SnO2 nanospheres functionalized with Pd nanoparticles and rGO nanosheets. Appl. Surf. Sci. 2022, 575, 151698.
2. Amu-Darko, J. N. O.; Hussain, S.; Gong, Q.; et al. Highly sensitive In2O3/PANI nanosheets gas sensor for NO2 detection. J. Environ. Chem. Eng. 2023, 11, 109211.
3. Inaba, M.; Oda, T.; Kono, M.; et al. Effect of mixing ratio on NO2 gas sensor response with SnO2-decorated carbon nanotube channels fabricated by one-step dielectrophoretic assembly. Sensor. Actuat. B. Chem. 2021, 344, 130257.
4. Chu, S. Y.; Wu, M. J.; Yeh, T. H.; Lee, C. T.; Lee, H. Y. Sensing mechanism and characterization of NO2 gas sensors using gold-black NP-decorated Ga2O3 nanorod sensing membranes. ACS. Sens. 2024, 9, 118-25.
5. Yuan, Z.; Zhao, Q.; Duan, Z.; et al. Ag2Te nanowires for humidity-resistant trace-level NO2 detection at room temperature. Sensor. Actuat. B. Chem. 2022, 363, 131790.
6. Sihag, S.; Dahiya, R.; Rani, S.; et al. Low ppm NO2 detection through advanced ultrasensitive copper oxide gas sensor. Discov. Nano. 2024, 19, 107.
7. Bonyani, M.; Zebarjad, S. M.; Janghorban, K.; Kim, J.; Kim, H. W.; Kim, S. S. Enhanced NO2 gas sensing properties of ZnO-PANI composite nanofibers. Ceram. Int. 2023, 49, 1238-49.
8. Feng, S.; Farha, F.; Li, Q.; et al. Review on smart gas sensing technology. Sensors 2019, 19, 3760.
9. Xin, C.; Zhao, L.; Xing, Y.; et al. An in situ exsolution strategy to prepare metallic Sn modified SnO2 with oxygen vacancies for ppb-level NO2 detection. Sensor. Actuat. B. Chem. 2024, 419, 136232.
10. Jiakun, W.; Hirata, M. Research into normal temperature gas-sensitive characteristics of polyaniline material. Sensor. Actuat. B. Chem. 1993, 12, 11-3.
11. Byoun, Y.; Jin, C.; Choi, S. Strategy for sensitive and selective NO2 detection at low temperatures utilizing p-type TeO2 nanowire-based sensors by formation of discrete n-type ZnO nanoclusters. Ceram. Int. 2020, 46, 19365-74.
12. Li, Y.; Jiang, H.; Yan, X.; et al. 3D nanostructured CuxO modified copper foam as a binder-free electrode for all-solid-state supercapacitor. Ceram. Int. 2021, 47, 31138-48.
13. Nakate, U. T.; Patil, P.; Na, S.; Yu, Y.; Suh, E.; Hahn, Y. Fabrication and enhanced carbon monoxide gas sensing performance of p-CuO/n-TiO2 heterojunction device. Colloid. Surface. A. 2021, 612, 125962.
14. Hmeed, A. A. A.; Al-Jumaili, H. S. NO2 gas sensor properties of In2O3-CuO nanocomposite thin films prepared by chemical spray pyrolysis. IOP. Conf. Ser. Mater. Sci. Eng. 2021, 1095, 012008.
15. Wang, N.; Tao, W.; Gong, X.; et al. Highly sensitive and selective NO2 gas sensor fabricated from Cu2O-CuO microflowers. Sensor. Actuat. B. Chem. 2022, 362, 131803.
16. Ambedkar, A. K.; Gautam, D.; Vikal, S.; et al. Ocimum sanctum leaf extract-assisted green synthesis of Pd-doped CuO nanoparticles for highly sensitive and selective NO2 gas sensors. ACS. Omega. 2023, 8, 29663-73.
17. Hsueh, T.; Li, P.; Fang, S.; Hsu, C. A vertical CuO-NWS/MEMS NO2 gas sensor that is produced by sputtering. Sensor. Actuat. B. Chem. 2022, 355, 131260.
18. Govind, A.; Bharathi, P.; Mohan, M. K.; Archana, J.; Harish, S.; Navaneethan, M. Highly sensitive near room temperature operable NO2 gas-sensor for enhanced selectivity via nanoporous CuO@ZnO heterostructures. J. Environ. Chem. Eng. 2023, 11, 110056.
19. Wang, Z.; Chang, X.; Li, J.; et al. Room-temperature NH3 sensor based on CoFe2O4/PANI composite with porous structure. Mater. Res. Bull. 2024, 175, 112774.
20. Zhang, B.; Shang, F.; Shi, X.; et al. Polyaniline/CuO nanoparticle composites for use in selective H2S sensors. ACS. Appl. Nano. Mater. 2023, 6, 18413-25.
21. Wang, Y.; Liu, A.; Han, Y.; Li, T. Sensors based on conductive polymers and their composites: a review. Polym. Int. 2020, 69, 7-17.
22. Das, M.; Roy, S. Polypyrrole and associated hybrid nanocomposites as chemiresistive gas sensors: a comprehensive review. Mat. Sci. Semicon. Proc. 2021, 121, 105332.
23. Liu, A.; Lv, S.; Jiang, L.; et al. The gas sensor utilizing polyaniline/MoS2 nanosheets/SnO2 nanotubes for the room temperature detection of ammonia. Sensor. Actuat. B. Chem. 2021, 332, 129444.
24. Kumawat, M.; Thapliyal, D.; Verros, G. D.; et al. PANI-based hydrogen sulfide gas sensors. Coatings 2022, 12, 186.
25. Esmaeili, C.; Ashtiani, S.; Regmi, C.; et al. Preparation and characterisation of NH3 gas sensor based on PANI/Fe-doped CeO2 nanocomposite. Heliyon 2024, 10, e34801.
26. Xu, H.; Ju, D.; Li, W.; et al. Low-working-temperature, fast-response-speed NO2 sensor with nanoporous-SnO2/polyaniline double-layered film. Sensor. Actuat. B. Chem. 2016, 224, 654-60.
27. Nguyet, T. T.; Van, D. L.; Nguyet, Q. T. M.; et al. Novel synthesis of a PANI/ZnO nanohybrid for enhanced NO2 gas sensing performance at low temperatures. J. Electron. Mater. 2023, 52, 304-19.
28. Patowary B, Laskar S, Narzary R, Mondal A. Synthesis, characterization, and study of NO2 gas sensing behavior of CSA doped PANi-Ta2O5 nanocomposite. IEEE. Sensors. J. 2020, 20, 3429-41.
29. Jian, Y.; Hu, W.; Zhao, Z.; et al. Gas sensors based on chemi-resistive hybrid functional nanomaterials. Nanomicro. Lett. 2020, 12, 71.
30. Ghaleghafi, E.; Rahmani, M. B. Reversible room temperature ammonia sensor based on the synthesized MoS2/PANI nanocomposites. Phys. Scr. 2023, 98, 075801.
31. Han, D.; Wang, Y.; Wang, Y.; et al. Machine-learning-assisted n-GaN-Au/PANI gas sensor array for intelligent and ultra-accurate ammonia recognition. Chem. Eng. J. 2024, 495, 153705.
32. Li, F.; Jing, J.; Li, J.; et al. Fabrication of ZnO-SnO2 heterojunction inverse opal photonic balls for chemiresistive acetone sensing. Sensor. Actuat. B. Chem. 2024, 400, 134887.
33. Li, F.; Jing, J.; Li, J.; et al. Fabrication of Pt cluster-loaded In2O3 inverse opal photonic crystals for fast and highly sensitive ethanol sensing. J. Mater. Chem. C. 2023, 11, 1899-907.
34. Sui, C.; Zhang, M.; Li, Y.; et al. Pd@Pt core-shell nanocrystal-decorated ZnO nanosheets for ppt-level NO2 detection. ACS. Sens. 2024, 9, 1967-77.
35. Li, P.; Wang, Z.; Feng, Y.; Feng, B.; Cheng, D.; Wei, J. Synergistic sensitization effects of single-atom gold and cerium dopants on mesoporous SnO2 nanospheres for enhanced volatile sulfur compound sensing. Mater. Horiz. 2024, 11, 3038-47.
36. Liu, W.; Zou, J.; Li, S.; et al. Pd/In2O3-based bilayer H2 sensor with high resistance to silicone toxicity and ultra-fast response. Int. J. Hydrogen. Energy. 2023, 48, 5743-53.
37. Chen, Y.; Fu, H.; Bai, Y.; et al. Room-temperature NH3-sensor: SnO2@PANI core-shell hollow spheres. Sensor. Actuat. B. Chem. 2024, 412, 135784.
38. Liu, F.; Zhao, J.; Chen, C.; et al. Dual enhancement in response and anti-humidity properties of a triethylamine sensor based on trimethoxypropylsilane self-assembled functionalized In2O3. Sensor. Actuat. B. Chem. 2024, 417, 136120.
39. Qiao, L.; Jia, X.; Zhang, J.; et al. A highly responsive, moisture resistant diabetes diagnostic gas sensor with Pt-loaded porous GO/ZnO. Sensor. Actuat. B. Chem. 2024, 418, 136275.
40. Haldar, T.; Shiu, J. W.; Yang, R. X.; et al. Exploring MOF-derived CuO/rGO heterostructures for highly efficient room temperature CO2 sensors. ACS. Sens. 2024, 9, 5856-65.
41. Li, S.; Yu, L.; Zhang, C.; et al. Controllable synthesis of heterostructured CuO–ZnO microspheres for NO2 gas sensors. Sensor. Actuat. B. Chem. 2024, 417, 136179.
42. Shi, Z.; Hsiao, Y.; Chang, S.; Chang, S. Synthesis and characterization of an oxygen-controlled CuO/SnO2 sensor for NO2 detection. Sensor. Actuat. B. Chem. 2024, 421, 136517.
43. Umar, A.; Ibrahim, A. A.; Algadi, H.; et al. Enhanced NO2 gas sensor device based on supramolecularly assembled polyaniline/silver oxide/graphene oxide composites. Ceram. Int. 2021, 47, 25696-707.
44. He, W.; Zhao, Y.; Xiong, Y. Bilayer polyaniline-WO3 thin-film sensors sensitive to NO2. ACS. Omega. 2020, 5, 9744-51.
45. Jain, S.; Karmakar, N.; Shah, A.; Shimpi, N. G. Development of Ni doped ZnO/polyaniline nanocomposites as high response room temperature NO2 sensor. Mater. Sci. Eng. B. 2019, 247, 114381.
46. Zhang, Y.; Li, Y.; Liu, W.; Li, F.; Zhan, Z. Design of SnO2-based CH4 sensors with reactive anti-poisoning layers: excellent stability and high resistance to hexamethyldisiloxane. J. Mater. Sci. 2023, 58, 2140-55.
47. Zhang, S.; Lai, X.; Xiao, R.; et al. Size-dependent response of hydrothermally grown SnO2 for a high-performance NO2 sensor and the impact of oxygen. ACS. Sens. 2024, 9, 195-205.
48. Guo, F.; Feng, C.; Zhang, Z.; et al. A room-temperature NO2 sensor based on Ti3C2TX MXene modified with sphere-like CuO. Sensor. Actuat. B. Chem. 2023, 375, 132885.
49. Köglmaier, M.; Joost, T.; Kronseder, M.; Kunz, W. Characterization of the interaction of nitric oxide/nitrogen dioxide with the polymer surfaces in ECMO devices. J. Polym. Res. 2024, 31, 4109.
50. Jin, Z.; Mou, Y.; Zhao, J.; et al. Anchoring Ni atomic clusters on yolk-shell In2O3 microspheres for highly sensitive, ultra-rapid responding/recovering ppb-level NO2 detections. Chem. Eng. J. 2024, 499, 156531.
51. Xin, Y.; Zhu, Q.; Gao, T.; et al. Photocatalytic NO removal over defective Bi/BiOBr nanoflowers: the inhibition of toxic NO2 intermediate via high humidity. Appl. Catal. B. Environ. 2023, 324, 122238.
52. Hadjiivanov, K. I. Identification of neutral and charged NxOy surface species by IR spectroscopy. Catal. Rev. 2000, 42, 71-144.
53. Chen, Q.; Long, H.; Chen, M.; Rao, Y.; Li, X.; Huang, Y. In situ construction of biocompatible Z-scheme α-Bi2O3/CuBi2O4 heterojunction for NO removal under visible light. Appl. Catal. B. Environ. 2020, 272, 119008.
54. Sivachandiran, L.; Thevenet, F.; Rousseau, A.; Bianchi, D. NO2 adsorption mechanism on TiO2: an in-situ transmission infrared spectroscopy study. Appl. Catal. B. Environ. 2016, 198, 411-9.
55. Suh, J. M.; Sohn, W.; Shim, Y. S.; et al. p-p Heterojunction of nickel oxide-decorated cobalt oxide nanorods for enhanced sensitivity and selectivity toward volatile organic compounds. ACS. Appl. Mater. Interfaces. 2018, 10, 1050-8.
56. Li, R.; Wang, Q.; Wang, Y.; et al. Unraveling the effect of oxygen vacancy on WO3 surface for efficient NO2 detection at low temperature. ACS. Appl. Mater. Interfaces. 2024, 16, 51738-47.
57. Yan, X.; Han, Z.; Yang, Y.; Tay, B. NO2 gas sensing with polyaniline nanofibers synthesized by a facile aqueous/organic interfacial polymerization. Sensor. Actuat. B. Chem. 2007, 123, 107-13.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].