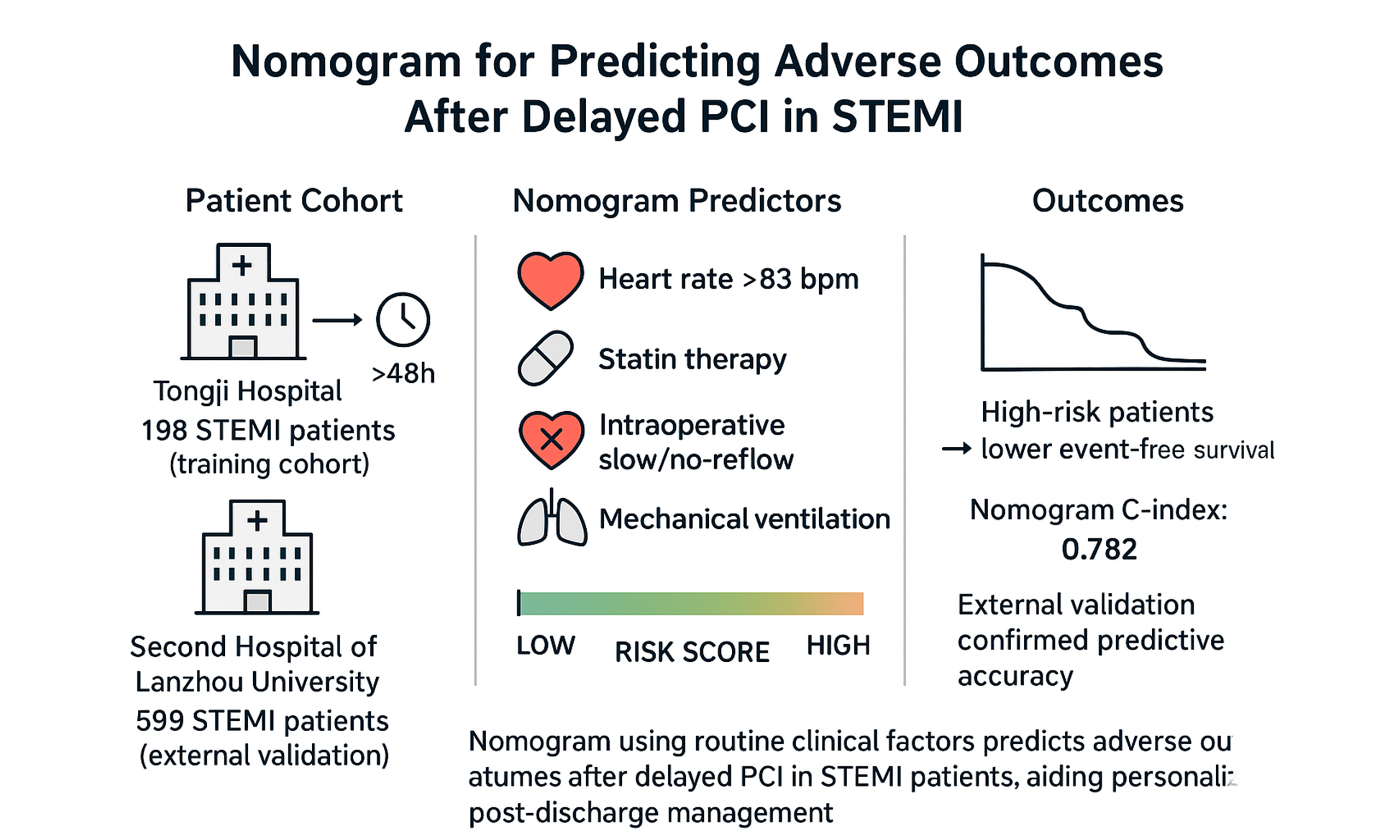
Development and external validation of a nomogram to predict adverse outcomes following delayed PCI in STEMI
Abstract
Aim: Reliable prognostic tools remain limited for patients with ST-segment elevation myocardial infarction (STEMI) undergoing percutaneous coronary intervention (PCI) more than 48 h after symptom onset. This study aimed to develop and externally validate a nomogram based on routinely available in-hospital clinical variables to predict post-discharge adverse outcomes in this population.
Methods: We retrospectively analyzed data from Tongji Hospital between June 2019 and August 2022 and identified 198 STEMI patients who underwent delayed PCI as the training cohort. Independent predictors of composite adverse events, defined as all-cause mortality, nonfatal myocardial infarction, and New York Heart Association class IV heart failure, were identified using multivariate Cox proportional hazards regression. A nomogram was subsequently constructed and internally validated using bootstrap resampling. External validation was performed in an independent cohort of 599 patients treated at the Second Hospital of Lanzhou University, with a median follow-up duration of 20 months.
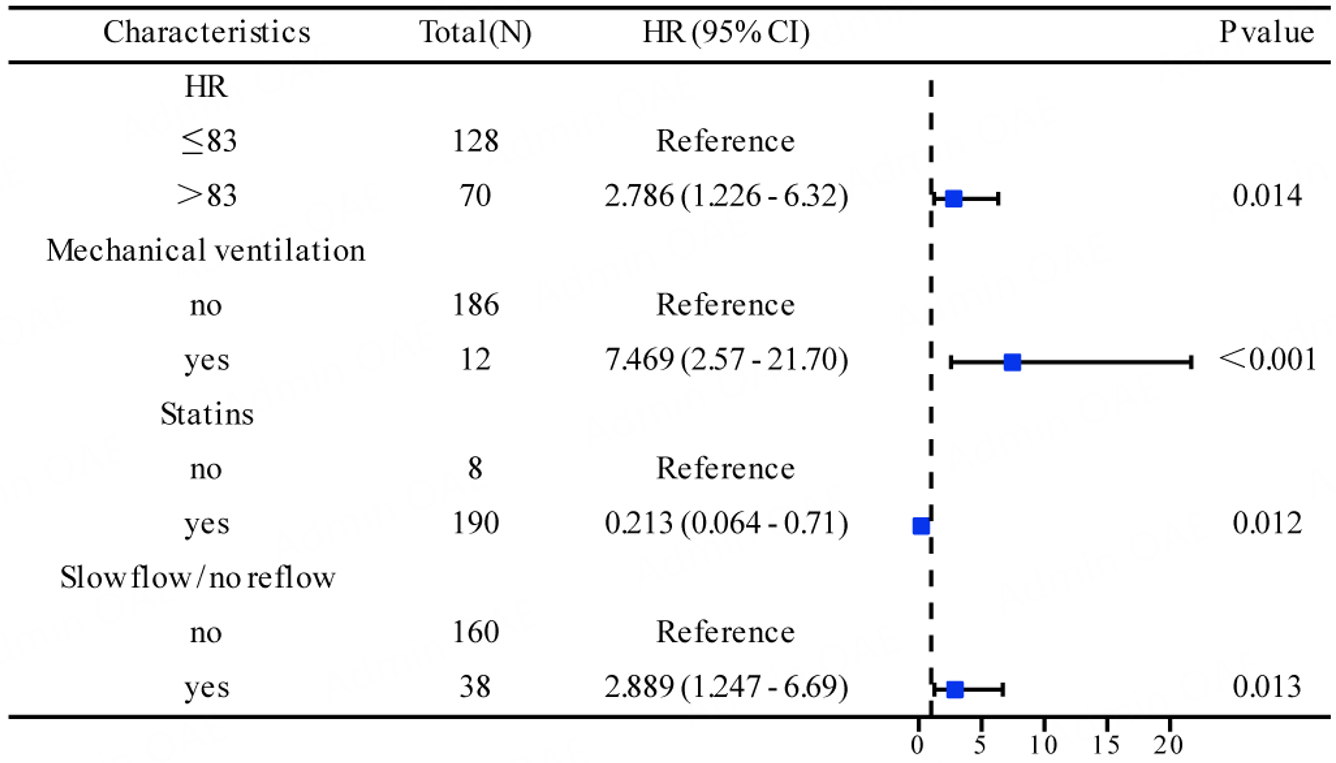
Results: Four variables were identified as independent predictors of adverse outcomes and incorporated into the nomogram: (1) heart rate > 83 beats per minute (hazard ratio [HR] 2.786, 95% confidence interval [CI]: 1.226-6.32, P = 0.014); (2) absence of statin therapy (HR 0.213, 95%CI: 0.064-0.71, P = 0.012); (3) intraoperative slow-flow/no-reflow phenomenon (HR 2.889, 95%CI: 1.247-6.69, P = 0.013); and (4) requirement for mechanical ventilation (HR 7.469, 95%CI: 2.57-21.70, P < 0.001). The nomogram demonstrated good discrimination and calibration in the training cohort, with a concordance index of 0.782. External validation confirmed its robust predictive performance. Patients classified as high risk exhibited significantly lower event-free survival compared with those at low risk (P < 0.0001).
Conclusion: This validated nomogram, derived from routinely collected clinical variables, provides reliable prediction of adverse outcomes in STEMI patients undergoing delayed PCI and may facilitate individualized risk stratification and optimized post-discharge management.
Keywords
INTRODUCTION
Acute ST-segment elevation myocardial infarction (STEMI) represents a major global health burden, associated with substantial morbidity and mortality worldwide[1]. The primary therapeutic goal in STEMI management is the prompt restoration of coronary blood flow to salvage ischemic myocardium, most commonly achieved through percutaneous coronary intervention (PCI)[2]. Despite advances in interventional techniques, long-term outcomes after PCI remain suboptimal, with a substantial proportion of patients experiencing major adverse cardiac events, including death, recurrent myocardial infarction, and advanced heart failure[3-5]. Given the heterogeneity in clinical presentation and disease severity, accurate risk stratification is essential to guide post-discharge management and long-term follow-up.
In clinical settings, a subset of STEMI patients receive PCI treatment more than 48 h after their symptoms initially emerged, sometimes due to delayed medical attention, diagnostic uncertainty among healthcare personnel, or organizational challenges. While extensive evidence has focused on early reperfusion, the prognostic profile of patients undergoing delayed PCI remains insufficiently characterized. Identifying high-risk individuals in this specific population is critical for optimizing subsequent management strategies.
In-hospital clinical variables—such as vital signs, pharmacological treatments, intraoperative coronary flow status, and the need for mechanical ventilation—may reflect disease severity and procedural complexity and therefore provide valuable prognostic information. However, existing prediction models have largely focused on early PCI populations or short-term outcomes and have not been specifically developed for STEMI patients treated with delayed PCI.
Nomograms offer a practical and intuitive approach to individualized risk prediction by integrating multiple prognostic variables into a single quantitative tool[6]. As a result, the study sought to develop and validate a nomogram based on routine clinical data to predict the likelihood of long-term adverse outcomes in STEMI patients undergoing delayed PCI.
MATERIALS AND METHODS
Study population
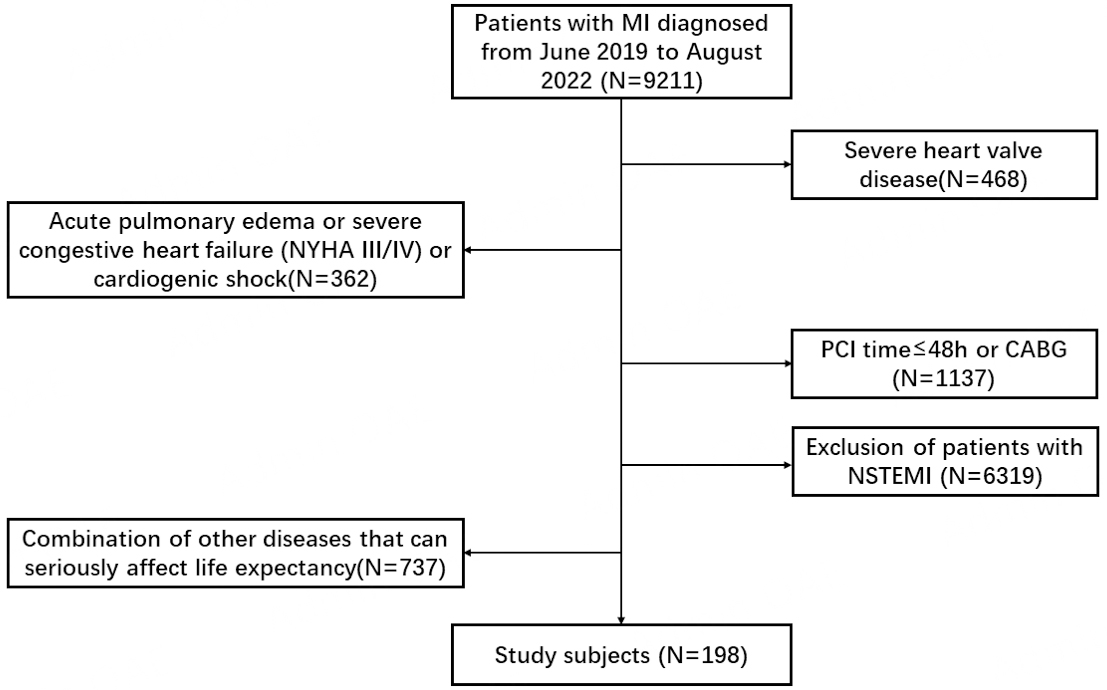
Between June 2019 and August 2022, Tongji Hospital, affiliated with Tongji Medical College of Huazhong University of Science and Technology, received 198 consecutive patients diagnosed with STEMI who subsequently underwent delayed PCI [Figure 1]. Delayed PCI was defined as a procedure performed at least 48 h after the initial onset of ischemic symptoms.
The diagnosis of STEMI was established according to current guideline criteria[7], including: (1) Chest pain caused by insufficient blood flow to the heart that lasts more than 20 min and does not go away even after taking nitroglycerin, and (2) Electrocardiograph (ECG) signs indicating ST-segment elevation in at least two neighboring leads (measuring up to 0.2 mV in chest leads or 0.1 mV in limb leads), or the sudden appearance of a complete left or right bundle branch block.
Eligible patients were required to meet the following inclusion criteria: confirmed STEMI with symptom onset ≥ 48 h at the time of PCI. The exclusion criteria were: (1) abnormalities of the heart's critical valves; (2) acute pulmonary edema or advanced heart failure (New York Heart Association (NYHA) class III or IV upon entry, or heart-related shock); (3) people undergoing urgent angioplasty or bypass surgery for massive heart attacks; (4) heart attacks that do not exhibit the usual ST-segment changes; and (5) other health issues that could significantly reduce life expectancy, such as cancer or severe dysfunction of the liver, kidneys, lungs, endocrine system, nerves, or blood. The ethics review committees of Tongji Hospital, Tongji Medical College of Huazhong University of Science and Technology (Wuhan, China), and The Second Hospital of Lanzhou University (Lanzhou, China) approved this study, which was conducted in accordance with the Declaration of Helsinki.
PCI procedure
Cardiac catheterization was performed with 5-Fr Judkins catheters through radial or femoral access, as per normal protocol. Prior to the intervention, patients were treated according to normal coronary artery disease guidelines and dual antiplatelet regimens. Culprit lesions were identified based on coronary angiographic findings in conjunction with electrocardiographic and echocardiographic assessments. All procedures were performed by experienced interventional cardiologists. Based on the distinct anatomical characteristics of each coronary blockage, the interventional team selected between balloon angioplasty and coronary stent implantation; the doctor made the ultimate decision.
In cases of intraoperative slow-flow or no-reflow, prompt rescue treatments were administered as clinically indicated, including intracoronary pharmacological therapy and/or mechanical interventions. Coronary artery disease was classified as serious when there was a 70% or greater stenosis in arteries other than the left main, or when the left main artery had a 50% or more occlusion[8].
Myocardial perfusion was measured using the Thrombolysis In Myocardial Infarction (TIMI) flow classification system[9], and evaluated using the Rentrop grading scheme[10]. No-reflow was defined as a significant decrease in coronary blood flow following the procedure, even when there was no major heart artery obstruction, arterial lining tears, muscle constriction, or blood clot formation at the initial site. TIMI flow grades 0-1 were classified as no-reflow, whereas TIMI flow grade 2 was defined as slow-flow[11,12].
Follow-up process
The primary outcome of this study was event-free survival (EFS)[13], measured as the interval between initial coronary catheterization and the first composite adverse outcome. The composite outcome included all-cause mortality, nonfatal myocardial infarction, or progression to advanced heart failure (NYHA class IV), assessed up to the end of the study period or the final follow-up date. Subsequent assessments were conducted during scheduled phone calls and standard clinic check-ups at 1, 3, and 6 months after hospital discharge. After that first time, monitoring continued every six months. The median observation time was 20 months (range: 1–51 months), with data collection ending in September 2023.
Healthcare practitioners identified key incidents by meticulously reviewing hospital documentation, analyzing doctor ratings from routine check-ups, and maintaining open lines of communication with patients and their families. All clinical endpoints were independently adjudicated by two experienced cardiologists. All subjects completed the study.
Statistical analysis
Associations between continuous variables and EFS were first explored to determine their functional relationships. Optimal cutoff values for continuous variables included in the nomogram were identified using maximally selected rank statistics implemented in the survminer package in R, thereby enhancing clinical interpretability.
Missing data were present across several variables; however, the proportion of missing values did not exceed 30% for any variable. The pattern of missingness for all study variables is presented in
Prior to multivariable modeling, univariate Cox proportional hazards regression analyses were performed to evaluate the associations between candidate variables and EFS, allowing for an inclusive initial screening. Given the limited number of outcome events, inclusion of all candidate variables in a multivariable model could have resulted in overfitting. Therefore, a parsimonious multivariable Cox proportional hazards regression model was constructed by including not only variables with statistical significance in univariate analyses (P < 0.05) but also clinically relevant factors identified by experienced clinicians.
Based on the results of the multivariable analysis, nomograms were developed to estimate 6-month and 12-month EFS probabilities. Model discrimination was assessed using the concordance index (C-index). Calibration was evaluated by comparing predicted and observed outcomes using calibration plots generated from 1,000 bootstrap resamples. The clinical utility of the nomogram was further examined using decision curve analysis (DCA).
Patients were stratified into different risk groups according to nomogram-derived risk scores, and Kaplan-Meier survival curves were generated to compare EFS across risk strata. Differences between groups were assessed using the log-rank test.
All statistical analyses were conducted using SPSS software (version 26.0; IBM Corp., Chicago, IL, USA) and R software (version 4.3.1; R Foundation for Statistical Computing, Vienna, Austria). Nomograms and calibration plots were generated using the rms package. DCA was performed with the dcurves package, time-dependent receiver operating characteristic (ROC) curves were constructed using the survival ROC package, and Kaplan-Meier analyses were conducted using the survival and survminer packages. Internal validation was performed using bootstrap resampling with 1,000 iterations. All statistical tests were two-sided, and a P value < 0.05 was considered statistically significant.
RESULTS
Patient characteristics
A total of 198 patients with STEMI who underwent delayed PCI were included in the study. Their demographic, clinical, laboratory, and procedural details are neatly outlined in Table 1 (refer to Figure 1 for more information). The mean age was 62 years (range, 49-75). Of the 198 patients, 146 (73.7%) were male. Continuous variables were categorized according to predefined optimal cutoff values. Overall, patients exhibited substantial heterogeneity in baseline clinical and laboratory parameters, reflecting the broad spectrum of disease severity among STEMI patients undergoing delayed PCI. Detailed distributions of laboratory indices, hemodynamic parameters, and cardiac biomarkers are presented in Table 1.
Baseline demographic and clinical characteristics of STEMI patients undergoing delayed PCI
| Characteristics | Overall (N = 198), n (%) | Events (N = 33), n (%) |
| Male | 146 (73.7) | 20 (60.6) |
| Age (years) | ||
| ≤ 55 | 62 (31.3) | 4 (12.1) |
| > 55 | 136 (68.7) | 29 (87.9) |
| Hypertension | 102 (51.5) | 22 (66.7) |
| Diabetes | 56 (28.3) | 12 (36.4) |
| Hyperlipidemia | 16 (8.1) | 3 (9.1) |
| Renal insufficiency | 14 (7.1) | 2 (6.1) |
| BMI (kg/m2) | ||
| ≤ 20.48 | 26 (13.1) | 8 (24.2) |
| > 20.48 | 172 (86.9) | 25 (75.8) |
| HR (beats/min) | ||
| ≤ 83 | 128 (64.6) | 11 (33.3) |
| > 83 | 70 (35.4) | 22 (66.7) |
| SBP (mmHg) | ||
| ≤ 102 | 24 (12.1) | 8 (24.2) |
| > 102 | 174 (87.9) | 25 (75.8) |
| DBP (mmHg) | ||
| ≤ 65 | 38 (19.2) | 11 (33.3) |
| > 65 | 160 (80.8) | 22 (66.7) |
| WBC (×109/L) | ||
| ≤ 11.42 | 148 (74.7) | 19 (57.6) |
| > 11.42 | 50 (25.3) | 14 (42.4) |
| Hb (g/L) | ||
| ≤ 122 | 66 (33.3) | 20 (60.6) |
| > 122 | 132 (66.7) | 13 (39.4) |
| PLT (×109/L) | ||
| ≤ 340 | 178 (89.9) | 27 (81.8) |
| > 340 | 20 (10.1) | 6 (18.2) |
| ALT (U/L) | ||
| ≤ 54 | 165 (83.3) | 22 (66.7) |
| > 54 | 33 (16.7) | 11 (33.3) |
| AST (U/L) | ||
| ≤ 150 | 168 (84.8) | 22 (66.7) |
| > 150 | 30 (15.2) | 11 (33.3) |
| LDH (U/L) | ||
| ≤ 543 | 151 (76.3) | 21 (63.6) |
| > 543 | 47 (23.7) | 12 (36.4) |
| Cr (μmol/L) | ||
| ≤ 113 | 151 (76.3) | 19 (57.6) |
| > 113 | 47 (142.4) | 14 (7.1) |
| eGFR (mL/min/1.73 m2) | ||
| ≤ 30.9 | 24 (12.1) | 10 (30.3) |
| > 30.9 | 174 (87.9) | 23 (69.7) |
| TC (mmol/L) | ||
| ≤ 2.72 | 19 (9.6) | 7 (21.2) |
| > 2.72 | 179 (90.4) | 26 (78.8) |
| TG (mmol/L) | ||
| ≤ 2.10 | 159 (80.3) | 31 (93.9) |
| > 2.10 | 39 (19.7) | 2 (6.1) |
| HDL-C (mmol/L) | ||
| ≤ 0.66 | 128 (64.6) | 20 (60.6) |
| > 0.66 | 70 (35.4) | 13 (39.4) |
| LDL-C (mmol/L) | ||
| ≤ 1.45 | 19 (9.6) | 6 (18.2) |
| > 1.45 | 179 (90.4) | 27 (81.8) |
| K+ (mmol/L) | ||
| ≤ 4.54 | 162 (81.8) | 21 (63.6) |
| > 4.54 | 36 (18.2) | 12 (36.4) |
| Glu (mmol/L) | ||
| ≤ 10.03 | 165 (83.3) | 22 (66.7) |
| > 10.03 | 33 (16.7) | 11 (33.3) |
| Hs-CRP (mg/L) | ||
| ≤ 26.80 | 128 (64.6) | 17 (51.5) |
| > 26.80 | 70 (35.4) | 16 (48.5) |
| ESR (mm/h) | ||
| ≤ 15 | 119 (60.1) | 14 (42.4) |
| > 15 | 79 (39.9) | 19 (57.6) |
| cTnI (pg/mL) | ||
| ≤ 4,9025.8 | 161 (81.3) | 20 (60.6) |
| > 4,9025.8 | 37 (18.7) | 13 (39.4) |
| NT-proBNP (pg/mL) | ||
| ≤ 6,587 | 162 (81.8) | 18 (54.5) |
| > 6,587 | 36 (18.2) | 15 (45.5) |
| LVEF (%) | ||
| ≤ 37 | 40 (20.2) | 16 (48.5) |
| > 37 | 158 (79.8) | 17 (51.5) |
| Oxygen therapy | 124 (62.6) | 25 (75.8) |
| Mechanical ventilation | 12 (6.1) | 8 (24.2) |
| IABP | 18 (9.1) | 10 (30.3) |
| Temporary pacemaker | 4 (2) | 1 (3.0) |
| Aspirin | 173 (87.4) | 23 (69.7) |
| Indobufen | 17 (8.6) | 5 (15.2) |
| Clopidogrel | 70 (35.4) | 13 (39.4) |
| Ticagrelor | 124 (62.6) | 17 (51.5) |
| Statins | 190 (96) | 29 (87.9) |
| Ezetimibe | 26 (13.1) | 7 (21.2) |
| PCSK9i | 18 (9.1) | 3 (9.1) |
| β-blocker | 151 (76.3) | 22 (66.7) |
| ACEI/ARB/ARNI | 142 (71.7) | 16 (48.5) |
| Nitrates | 29 (14.6) | 4 (12.1) |
| CCB | 28 (14.1) | 2 (6.1) |
| Diuretic | 68 (34.3) | 17 (51.5) |
| Ivabradine | 21 (10.6) | 7 (21.2) |
| Trimetazidine | 29 (14.6) | 6 (18.2) |
| Coenzyme Q | 23 (11.6) | 4 (12.1) |
| Digitalis | 7 (3.5) | 2 (6.1) |
| Low molecular heparin | 55 (27.8) | 12 (36.4) |
| Number of diseased vessels (n (%)) | ||
| 1 | 175 (88.4) | 30 (90.9) |
| 2 | 23 (11.6) | 3 (9.1) |
| LAD | 119 (60.1) | 15 (45.5) |
| LCX | 28 (14.1) | 6 (18.2) |
| RCA | 74 (37.4) | 15 (45.5) |
| Total occlusion | 113 (57.1) | 22 (66.7) |
| TIMI, n (%) | ||
| 0 | 4 (2.0) | 4 (12.1) |
| 1 | 17 (8.6) | 4 (12.1) |
| 2 | 17 (8.6) | 2 (6.1) |
| 3 | 160 (80.8) | 23 (69.7) |
| Slow flow/no reflow | 38 (19.2) | 10 (30.3) |
| Thrombus aspiration | 15 (7.6) | 5 (15.2) |
| Tirofiban | 57 (28.8) | 13 (39.4) |
| Nicorandil | 16 (8.1) | 3 (9.1) |
| Nitroglycerin | 7 (3.5) | 2 (6.1) |
The detailed cutoff values for continuous variables are provided in Supplementary Table 1.
Nomogram construction based on clinical characteristics and hospitalization status
Four key factors emerged as independent predictors of poor outcomes in STEMI patients through both univariate and multivariable Cox proportional hazards regression analyses: (1) heart rates exceeding 83 beats per minute; (2) the need for mechanical ventilation; (3) absence of statin therapy; and (4) occurrence of intraoperative slow-flow or no-reflow phenomenon (all with P-values < 0.05; see Table 2). While left ventricular ejection fraction showed a significant correlation with EFS in the initial single-variable assessment, it failed to maintain its independent prognostic value when all variables were considered together, leading to its exclusion from the final predictive model.
Univariate and multivariable Cox proportional hazards regression analyses for composite adverse events
| Variable | Univariate HR | 95%CI | P value | Multivariate HR | 95%CI | P value |
| Male | 0.545 | 0.266-1.116 | 0.097 | |||
| Age > 55 years | 3.498 | 1.227-9.978 | 0.019 | 2.627 | 0.864-7.991 | 0.089 |
| Hypertension | 1.835 | 0.885-3.807 | 0.103 | |||
| Diabetes | 1.56 | 0.762-3.193 | 0.224 | |||
| Hyperlipidemia | 1.243 | 0.378-4.082 | 0.720 | |||
| Renal insufficiency | 0.847 | 0.202-3.548 | 0.821 | |||
| BMI > 20.48 kg/m2 | 0.534 | 0.231-1.235 | 0.142 | |||
| HR > 83 beats/min | 3.904 | 1.881-8.104 | 0.000 | 2.786 | 1.226-6.328 | 0.014 |
| SBP > 102 mmHg | 0.363 | 0.163-0.808 | 0.013 | |||
| DBP > 65 mmHg | 0.406 | 0.195-0.842 | 0.015 | |||
| WBC > 11.42× 109/L | 2.583 | 1.284-5.198 | 0.008 | |||
| Hb > 122 g/L | 0.311 | 0.153-0.63 | 0.001 | 0.53 | 0.238-1.181 | 0.120 |
| PLT > 340 × 109/L | 2.177 | 0.895-5.294 | 0.086 | |||
| ALT > 54 U/L | 3.086 | 1.482-6.425 | 0.003 | |||
| AST > 150 U/L | 3.48 | 1.671-7.249 | 0.001 | |||
| LDH > 543 U/L | 2.127 | 1.039-4.354 | 0.039 | |||
| Cr > 113 μmol/L | 2.884 | 1.43-5.813 | 0.003 | |||
| eGFR > 30.9 mL/min/1.73 m2 | 0.231 | 0.109-0.493 | 0.000 | |||
| TC > 2.72 mmol/L | 0.352 | 0.152-0.816 | 0.015 | 0.451 | 0.179-1.141 | 0.093 |
| TG > 2.10 mmol/L | 0.248 | 0.059-1.036 | 0.056 | |||
| HDL-C > 0.66 mmol/L | 1.276 | 0.63-2.584 | 0.499 | |||
| LDL-C > 1.45 mmol/L | 0.442 | 0.182-1.074 | 0.072 | |||
| K+ > 4.54 mmol/L | 3.114 | 1.52-6.377 | 0.002 | |||
| Glu > 10.03 mmol/L | 3.026 | 1.458-6.279 | 0.003 | |||
| Hs-CRP > 26.8 mg/L | 1.985 | 0.992-3.97 | 0.053 | |||
| ESR > 15 mm/h | 2.065 | 1.027-4.154 | 0.042 | |||
| CTnI > 4,9025.8 pg/mL | 3.005 | 1.468-6.152 | 0.003 | |||
| NT-proBNP > 6,587 pg/mL | 4.446 | 2.198-8.992 | 0.000 | |||
| LVEF > 37% | 0.204 | 0.102-0.409 | 0.000 | 0.502 | 0.204-1.233 | 0.133 |
| Oxygen therapy | 1.966 | 0.883-4.377 | 0.098 | |||
| Mechanical ventilation | 9.856 | 4.363-22.262 | 0.000 | 7.469 | 2.57-21.704 | 0.000 |
| IABP | 7.172 | 3.363-15.298 | 0.000 | |||
| Temporary pacemaker | 1.663 | 0.227-12.189 | 0.617 | |||
| Aspirin | 0.246 | 0.116-0.521 | 0.000 | 0.479 | 0.207-1.108 | 0.085 |
| Indobufen | 2.321 | 0.891-6.049 | 0.085 | |||
| Clopidogrel | 1.274 | 0.629-2.583 | 0.501 | |||
| Ticagrelor | 0.561 | 0.28-1.122 | 0.102 | |||
| Statins | 0.229 | 0.08-0.655 | 0.006 | 0.213 | 0.064-0.71 | 0.012 |
| Ezetimibe | 1.992 | 0.862-4.608 | 0.107 | |||
| PCSK9i | 1.045 | 0.318-3.431 | 0.942 | |||
| β-blocker | 0.546 | 0.263-1.132 | 0.104 | |||
| ACEI/ARB/ARNI | 0.344 | 0.172-0.689 | 0.003 | 0.51 | 0.239-1.091 | 0.083 |
| Nitrates | 0.783 | 0.274-2.234 | 0.647 | |||
| CCB | 0.367 | 0.088-1.536 | 0.170 | |||
| Diuretic | 2.266 | 1.132-4.539 | 0.021 | |||
| Ivabradine | 2.589 | 1.12-5.988 | 0.026 | |||
| Trimetazidine | 1.378 | 0.567-3.35 | 0.480 | |||
| Coenzyme Q | 1.063 | 0.373-3.032 | 0.909 | |||
| Digitalis | 1.87 | 0.447-7.826 | 0.391 | |||
| Low molecular heparin | 1.44 | 0.694-2.987 | 0.327 | 0.42 | 0.17-1.038 | 0.060 |
| LAD | 0.586 | 0.293-1.175 | 0.132 | |||
| LCX | 1.412 | 0.581-3.433 | 0.446 | |||
| RCA | 1.29 | 0.641-2.594 | 0.476 | |||
| Total occlusion | 1.544 | 0.744-3.203 | 0.244 | |||
| Slow flow or no reflow | 2.201 | 1.041-4.654 | 0.039 | 2.889 | 1.247-6.691 | 0.013 |
| Thrombus aspiration | 2.621 | 1.006-6.831 | 0.049 | 0.338 | 0.095-1.205 | 0.095 |
| Tirofiban | 1.635 | 0.799-3.347 | 0.179 | |||
| Nicorandil | 1.29 | 0.393-4.239 | 0.675 | |||
| Nitroglycerin | 2.58 | 0.614-10.847 | 0.196 |
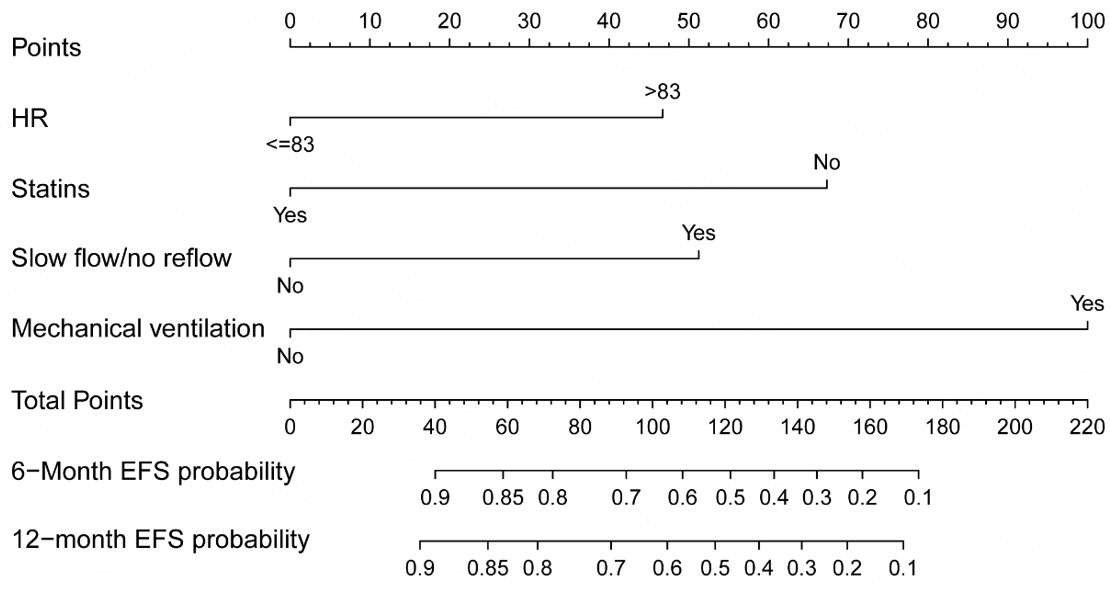
To create a predictive tool for 6-month and 12-month EFS in patients receiving delayed PCI, we integrated these four key factors into a nomogram (depicted in Figures 2 and 3). Within this visual aid, each predictor was given a points value that reflected its individual impact on risk assessment, allowing clinicians to tally up these scores to gauge the likelihood of EFS. The higher the cumulative score, the steeper the downhill trajectory toward potential complications.
Figure 2. Hazard ratios (HRs) and 95% confidence intervals (CIs) of the four independent prognostic factors for EFS.
Goodness of fit and discrimination of the nomogram
The nomogram’s discriminatory ability was assessed using the C-index. It showed robust discrimination, with a C-index of 0.782 (95% confidence interval [CI]: 0.690-0.874).
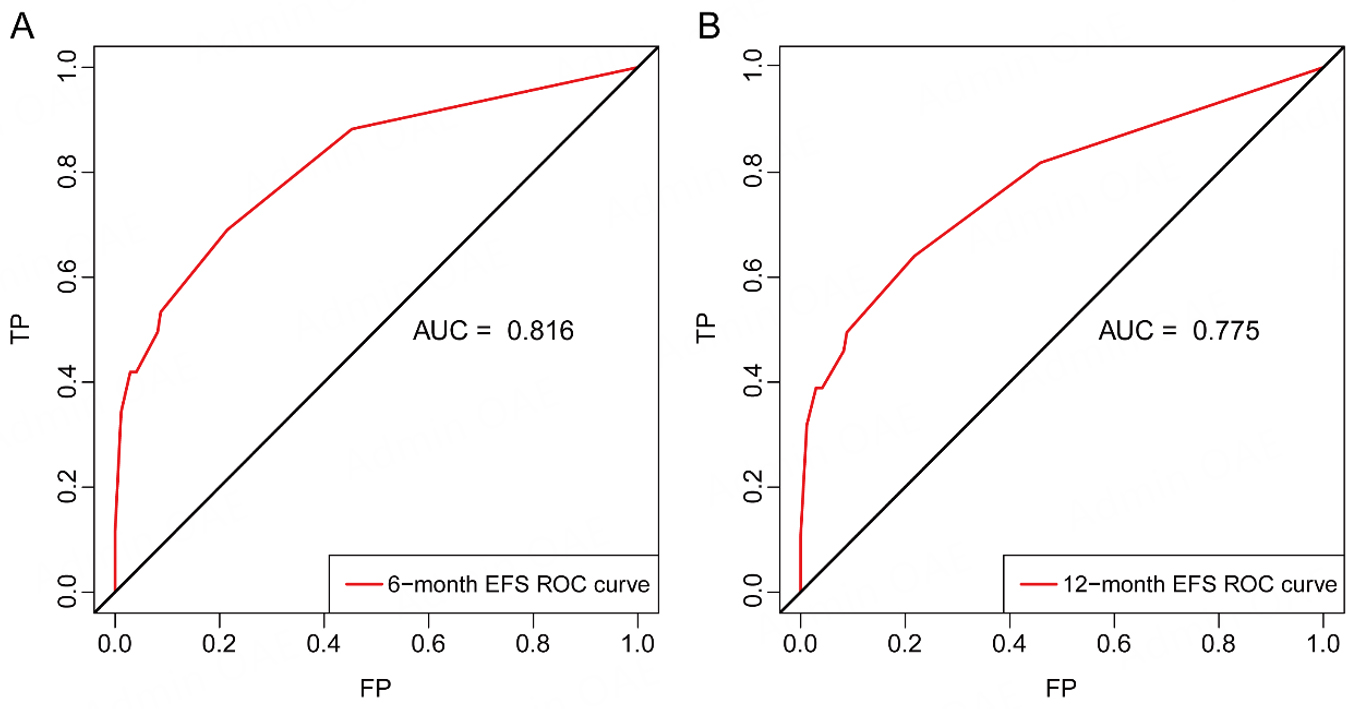
The ROC curve analysis demonstrating time dependence further validates the nomogram's predictive precision. The areas under the ROC curve (AUCs) were 0.816 for 6-month EFS and 0.775 for 12-month EFS, indicating good short- and intermediate-term prognostic performance [Figure 4].
Figure 4. Time-dependent ROC curves of the nomogram for predicting EFS. (A) ROC curve for 6-month EFS; (B) ROC curve for 12-month EFS.
The nomogram incorporates four independent prognostic factors: heart rate, mechanical ventilation, statin therapy, and intraoperative slow-flow or no-reflow phenomenon. The aggregate score results from adding up the points for each predictor and reflects the projected likelihood of EFS occurring at 6 and 12-month intervals.
Net benefit and predictive power of the nomogram
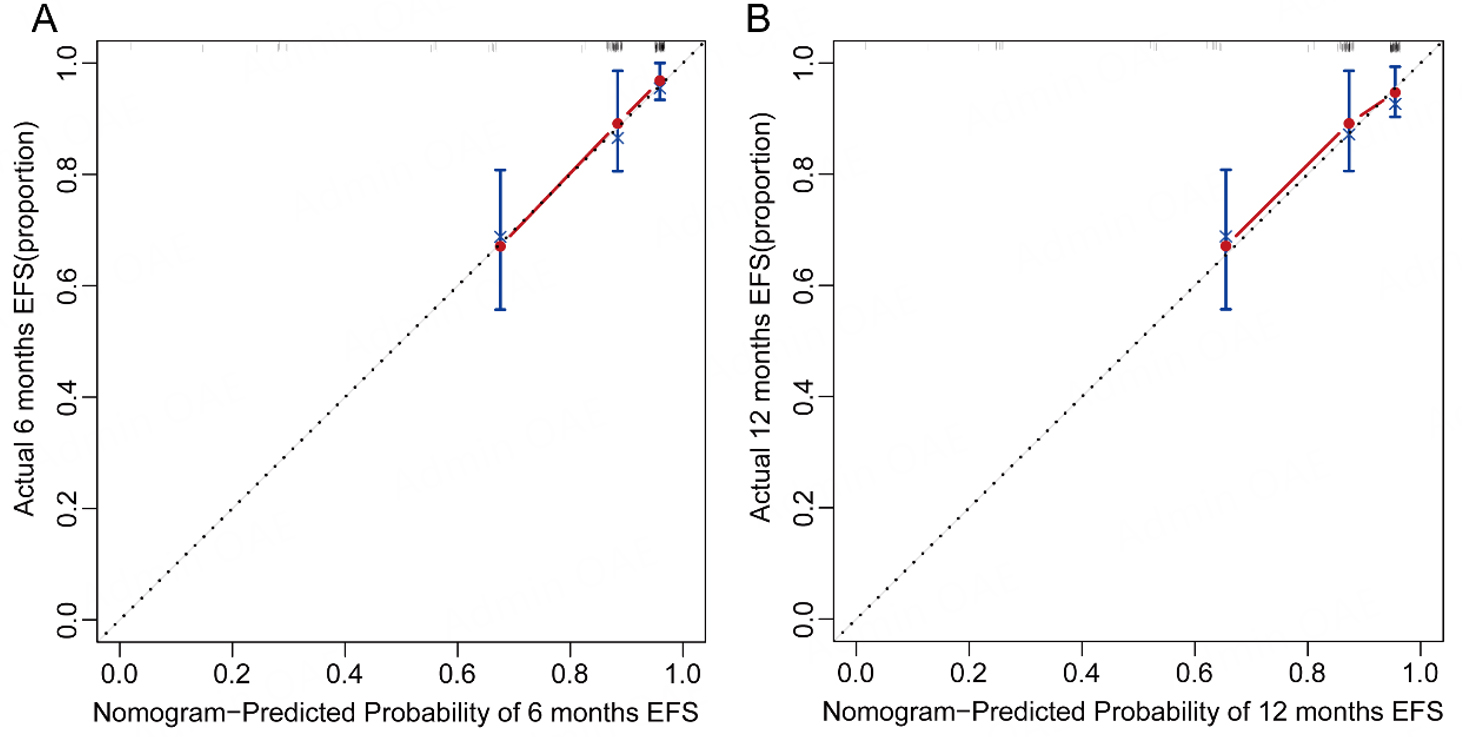
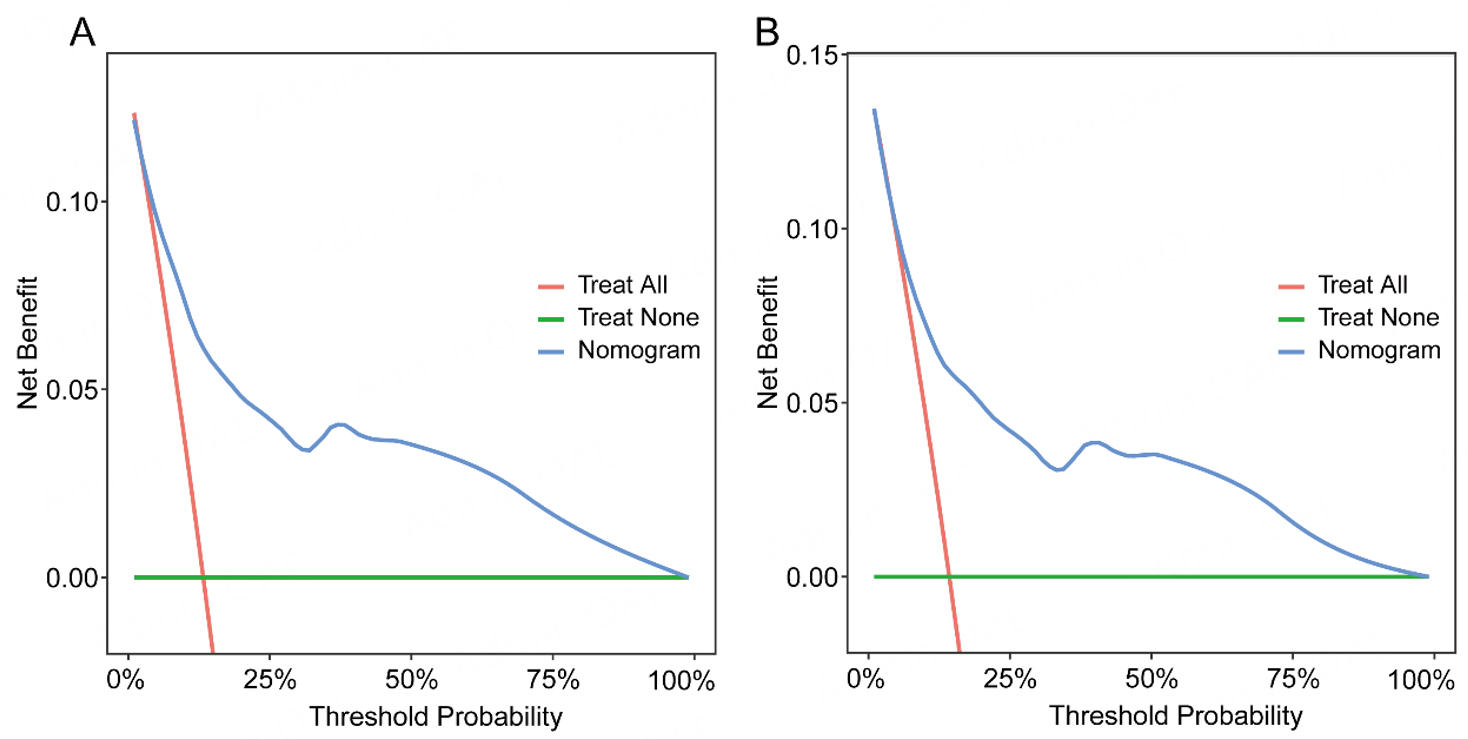
The calibration curve estimates the closeness between the risk predicted by the nomogram and the observed risk. As shown in Figure 5, the nomogram demonstrates good calibration for predicting 6-month and 12-month EFS. The nomogram's net advantage and forecasting prowess were evaluated through decision curve and calibration curve analyses[14]. In Figure 6, we can clearly see that the nomogram outshines the "treat-all" and "treat-none" approaches. This is thanks to its superior ability to refine risk categorization and support tailored post-discharge care, yielding a higher overall net benefit across a broad spectrum of threshold probabilities. In conclusion, these findings suggest that our nomogram exhibits strong performance in predicting EFS in STEMI patients.
Figure 5. Calibration curves of the nomogram for predicting 6-month (A) and 12-month (B) EFS. Dots represent deciles of predicted risk. The diagonal line indicates ideal agreement between predicted and observed outcomes, and the solid line represents the bootstrap-corrected calibration curve based on 1,000 bootstrap resamples.
Figure 6. Decision curve analysis (DCA) of the nomogram for predicting 6-month (A) and 12-month (B) EFS in patients with STEMI.
The decision curves clearly demonstrate the nomogram's overall clinical utility when evaluated against various threshold probability scenarios. In this visualization, the red line indicates the outcome of adopting a blanket treatment approach for every patient, while the green line depicts the results of implementing a wait-and-see strategy that withholds treatment from all individuals. The net advantage, which is determined from the ratio of true positives to false positives, is displayed on the vertical axis.
External validation
To validate our findings, we set up an independent external validation group using data from 599 STEMI patients who received delayed PCI treatment at the Second Hospital of Lanzhou University. These individuals were followed for a median of 20 months, during which we documented a total of 100 composite adverse outcomes. The participant recruitment process for this validation group is shown in
Risk stratification based on nomogram
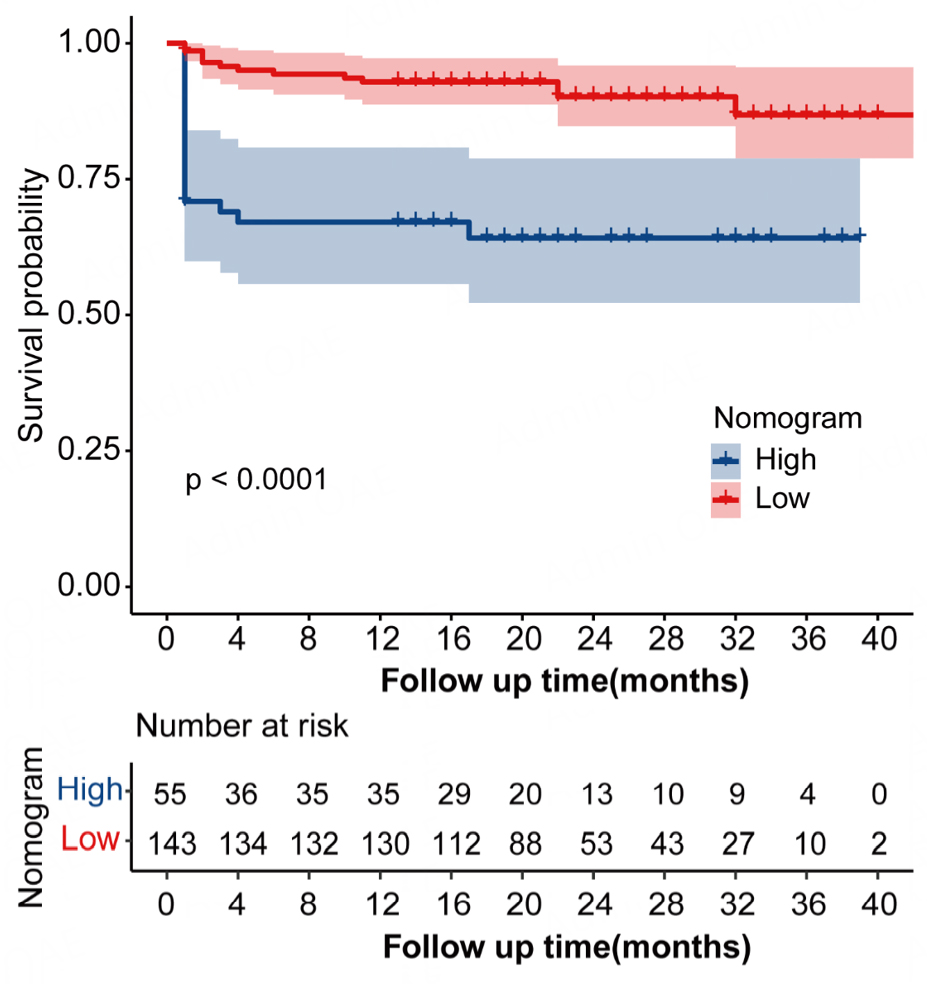
To evaluate how well the nomogram could stratify patients by risk level, we calculated each patient’s predicted probability of EFS. The median value of these predictions (62.11%) was used as the cutoff to divide patients into low-risk and high-risk categories.
Kaplan-Meier survival analysis showed clearly different outcomes between the two groups. The high-risk patients had significantly worse EFS than their low-risk counterparts (log-rank P < 0.0001; Figure 7).
DISCUSSION
Guideline-endorsed PCI stands as the preferred reperfusion method for STEMI patients[16-18]. However, due to delayed presentation, logistical constraints, and limited access to catheterization facilities, a subset of patients undergoes PCI more than 48 h after symptom onset. Clinical outcomes in this population remain heterogeneous and are influenced by multiple clinical and procedural factors.
In this study, we developed a predictive nomogram to estimate EFS among STEMI patients slated for delayed PCI. Through rigorous multivariate Cox regression analysis, we identified four readily available clinical markers—heart rate, statin administration, mechanical ventilation, and the occurrence of intraoperative slow flow or no-reflow—as key prognostic indicators, which were subsequently incorporated into our comprehensive predictive model. Although left ventricular ejection fraction, eGFR ≤ 30.9 mL/min/1.73 m2, and use of an intra-aortic balloon pump were associated with unfavorable outcomes in the initial analysis, they did not maintain prognostic independence in the multivariable analysis and were therefore excluded from the model to streamline its complexity.
The nomogram revealed effective discriminatory capabilities, boasting a C-index of 0.782, and exhibited accurate calibration in forecasting 6- and 12-month EFS. DCA further indicated meaningful clinical net benefit across a wide range of threshold probabilities. Importantly, the model effectively stratified patients into high- and low-risk groups, with significantly worse EFS observed in the high-risk group, supporting its potential utility for individualized risk assessment and post-discharge management.
The prognostic relevance of the variables included in the nomogram is clinically plausible. Intraoperative slow-flow or no-reflow reflects microvascular dysfunction and procedural complexity and has been consistently associated with adverse outcomes after PCI[19]. Statin therapy is known to exert cardioprotective and pleiotropic effects beyond lipid lowering and was associated with improved prognosis in our cohort[20-22]. Mechanical ventilation[22-24] and elevated heart rate[25-29] both reflect more severe hemodynamic and clinical instability and have been linked to worse outcomes in patients with acute coronary syndromes.
This study has several strengths. It focuses on a clinically relevant but under-investigated population of STEMI patients undergoing delayed PCI, incorporates routinely available clinical and procedural variables, and includes external validation in an independent cohort[30,31]. Together, these features enhance the robustness and generalizability of the proposed nomogram[32].
It is important to recognize a few key limitations. For one, the study's observational nature could potentially lead to residual bias. Secondly, the scope of outcomes was narrowed down to a combination of major adverse events, leaving out other critical clinical measures. Additionally, the patient pool was exclusively of Chinese descent, which might restrict the applicability to diverse populations. Lastly, to validate the effectiveness of nomogram-based care in enhancing clinical results, further confirmation in longitudinal studies is necessary.
To summarize, this externally validated nomogram may provide a useful and practical tool for forecasting long-term outcomes among STEMI patients undergoing delayed PCI. It could play a crucial role in tailoring risk assessment and guiding clinical decision-making for this particularly vulnerable patient group.
Conclusion
This study introduces a user-friendly nomogram leveraging standard clinical parameters and inpatient metrics that effectively predicts individualized Event-Free Survival in STEMI patients undergoing delayed Percutaneous Coronary Intervention. If further validated in large-scale, multicenter prospective studies, this nomogram may serve as a practical and accessible prognostic tool in clinical practice, supporting individualized risk stratification and informed post-discharge management and decision-making.
DECLARATIONS
Authors' contributions
Conceptualization and study design: Li Y, Liu X
Manuscript drafting: Li Y
Feasibility analysis: Li Y, Yue X, Sun R, Li H, Li Q
Literature search and data analysis: Li Y, Yue X, Wang F
Quality control, proofreading, and manuscript review: Li Y, Wang F, Zhou L, Zhao C
Final manuscript review and approval: Li Y, Liu X, Yue X, Sun R, Li H, Li Q, Zhou L, Zhao C, Wang F
Availability of data and materials
The data generated and/or analyzed in this study are not publicly available due to patient confidentiality and institutional policies. However, de-identified data may be available from the corresponding author upon reasonable request. Any data sharing requires approval from the relevant ethics committee. Once approved, access will be granted to qualified researchers for academic purposes under appropriate data governance regulations.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
The study protocol was approved by the Ethics Committees of Tongji Hospital (TJ-IRB202401016), affiliated with Tongji Medical College of Huazhong University of Science and Technology, and The Second Hospital of Lanzhou University (2024A-1270). Written informed consent was obtained from all participants or their legal representatives prior to enrollment. All procedures were conducted in accordance with the ethical standards of the Declaration of Helsinki. The dataset used in this study was fully de-identified, ensuring that individual patients could not be identified. The study complied with all applicable institutional and regulatory requirements.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
2. Grines CL, Mehta S. ST-segment elevation myocardial infarction management: great strides but still room for improvement. Eur Heart J. 2021;42:4550-2.
3. Stone GW, Kappetein AP, Sabik JF, et al. Five-year outcomes after PCI or CABG for left main coronary disease. N Engl J Med. 2019;381:1820-30.
4. Ono M, Serruys PW, Hara H, et al. 10-year follow-up after revascularization in elderly patients with complex coronary artery disease. J Am Coll Cardiol. 2021;77:2761-73.
5. Ye Z, Xu Y, Tang L, et al. Predicting long-term prognosis after percutaneous coronary intervention in patients with new onset ST-elevation myocardial infarction: development and external validation of a nomogram model. Cardiovasc Diabetol. 2023;22:87.
6. Yang XG, Feng JT, Wang F, et al. Development and validation of a prognostic nomogram for the overall survival of patients living with spinal metastases. J Neurooncol. 2019;145:167-76.
7. Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). Eur Heart J. 2019;40:237-69.
8. Virani SS, Newby LK, Arnold SV, et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2023;148:e9-119.
9. Ocak N, Yesilaras M, Kilicaslan B, Eyler Y, Mutlu İ, Kutlu M. Comparing TIMI, HEART, and GRACE risk scores to predict angiographic severity of coronary artery disease and 30-day major adverse cardiac events in emergency department patients with NSTEACS. Prehospital Disaster Med. 2023;38:740-8.
10. Demirkıran A, Aydın C, Yılmaz A, et al. A new marker of coronary collateral flow in patients presenting with acute myocardial infarction. Eur Rev Med Pharmacol Sci. 2023;27:6256-63.
11. Katayama Y, Taruya A, Kashiwagi M, et al. No-reflow phenomenon and in vivo cholesterol crystals combined with lipid core in acute myocardial infarction. Int J Cardiol Heart Vasc. 2022;38:100953.
12. Namazi M, Mahmoudi E, Safi M, et al. The no-reflow phenomenon: is it predictable by demographic factors and routine laboratory data? Acta Biomed. 2021;92:e2021297.
13. Perera D, Clayton T, O'Kane PD, et al. Percutaneous revascularization for ischemic left ventricular dysfunction. N Engl J Med. 2022;387:1351-60.
14. Tang L, Wu M, Xu Y, et al. Multimodal data-driven prognostic model for predicting new-onset ST-elevation myocardial infarction following emergency percutaneous coronary intervention. Inflamm Res. 2023;72:1799-809.
15. Zhong L, Min J, Zhang J, Hu B, Qian C. Risk prediction models for successful discontinuation in acute kidney injury undergoing continuous renal replacement therapy. iScience. 2024;27:110397.
16. Fazel R, Joseph TI, Sankardas MA, et al. Comparison of reperfusion strategies for ST-segment-elevation myocardial infarction: a multivariate network meta-analysis. J Am Heart Assoc. 2020;9:e015186.
17. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019;40:87-165.
18. Levine GN, Bates ER, Blankenship JC, et al. 2015 ACC/AHA/SCAI focused update on primary percutaneous coronary intervention for patients with ST-elevation myocardial infarction: an update of the 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention and the 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Circulation. 2016;133:1135-47.
19. Song J, Liu Y, Wang W, et al. A nomogram predicting 30-day mortality in patients undergoing percutaneous coronary intervention. Front Cardiovasc Med. 2022;9:897020.
20. Oesterle A, Laufs U, Liao JK. Pleiotropic effects of statins on the cardiovascular system. Circ Res. 2017;120:229-43.
21. Leoncini M, Toso A, Maioli M, Tropeano F, Bellandi F. Statin treatment before percutaneous cononary intervention. J Thorac Dis. 2013;5:335-42.
22. Zachariah P, Johnson CL, Halabi KC, et al. Epidemiology, clinical features, and disease severity in patients with coronavirus disease 2019 (COVID-19) in a children's hospital in New York City, New York. JAMA Pediatr. 2020;174:e202430.
23. Cockburn J, Kemp T, Ludman P, et al. Percutaneous coronary intervention in octogenarians: a risk scoring system to predict 30-day outcomes in the elderly. Catheter Cardiovasc Interv. 2021;98:1300-7.
24. Kleinbongard P, Gedik N, Witting P, Freedman B, Klöcker N, Heusch G. Pleiotropic, heart rate-independent cardioprotection by ivabradine. Br J Pharmacol. 2015;172:4380-90.
25. Halliday BP, Wassall R, Lota AS, et al. Withdrawal of pharmacological treatment for heart failure in patients with recovered dilated cardiomyopathy (TRED-HF): an open-label, pilot, randomised trial. Lancet. 2019;393:61-73.
26. Aro AL, Reinier K, Rusinaru C, et al. Electrical risk score beyond the left ventricular ejection fraction: prediction of sudden cardiac death in the Oregon Sudden Unexpected Death Study and the Atherosclerosis Risk in Communities Study. Eur Heart J. 2017;38:3017-25.
27. Parikh KS, Greiner MA, Suzuki T, et al. Resting heart rate and long-term outcomes among the African American population: insights from the jackson heart study. JAMA Cardiol. 2017;2:172-80.
28. O'Hare C, Kuh D, Hardy R. Association of early-life factors with life-course trajectories of resting heart rate: more than 6 decades of follow-up. JAMA Pediatr. 2018;172:e175525.
29. Aziz F, Malek S, Ibrahim KS, et al. Short- and long-term mortality prediction after an acute ST-elevation myocardial infarction (STEMI) in Asians: a machine learning approach. PLoS ONE. 2021;16:e0254894.
30. Gao J, Lu Y, Ashrafi N, Domingo I, Alaei K, Pishgar M. Prediction of sepsis mortality in ICU patients using machine learning methods. BMC Med Inform Decis Mak. 2024;24:228.
31. Tan J, Zhang Z, He Y, et al. Development and validation of a risk prediction model for osteoporosis in elderly patients with type 2 diabetes mellitus: a retrospective and multicenter study. BMC Geriatr. 2023;23:698.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].