Advanced wearable biosensing and wireless integration technologies for next-generation healthcare monitoring
Abstract
Wearable sensors play a crucial role in biomedical applications, enabling real-time and long-term monitoring of physiological and metabolic signals that are essential for disease prevention and personalized healthcare. With excellent stretchability, biocompatibility, and self-healing capability, these novel devices continuously track human biophysical signals such as motion, respiration, and blood pressure, as well as chemical biomarkers including electrolytes, glucose, and lactate in body fluids. Recent advances in material design, sensing mechanisms, and integration technologies have greatly fueled the enhanced capabilities of wearable monitoring systems and their implementation in next-generation healthcare platforms. This review summarizes the key material properties required for reliable long-term monitoring, the main detectable physiological biomarkers, and the latest progress and challenges in sensor integration and communication.
Keywords
INTRODUCTION
In the biomedical field, the precise, real-time, and non-invasive acquisition of human physiological signals serves as a cornerstone for disease prevention, health management, and clinical diagnosis and treatment[1-3]. However, the human skin - which is soft, deformable, naturally humid, and continuously subjected to complex stretching and deformation during daily activities - presents significant challenges for the stable and reliable acquisition of such signals. Conventional medical monitoring devices, such as Holter electrocardiographs and continuous glucose monitoring systems, though capable of delivering high-precision data, are constrained by their rigid structures, single functionalities, and frequently invasive designs[4-6]. These limitations not only reduce their suitability for long-term, skin-conformal monitoring in dynamic environments but also hinder the formation of intimate and stable interfaces with the skin, thereby inducing signal interference, motion artifacts, and discomfort during wear[7]. Such issues become particularly critical in scenarios requiring continuous and dynamic monitoring, including physical exercise, chronic disease management, sleep monitoring, and rehabilitation training. In recent years, propelled by the deep convergence of materials science, microelectronics, and biosensing technologies, wearable sensors have been progressively reshaping the paradigm of epidermal biomedical monitoring[8,9]. In 2013, Yeo et al. pioneered the direct printing of electronic devices onto human skin[10]. This seminal work triggered an explosive growth in subsequent research on wearable sensors. Notably, Rogers et al. published 18 papers in this field in 2025 (https://rogersgroup.northwestern.edu/). Meanwhile, Professor Zhenan Bao et al. have also made significant contributions to this area, publishing 8 related papers in 2025 (https://baogroup.stanford.edu/people/zhenan-bao). Owing to their excellent flexibility, stretchability, and biocompatibility, wearable sensors can form conformal and stable interfaces with the skin, enabling high-fidelity signal acquisition even under conditions of motion, perspiration, or micro-strain. This technological progress has greatly facilitated the development of long-term, dynamic, and unobtrusive health monitoring systems.
The proliferation of traditional rigid wearable electronics has led to significant advancements, yet several challenges do persist. The pronounced Young’s modulus mismatch - where traditional electronic materials such as silicon can be as stiff as ~100 GPa, compared to the kPa-MPa range of human skin, which constitutes a fundamental challenge[11]. This difference prevents traditional rigid wearable devices from establishing and maintaining conformal contact with the biological interface. The resulting poor adhesion and micro-movements generate substantial noise, leading to low signal-to-noise ratios (SNRs) and consequently compromising diagnostic efficiency and accuracy. Additionally, the wireless communication modules of conventional devices often rely on centralized cloud computing resources for data processing and analysis. However, this approach presents several challenges, including increased latency, bandwidth limitations, and concerns about data privacy[12,13]. Therefore, the exploration of novel flexible materials and the development of self-powered and wireless data transmission technologies are of great significance for advancing the next generation of wearable sensing systems.
This review provides a comprehensive summary of recent advances in wearable sensors for health monitoring [Figure 1], encompassing key material properties, sensor types, and wireless communication applications. Firstly, the material requirements for high-performance sensors are discussed, including excellent stretchability, self-healing capability, and biocompatibility, which are essential for ensuring device stability and comfort under complex human motion conditions. Subsequently, the major sensor types employed for monitoring physiological signals and metabolites are reviewed: temperature sensors enable precise monitoring of core body temperature; capacitive, piezoresistive, and piezoelectric pressure sensors facilitate real-time detection of physiological signals such as blood pressure, pulse, and gait; in addition, sweat, tear, and saliva sensors allow the analysis of metabolites in body fluids, providing insights into individual health status. Finally, recent progress in wireless data transmission and communication technologies is also discussed, offering critical support for the development of multimodal, real-time, and personalized smart health monitoring systems.
REQUIRED MATERIAL PROPERTIES FOR WEARABLE SENSORS
Given the unique and dynamic nature of human skin, wearable sensors must incorporate essential properties such as flexibility, stretchability, self-healing capability, biocompatibility, and high sensing performance to ensure seamless and conformal adaptation[1,14,15]. The practical application environments for wearable sensors are often complex, necessitating the integration of multiple key characteristics for optimal adaptability. These include self-powered operation, real-time monitoring, breathability, water resistance, and strong adhesion. This section examines recent advancements in the materials and fabrication technologies that underpin these features[16-18].
Flexibility
Flexibility is one of the most fundamental and critical functionalities of wearable sensors. Devices incorporating flexible substrates can conform more stably to human skin, enabling intimate and reliable interfacial contact[19,20]. Such stable coupling effectively reduces signal attenuation and noise caused by interfacial gaps or relative slippage, thereby significantly improving sensing accuracy, repeatability, and long-term monitoring reliability. Consequently, flexibility is regarded as a prerequisite for achieving high-quality signal acquisition and practical wearable applications[21].
Polyethylene terephthalate (PET) is a heat-resistant polyester film with a thermal tolerance of up to 200 °C[22]. It also exhibits excellent optical transparency, chemical resistance, mechanical properties, and overall stability, making it a preferred substrate material for flexible and transparent conductive films[23]. Zhao et al. proposed a PET-based flexible interdigitated electrode (IDE) pressure sensor fabricated by spraying silver nanoparticles (AgNPs)/carbon nanotubes (CNTs) onto a nonwoven fabric[24]. PET serves as the substrate for the flexible IDEs, providing a stable and flexible supporting structure for the sensor. The mechanical stability of PET, combined with the high flexibility of CNTs and the excellent conductivity of AgNPs, ensures both the structural integrity of the sensor under repeated deformation (up to 6,000 cycles) and its superior electrical performance. Similarly, polyimide (PI), as a widely used high–glass-transition temperature (high-Tg) engineering material, exhibits superior high-temperature resistance and structural stability compared with other polymers; therefore, it can effectively serve as a flexible substrate in high-temperature environments[25,26]. Liu et al. employed a
Naturally derived materials, such as cellulose nanofibers (CNFs) and silk fibroin (SF), are also considered ideal substrate candidates for wearable sensors due to their favorable mechanical properties and biocompatibility[27]. CNFs are considered ideal substrate materials due to their biodegradability, excellent mechanical properties, flexibility, and environmental sustainability[28,29]. Huang et al. employed CNFs as the core substrate and proposed a novel piezoresistive sensor based on an indium tin oxide nanoparticle (ITO NP)/CNF composite[29]. Hydrogen-bonding interactions between the CNF substrate and ITO NP tightly anchored the conductive fillers onto the fiber surfaces, preventing detachment even under repeated deformation. Combined with the flexible design of paper-based IDEs, the sensor maintained stable performance over more than 5,000 loading cycles. Li et al. developed a piezoresistive sensor based on a SF/sodium alginate (SA) bilayer sponge incorporated with aniline tetramer (AT)[27]. Naturally biodegradable SF and SA were used as the substrate materials, while in situ polymerization enabled strong interfacial bonding between the AT and the substrate. Even after 10 compression cycles with strains ranging from 10% to 50%, the maximum stress could recover to more than 86% of its initial value, demonstrating good flexibility.
Stretchability
The pursuit of conformal skin-device interfaces for healthcare monitoring necessitates sensors with mechanical compatibility. This is crucial as human skin is a dynamic curvilinear surface with a Young’s modulus of approximately 0.1-2 MPa and can undergo strains of 30%-70%[30]. Consequently, wearable sensors must match this soft and stretchable nature to maintain reliable contact[31]. Resistance to mechanical stress is a key requirement for wearable healthcare sensors during use. Performance degradation under dynamic deformation remains a major challenge, and prior studies have identified several underlying mechanisms responsible for this phenomenon. For example, conductive hydrogels often undergo energy dissipation during deformation, a behavior commonly referred to as hysteresis[32]. When a hydrogel exhibits pronounced hysteresis, the cumulative energy loss over thousands of deformation cycles can eventually lead to structural failure. The two-dimensional honeycomb-like carbon-carbon network in graphene lacks sufficient energy-dissipation pathways to accommodate external strain[33]. As a result, graphene films are prone to cracking at strains as low as 5%. Stretchability is therefore paramount, enabling extensive elongation and bending without permanent deformation or failure while maintaining the body’s natural range of motion. This capability is typically achieved through one of two core strategies: geometric engineering applied to rigid materials or the use of intrinsically stretchable soft materials[34-36].
One effective strategy for achieving stretchability is through the design of appropriate geometrical structures. Geometric design strategies such as metal serpentine structures[37,38], kirigami structures[39,40], and microcrack structures[41,42] have been demonstrated to effectively transform intrinsically rigid materials into stretchable ones while maintaining outstanding electrical performance. The serpentine and helical structures are representative types of island-bridge configurations, consisting of conductive interconnects (bridges) and functional components (islands) positioned at fixed locations. Yan et al. proposed a hierarchical serpentine-spiral composite structure that maintains stable performance under multidirectional stretching[43]. This 3D helical configuration enhanced transverse stretchability by increasing the helix pitch, while the serpentine units enable longitudinal and vertical deformation through buckling [Figure 2A]. Consequently, this hybrid structure achieved excellent stretchability (≥ 200%) along the x, y, and z-axes, imparting high flexibility to otherwise rigid materials. Liu et al. reported that introducing patterned incisions into a composite nanofiber framework hydrogel film via laser cutting significantly enhanced its stretchability[44]. As shown in Figure 2B, the kirigami-inspired structure accommodated macroscopic deformation through mesoscale bending and twisting, achieving an elongation of up to 130% while maintaining stable electrical resistance. Moreover, the film exhibited negligible performance degradation after 5,000 stretching cycles at 60% strain, demonstrating excellent mechanical durability and electrical stability. These strategies can effectively convert rigid materials into stretchable ones while preserving electrical properties. Under mechanical stress, these stretchable microstructures frequently form out-of-plane patterns such as wrinkling, necessitating the strategic development of packaging schemes to mitigate interfacial failure and maintain electrical functionality.
Figure 2. Stretchability enhancement strategies. (A) Bioinspired design of hierarchical 3D serpentine-helix combination. Reproduced with permission[43]. Copyright 2023, Wiley-VCH; (B) Kirigami membrane and its stability. Reproduced with permission[44]. Copyright 2022, Wiley-VCH; (C) Chemical structures of IDTBT and n-NIPS and Photocrosslinking process. Reproduced with permission[54]. Copyright 2023, Wiley-VCH; (D) Schematic of mechanism of stretchability and elasticity. Reproduced with permission[59]. Copyright 2024, Wiley-VCH; (E) Preparation diagram of the composite fiber and EDS images of it. Reproduced with permission[73]. Copyright 2023, American Chemical Society. IDTBT: Indacenodithiophene-benzothiadiazole; n-INPS: nitrene-induced photocrosslinkers, n denotes the number of ethylene glycol repeating units in the crosslinker molecule; EDS: energy dispersive spectroscopy; PAAm: poly(acrylamide); PEGDA: poly(ethylene glycol) diacrylate; PEDOT: poly(3,4-ethylenedioxythiophene); PSS: poly(styrenesulfonate); IPA: isopropyl alcohol; DMSO: dimethyl sulfoxide; PU: polyurethane.
Another strategy for achieving stretchability is the utilization of intrinsically stretchable materials[45,46]. Stretchable substrates fabricated from polymers [polydimethylsiloxane (PDMS)[47-49], polyurethane (PU)[50,51], styrene–ethylene–butylene–styrene (SEBS)[52,53], etc.] offer limited extensibility. To surpass these limitations, researchers use reversible dynamic covalent bonds to build reconfigurable networks, enabling high toughness and recoverable ultrahigh stretchability. For example, researchers synthesized indacenodithiophene-benzothiadiazole (IDTBT)/nitrene-induced photocrosslinker (1-NIPS) thin films via azide-based photocrosslinking [Figure 2C][54]. The variation of the dichroic ratio as a function of tensile strain demonstrates how the azide-based crosslinker regulates the molecular chain dynamics of the conjugated polymer film during mechanical deformation. The crosslinked film exhibited pronounced variations in tensile stress, demonstrating its exceptional stretchability. These films exhibited a uniform morphology that enabled homogeneous stress distribution. Under 80% strain, the crosslinked films retained 96% of their initial hole mobility, and their electrical performance almost fully recovered upon strain release[54]. Weak hydrogen bonds and electrostatic interactions within polymers can dissipate strain-induced energy via reversible breakage and recombination. Hydrogels[55-57], owing to their excellent flexibility and biocompatibility, are ideal materials for wearable sensors. Traditional hydrogel-based wearable sensors typically employ thicker hydrogels, which limit their sensitivity. Pan et al. designed a highly stretchable, fast-response humidity sensor utilizing a unique ultra-thin hydrogel-carbon nanocomposite material[58]. This sensor employs a micro-nano hierarchical structure and a two-stage solvent-free method, ultimately resulting in the preparation of a nanocomposite sensor. Even under extreme bending at 180°, the sensor retains impressive sensing performance, with a sensitivity of 32.94 nA/% [nA%: the change in output current (in nA) per 1% variation in the measured quantity] and a response/recovery time of 25.2/0.4 s. In addition, incorporating dynamic noncovalent crosslinks between flexible polymer chains [Figure 2D] enables further enhancement of the polymer’s tensile properties. The right Figure shows the Young’s modulus of the printed hydrogel after 1, 3, 7, and 10 days. The silk fiber-reinforced poly(acrylamide)/poly(acrylic acid) (SF-PAAm/PAAc) hydrogel developed by Wu et al. exhibited excellent mechanical stability, maintaining consistent performance during 80 successive stretch-release cycles under 100% strain, with a gauge factor (GF) of ~1.29[59]. As the acrylic acid (AAc) content increases from 0% to 30% by weight relative to acrylamide (AAm), the Young’s modulus of the hydrogel increases from 40 to 80 kPa, while the maximum elongation decreases from 1,200% to ~680%.
Conductive composite materials represent an effective solution, enabling the combination of high stretchability and strain-resistant conductivity. Conductive fillers - including metal nanoparticles[60,61], nanowires[11,62], nanosheets[63,64], and CNTs[65,66], integrated with stretchable elastomers to form such conductive composites[67,68]. Jiang et al. engineered a Prussian blue (PB)–coated silver nanowires (AgNWs) core–shell architecture, which facilitates rapid electron transport along the low-resistance AgNWs pathways; the resulting sensor achieved a uric-acid sensitivity of 50.6 μA mM-1·cm-2[69]. Lin et al. combined silk sericin with CNTs and fabricated carboxylated styrene–butadiene rubber (XSBR)/sericin non-covalently modified carbon nanotubes (SSCNT) sensors via a latex film-forming process[70]. The resulting sensors exhibit high ductility with a maximum tensile strain of 217%, an ultimate tensile strength of 12.58 MPa, and an enhanced toughness of 6.23 MJ·m-3. Moreover, the devices can sustain a 2 kg load and withstand stretching to 400% without rupture.
Furthermore, stretchable fibers composed of conductive filler-polymer composites have been successfully engineered for sensing applications[71,72]. The PU/graphene-encapsulated poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS) fiber developed by Fan et al. demonstrated excellent tensile performance [Figure 2E], which surpassed that of pure PEDOT:PSS fibers (12.84%) and exhibited a tensile strain 2.17 times higher than that of cotton yarn[73]. This fiber exhibited minimal resistance variation (only 2% increase near fracture), demonstrating exceptional electromechanical stability. This reliability enabled its broad application in long-term body temperature monitoring for intelligent healthcare and athlete management, as well as in specialized scenarios such as underwater or low-temperature operations. Despite progress in breathable textile-based multimodal sensors, achieving comprehensive perception (strain, pressure, temperature, humidity) with good permeability, biocompatibility, comfort, and a stable skin-electrode interface under wet conditions remains challenging. Zheng et al. developed a skin-like multimodal device (SMED) by screen-printing MXene/PEDOT:PSS onto an ultra-thin thermoplastic polyurethane (TPU) nanofiber pad[74]. The SMED exhibits high transparency, strong wet adhesion, and stable skin-electrode interface. The electrospun TPU pad (1-2 μm thick) maintains excellent stretchability with no visible damage even when stretched by 50%. The above strategies for enhancing the stretchability of wearable sensors are summarized in Table 1.
Summary of typical strategies for enhancing stretchability and their advantages
| Strategies | Structures | Advantages | Ref. |
| Geometrical structures | Metal serpentine | Real-time strain visualization, quantitative–qualitative strain analysis | Han et al.[37] |
| Kirigami | Geometrically programmable, highly stretchable | Zhang et al.[39] | |
| Microcrack | Tensile durability and cyclic stability | Lee et al.[41] | |
| Intrinsically stretchable materials | IDTBT/1-NIPS | Excellent electrical stability | Kim et al.[54] |
| Silk fiber-reinforced poly | Excellent biocompatibility, high precision fabrication | Wu et al.[59] | |
| Conductive composite materials | Ag nanowires | High electrical conductivity and high pressure sensitivity | Li et al.[11] |
| CNTs | Enhanced tensile strength and electrical stability | Alam et al.[66] | |
| Stretchable fibers | High strain accuracy, rapid response | Fan et al.[73] |
Self-healing capability
The integration of self-healing capabilities into wearable sensors, enabling autonomic repair after mechanical damage, is highly desirable for improving device reliability, safety, and fatigue resistance[75-77]. Wearable sensors with such functionality can achieve repeated self-repair after mechanical damage[78-80]. Therefore, service life is expected to extend and maintenance costs are reduced. The ideal self-healing capability for wearable sensors is defined by autonomy - restoring both mechanical and electrical properties promptly and completely without external energy input.
A prevalent strategy to impart self-healing capability to materials involves the use of composite systems containing either conductive fillers or microcapsules loaded with healing agents[66]. Damage repair and conductivity restoration can occur through the release of conductive fillers from ruptured capsules or via the redistribution of inherent conductive components within the matrix. For instance, Li et al. developed an electrically self-healing conductor based on a styrene-isoprene-styrene (SIS) elastomer [Figure 3A][81]. This conductor adopted a bilayer structure comprising an electroless copper-plated upper layer and a lower layer embedded with liquid metal microcapsules. Under mechanical deformation, microcracks form in the copper layer, and the associated stress punctures the underlying microcapsules. The released liquid metal then fills the cracks, re-establishing the electrical pathway and restoring conductivity. Dynamic conductive hydrogels represent another class of intrinsic self-healing materials, in which repair capability arises from reversible interactions such as metal coordination, hydrogen bonding, and ionic chelation[82,83]. Wei et al. fabricated polyacrylic acid–amorphous calcium carbonate–liquid metal–tannic acid [PAA-ACC-LM@TA (PATL)] hydrogels by incorporating tannic acid-coated gallium-indium liquid metal (EGaIn@TA) into PAA-ACC networks [Figure 3B][84]. Driven by a dynamic network of reversible interactions - including ionic chelation (PAA and Ca2+), hydrogen bonding (PAA and TA), and metal coordination (ACC and EGaIn@TA) - the hydrogel demonstrates rapid (< 1 s) and efficient (88.8 %) self-healing at room temperature. These mechanisms collectively enable macroscopic network reconstruction, microscopic crack healing, and conductive pathway restoration[85-87].
Figure 3. Self-healing strategies. (A) Schematic illustration of the ultrasensitive self-healing mechanism. Reproduced with permission[81]. Copyright 2023, Proceedings of the National Academy of Sciences; (B) Illustration of PAA-ACC-LM@TA hierarchical hydrogel and the corresponding self-healing mechanism. Reproduced with permission[84]. Copyright 2025, American Chemical Society; (C) Self-healing mechanism of PLOD and its electrical conductivity. Reproduced with permission[89] Copyright 2025, Elsevier B.V. PAA-ACC-LM@TA: Polyacrylic acid–amorphous calcium carbonate–liquid metal–tannic acid; PLOD: the exclusive abbreviation developed in-house for the nanocomposite hydrogel independently synthesized in this study, where P stands for PEDOT:PSS, L stands for LAP, O stands for OEGMA, D stands for DEGMA.
To achieve simultaneous mechanical robustness and electrical recoverability, researchers have integrated the reversible effects of dynamic bonds with conductive fillers[88]. Zeng et al. developed a novel self-healing composite conductive hydrogel strategy via synergizing multiple dynamic noncovalent bonds (hydrogen bonding, electrostatic interactions, π-π stacking) with conductive fillers[89]. They incorporated these bonds into an oligo(ethylene glycol) (OEG) copolymer matrix [from oligo(ethylene glycol) methyl ether methacrylate (OEGMA)/di(ethylene glycol) methyl ether methacrylate (DEGMA) copolymerization] and doped Laponite® (LAP)/PEDOT:PSS, forming a nanocomposite hydrogel network [PLOD (the exclusive abbreviation developed in-house for the nanocomposite hydrogel independently synthesized in this study, where P stands for PEDOT:PSS, L stands for LAP, O stands for OEGMA, D stands for DEGMA)] capable of both structural self-healing and conductive recovery [Figure 3C]. As shown in Figure 3C (right), when PLOD was cut, the resistance instantly increased to infinity. After self-healing, the resistance of PLOD could recover its initial value within 1.8 s.
Biocompatibility and comfort
The direct and prolonged skin contact of wearable sensors necessitates excellent biocompatibility to prevent adverse immune responses and ensure user comfort without restricting daily activities[90,91]. Therefore, wearable sensors require carefully selected materials to ensure functionality and biocompatibility during direct or indirect contact with biological tissues, thereby minimizing potential tissue damage[92]. For clinically relevant wearable devices, systematic clinical evaluation is necessary to monitor possible adverse or rejection responses and to comprehensively assess their safety. Regarding substrate materials for wearable sensors, the electronic skin concept proposed by Jiang et al. is based on elastomeric polymer matrices, which not only provide high flexibility and conformal contact with the skin but also effectively mimic the intrinsic elasticity and softness of human skin[93]. Commonly used elastomers include PDMS, TPU[94], styrene-based elastomers (e.g., Ecoflex)[95], and SEBS[96]. These materials exhibit excellent flexibility and a certain degree of breathability, making them well-suited for wearable medical devices. In addition, polymers such as PET, PI, polyvinyl alcohol (PVA), polyetherimide (PEI), and polyethylene naphthalate (PEN) are also widely employed. Ultrathin flexible substrates fabricated from these materials can achieve high sensitivity and favorable mechanical performance under deformation, pressure, or bending, while maintaining good biocompatibility. For example, Zhang et al. developed a flexible piezoresistive sensor based on a Ti3C2Tx [possesses a 2D layered structure consisting of three Ti atomic layers interleaved with two C atomic layers in a periodic manner, where Tx represents surface terminating groups whose type and average number (x) vary dynamically with the preparation conditions (e.g., etchant and temperature)] MXene/PET textile for a wearable pulse monitoring system[97]. This system demonstrated high accuracy in the classification and recognition of pulse signals from patients with cardiovascular diseases.
Electrodes for flexible wearable sensors are typically composed of a variety of materials, including solid metals, carbon-based materials (e.g., carbon black, graphene, and CNTs), conductive polymers, ionic liquids, and their hybrids. Solid metals are relatively rigid and prone to oxidation and mechanical fracture during long-term wear, which may induce skin irritation. In contrast, carbon-based materials, when integrated with flexible substrates, provide metal-like electrical conductivity while offering improved flexibility and biocompatibility. To enhance the breathability of wearable sensors, Gao et al. developed integrated dual-mode temperature and pressure sensors by decorating elastic polydopamine-modified PDMS (PDA@PDMS) foam with thermoelectric PEDOT:PSS/CNT composites[98]. The resulting electronic skin arrays enabled accurate monitoring of external temperature and pressure variations. Moreover, owing to their intrinsic biocompatibility, skin conformability, and breathability, conductive hydrogels with high electrical conductivity and mechanical stability have attracted increasing attention. Dang et al. developed multifunctional carboxymethyl cellulose (CMC)–based conductive hydrogels, whose properties were optimized by tuning the CMC content[99]. Cytotoxicity evaluations using Chinese hamster lung (CHL) cells showed no significant reduction in cell viability for eluates from PAAm–PAAc hydrogels without CMC or those containing 17% CMC, with cell viability consistently remaining ≥ 97%, demonstrating excellent biocompatibility.
Encapsulation of flexible wearable sensors enhances waterproofing, corrosion resistance, component durability, device lifetime, and overall sensing performance[92,100,101]. Medical tapes, made from non-toxic and porous materials, are widely used due to their skin compatibility and comfort when integrated with flexible electronics. Gai et al. developed a self-powered wearable sweat analysis system (SWSAS) using 3M double-sided medical tape as the adhesive layer, enabling breathable, skin-conformal attachment under sweating conditions while minimizing skin irritation[102]. A waterproof medical tape serves as the isolation layer to prevent direct electrode–skin contact and suppress interference from sweat impurities, ensuring stable signal acquisition. Chung et al. developed a novel skin-interface biosensor tailored for clinical monitoring in Neonatal Intensive Care Unit (NICU) and Pediatric Intensive Care Unit (PICU) settings, offering an advanced wireless solution for critically ill infants[103]. The device features a flexible silicone housing and a ~300 µm thick, ~4 kPa modulus silicone liner as its encapsulation, enhancing conformability to infants’ complex anatomy. Its gentle interface design ensures excellent biocompatibility with delicate neonatal skin, minimizing the risk of skin injury associated with conventional monitoring devices.
STRUCTURE OR DEVICE ENGINEERING FOR WEARABLE SENSORS
Wearable sensors are now ubiquitous in healthcare, allowing for the quantitative tracking of physiological signals from human motion and bodily fluids[104]. This is driven by research focused on optimizing device architecture and performance parameters - notably accuracy and sensitivity - for monitoring human biological indicators[105-107]. Functionally, wearable physical sensors are attached to the skin for vital signs collection such as arterial pulse, blood pressure, electrocardiography (ECG), and body temperature[104,108]. In parallel, chemical information extracted from bodily fluid components (electrolytes, proteins, metabolites, etc.) reflects health status and offers aid in the early disease diagnosis[17,109,110]. Table 2 summarizes the detectable analytes and potential related diseases. In this section, we summarize recent advances in wearable sensors for physiological signal monitoring.
Analytes and their corresponding associated diseases
| Analyte/parameter | Normal parameters | Reflecting human health status and diagnostic application | Ref. |
| Core body temperature | 36.0-37.0 °C | Sepsis diagnosis | Choi et al.[111] |
| Na+ | 135-145 mmol/L | Cystic fibrosis | Declercq et al.[112] |
| K+ | 3.5-5.5 mmol/L | Hypo or hyperkalemia | Calvo-López et al.[113] |
| Cl- | 96-106 mmol/L | Lung disease | Krishnan et al.[114] |
| Glucose | 3.9-6.1 mmol/L | Glycosuria | Zhou et al.[115] |
| Lactate | 0.5-1.6 mmol/L | Acidosis | Wu et al.[116] |
| Cholesterol | 0.28 ± 0.03 mM | Cardiovascular diseases | Song et al.[117] |
Wearable temperature sensors
As a critical physiological function, skin temperature sensing provides vital information for disease diagnosis, health assessment, and safety monitoring[118,119]. While a normal reading does not preclude illness, deviations from the stable core range of 37-37.5 °C are strong indicators of pathology. Consequently, wearable temperature sensors must be able to detect subtle, real-time fluctuations with high precision, typically demanding a sensitivity of ±0.1 °C within 37-39 °C and ±0.2 °C beyond this range[115,120].
Temperature sensing mechanisms capitalize on the strong temperature dependence of physical parameters such as electrical resistance, volumetric expansion, and optical properties[121,122]. A quantitative measurement model translates these dependencies into a readable signal. Common devices include resistance temperature detectors (RTDs), capacitive sensors, and thermistors, each based on a unique principle. RTDs, for example, function via the positive resistance-temperature relationship in conductive materials, where increased thermal lattice vibrations impede electron flow[70,123]. A notable application was the chipless, neuromorphic system by Choi et al.[111] [Figure 4A], which employed MXene for continuous core body temperature monitoring. Electrical conductivity through the MXene pathways increased markedly with temperature due to enhanced carrier mobility, resulting in a measurable resistance drop. The MXene line resistance decreased linearly from 8.8 to 4.4 kΩ as the temperature increased from 20 to 50 °C, and this resistance variation produced a corresponding voltage output ranging from 3.1 to 3.9 V [Figure 4A]. Notably, this work integrates sensing technology, signal processing units, and neural networks into a compact and wearable system tailored for medical applications such as sepsis diagnosis and monitoring. In wearable healthcare scenarios, the Hardware Neural Network (HWNN) demonstrates distinct technological advantages. Its fully hardware-implemented architecture eliminates reliance on external internet resources, with all data processing performed locally on the device. This not only significantly reduces power consumption (as low as 37.6 μW per case) but also effectively mitigates the risk of privacy leakage associated with the transmission of sensitive medical data.
Figure 4. Different types of temperature sensors and their performance. (A) Wearable sensor integrated with a RTD and its performance. Reproduced with permission[111]. Copyright 2025, Springer Nature; (B) Capacitive temperature sensors and their performance. Reproduced with permission[119]. Copyright 2022, American Association for the Advancement of Science; (C) Schematic illustration of the sensor and the resistance–temperature relationship of the NTC sensor. Reproduced with permission[129]. Copyright 2025, American Chemical Society. RTD: Resistance temperature detector; NTC: negative temperature coefficient; MCU: microcontroller unit.
The operation of capacitive temperature sensors relies on thermal changes affecting the dielectric constant or device geometry, thereby altering capacitance[124]. A notable advancement was the 8 × 8 capacitive temperature sensor array-based thermal patch [Figure 4B] by Kang et al.[119]. Its sensing layer exhibited a linear dielectric response over 25-40 °C [the coefficient of determination (R2) > 0.99], enabling a 0.3 °C resolution as evidenced by the relative change in capacitance (ΔC/C)-temperature plot in Figure 4B, and supported spatial temperature mapping. This innovative platform consolidated skin temperature mapping, thermal imaging, and thermotherapy, directly addressing the drawbacks of traditional infrared systems - including their large size, lack of spatial data, and segregated monitoring/therapy functions. Thermistors are fundamentally based on strong dependence of materials’ electrical resistance on temperature[125,126]. In negative temperature coefficient (NTC) thermistors, the resistance decreases exponentially with increasing temperature, as thermally excited charge carriers overcome potential barriers and enhance electrical conductivity[127,128]. Zhang et al. developed an in-ear wearable device [Figure 4C] for continuous core body temperature monitoring, utilizing a NTC thermistor as the sensing element[129]. This device measured core temperature via the cochlear canal, delivering precise and stable readouts. In treadmill tests, in-ear temperature deviated from rectal temperature by at most 0.4 °C, whereas skin temperature differed by
Wearable pressure sensors
Instrumental to continuous health monitoring and smart wearables, pressure sensors convert mechanical stimuli such as compression or stretch into quantifiable electrical outputs[130,131]. They are broadly categorized by transduction principle - primarily capacitive[132-134], piezoresistive[135-137], and piezoelectric[18,138,139]. Here, we summarize several representative studies on three types of pressure sensors, encompassing their device material architectures, sensitivity, and lifespan, as presented in Table 3[140,141]. Their applications in human physiological monitoring are summarized in this section. With the rapid advancement of artificial intelligence (AI), wearable pressure sensors are increasingly integrated with AI techniques to enhance data analysis and system intelligence. By leveraging machine learning (ML) or neural network models, these systems achieve improved sensing accuracy and robustness in complex environments. Accordingly, this section also highlights representative pressure-sensing applications, including selected works that incorporate AI-enabled approaches.
Performances of the flexible pressure sensors
| Mechanism | Materials | Sensitivity | lifespan | Ref. |
| Capacitive | PDMS/Au/GO/Au | 0.03 kPa-1 (10-250 kPa) | > 10,000 cycles | Hou et al.[140] |
| Poloxamer-based ionic conductive hydrogel | GF = 4.37 (0%-10% strain) | > 200 cycles at 50% strain | Shi et al.[141] | |
| Piezoresistive | Velostat | None | > 100 cycles at 16% strain | Kim et al.[156] |
| CNT/ACET/PDMS | 0.36 kPa-1 | > 180,000 cycles under a pressure of 3.5 kPa | Wang et al.[157] | |
| Piezoelectric | Cu/PZT/Au | 2.2 kPa/V | 30% strain with only 17% reduction | Yuan et al.[163] |
| PEDOT:PSS/rGO/PVDF-TrFE/PEDOT:PSS/rGO | 0.0309 kPa-1 | > 500 cycles under an applied force of 0.5 N | Kil et al.[164] |
Capacitive pressure sensors
Capacitive pressure sensors transduce external stimuli by pressure-induced variations in electrode spacing, dielectric permittivity, or effective electrode area. Applied pressure causes reversible deformation of the dielectric layer or modulates its dielectric distribution, thereby producing a measurable change in capacitance. By incorporating microstructured dielectrics, pressure-responsive dielectric materials, or IDE configurations, capacitive sensors can achieve high sensitivity under low pressures while maintaining mechanical flexibility suitable for wearable applications[134]. They typically consist of two parallel electrodes separated by a dielectric layer. Under external pressure, the change of distance between electrodes or dielectric thickness leads to a variation in capacitance[142]. Researchers enhance sensor sensitivity through structural optimization. Hou et al. developed a transparent and tactile wearable sensor that converts arterial pulsation into detectable capacitive signals[140]. The sensor demonstrated excellent linearity and a sensitivity of 0.03 kPa-1 within 10-250 kPa. Moreover, after undergoing 10,000 cycles of testing at pressures ranging from 1 to 20 kPa, the sensor demonstrated no significant degradation in sensitivity. The signal amplitude and waveform consistency remained exceptional, far exceeding the frequency of use typically observed in routine clinical pulse monitoring. Figure 5A exhibited its dynamic responses under different pressures. Shi et al. developed a biocompatible hydrogel-based capacitive strain sensor [Figure 5B] with a porous structure that enabled high sensitivity[141]. Although the overall distribution of ions within the hydrogel network remained essentially unchanged under compressive deformation, variations in electrode spacing and interfacial contact area modulated ion transport efficiency, thereby translating pressure differentials into capacitance variations. This sensor exhibited hierarchical sensitivity across different strain ranges. At 0%-10% strain, the GF (sensitivity coefficient) reached 4.37, whereas at 10%-50% strain, the GF was 1.34, enabling precise detection of subtle deformations. The porous hydrogel sensor showed no significant capacitance degradation after 200 cycles at 50% compressive strain and maintained stable signals during low-temperature (-20 °C) robotic gesture recognition under repeated deformation and friction. Han et al. developed a flexible, interference-resistant capacitive pressure sensor using a poly(vinylidene fluoride) (PVDF)@AgNWs@TiO2 dielectric[143]. Pressure-induced deformation modulates capacitance, which the flexible electrodes transduce into electrical signals. At 0.25 wt% AgNWs@TiO2, this device achieves a sensitivity of 0.0012 kPa-1 with excellent linearity (R2 = 0.98) across an ultrabroad range up to 1,500 kPa, and exhibits a response/recovery time of 166.9/199 ms. After 33,000 loading–unloading cycles at 1,122.5 kPa, the capacitive response showed negligible fluctuation and no degradation. Capacitance variations remained ~0.1 pF even under electromagnetic and close-proximity interference. Meanwhile, the development of short-range noncontact capacitive sensors has also progressed rapidly[144-146]. Huang et al. proposed a flexible proximity–pressure dual-mode sensor (PPDS) based on a theoretically guided complementary Archimedean spiral electrode design and a microstructured calcium-crosslinked alginate (CCA) dielectric membrane[147]. As the target object gradually approaches the sensor, the distance between the sensor electrodes and the target object decreases, resulting in a significant increase in mutual capacitance. Consequently, a detectable variation in the overall capacitance of the sensor is induced. Zhou and Yuan proposed a non-contact capacitive sensor based on a layer-stacked structure composed of Ecoflex elastomer and chitosan/poly(acrylic acid-co-[2-(methacryloyloxy) ethyl] dimethyl-(3-sulfopropyl) ammonium hydroxide-co-ethylene glycol methyl ether acrylate) (CPASM) conductive organohydrogel, along with a dual-capacitance generation mechanism[148]. As the target object (such as a finger, metal, etc.) gradually approaches the sensor, the approaching object acts as a third electrode, which reduces the electric field strength associated with the capacitor plates by weakening the coupling between the two conductive hydrogel electrodes. Consequently, a detectable variation in the mutual capacitance of the sensor is induced, enabling sensitive sensing of proximity distance, movement speed, and the number of approaching objects.
Figure 5. Different types of pressure sensors and their performance. (A) Device schematic of the capacitive-based wearable sensor and its relative capacitance variation under different applied pressures. Reproduced with permission[140]. Copyright 2025, American Chemical Society; (B) Poloxamer-based ionic hydrogel as a wearable sensor and Relative capacitance changes of the porous ionic hydrogels. Reproduced with permission[141]. Copyright 2024, American Chemical Society; (C) Schematic illustration and its electrode-skin contact impedances. Reproduced with permission[156]. Copyright 2019, American Association for the Advancement of Science; (D) The structure of CNT/ACET/PDMS sensors and the normalized current variation of sensor under different pressures. Reproduced with permission[157]. Copyright 2025, American Association for the Advancement of Science; (E) Structure schematic of the piezoelectric transducer cell and BP testing on volunteers with different BMI. Reproduced with permission[163]. Copyright 2025, American Association for the Advancement of Science; (F) Pulse monitoring sensor structure and comparison of the magnitude of pulse signals measured at different locations. Reproduced with permission[164]. Copyright 2023, Elsevier B.V. CNT: Carbon nanotube; ACET: acetylene black; PDMS: polydimethylsiloxane; BP: blood pressure; BMI: body mass index; GO: graphene oxide; sEMG: surface electromyography; FPSM: flexible perovskite solar module; PZT: lead zirconate titanate; PEDOT:PSS: poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate); rGO: reduced graphene oxide; PVA: polyvinyl alcohol; PVDF-TrFE: poly(vinylidene fluoride-co-trifluoroethylene); AgNW: silver nanowire.
In addition to the aforementioned studies, a novel ion-electronic thin-film sensor capable of generating hybrid supercapacitive behavior has been proposed[149,150]. The pseudocapacitive behavior originates from reversible redox reactions occurring at or near the surface of the electrode materials, enabling this class of pressure sensors to overcome the intrinsic performance limitations of conventional electric double-layer capacitive iontronic pressure sensors[151,152]. Based on this mechanism, Yoon et al. recently proposed a new class of iontronic films exhibiting hybrid supercapacitive phenomena[153]. The resulting iontronic sensor achieved a high sensitivity of 301.5 kPa-1 within a pressure range of 1.7-13 kPa, while maintaining a sensitivity of 11.6 kPa-1 in the low-pressure region (< 1.7 kPa). Moreover, the sensor demonstrated a broad operating pressure range of up to 63.3 kPa, along with excellent mechanical durability, showing stable performance over 5,000 loading–unloading cycles and retaining more than 90% of its specific capacitance after 1,000 cycles.
Piezoresistive pressure sensors
Piezoresistive sensors detect pressure via stress-induced resistance changes in a conductive composite layer, where deformation alters inter-filler contact geometry[154]. This deformation alters the contact distance between the conductive filaments or regions distributed in the material matrix[155]. Kim et al. engineered a submental sensor on a PI substrate, integrating dual-differential surface electromyography (sEMG) electrodes with a piezoresistive strain gauge for the first time[156]. The electrodes, arranged parallel to submental muscle fibers and coated with a 500 nm gold layer, enabled synchronous acquisition of muscle activity and laryngeal motion while effectively suppressing common-mode noise. Participants performed various swallowing tasks, during which sEMG signals were simultaneously recorded alongside videofluoroscopic swallowing study (VFSS), confirming the temporal correlation between the signals and swallowing events. The reusability of the sensor patch is primarily limited by reduced skin adhesion caused by contaminants such as dust, sweat, and facial hair. Consequently, the sensor can only be reused approximately 5-10 times. The recorded sEMG signals showed a correlation coefficient exceeding 0.95 with a commercial system [Figure 5C]. Wang et al. integrated a piezoresistive pressure sensor based on CNT/acetylene black (ACET)/PDMS into an insole, as shown in the upper panel of Figure 5D[157]. External pressure, such as plantar loading during gait, compresses the porous PDMS, reducing its porosity and increasing the contact area within the CNT/ACET conductive network. This results in the formation of additional conductive pathways, leading to changes in the sensor resistance. As shown in the lower panel of Figure 5D, the normalized current response of the sensor under varying applied pressures is presented. Voltage signals from 22 sensors embedded in the insole were recorded to capture temporal variations in plantar pressure during walking, running, and stair ascent/descent, from which key features were extracted. The core model for gait analysis and locomotion state recognition is a support vector machine (SVM), which enables accurate classification of eight distinct motion states. By exploiting the spatiotemporal characteristics of plantar pressure signals, this approach establishes a mapping between locomotion states and pressure distribution patterns, thereby achieving high-accuracy recognition of gait-related motion states. Recent advances have introduced nanomembranes as sensing elements, enabling high-resolution mechanical and electrophysiological sensing while promoting ultrathin biosensor technologies[158-160]. Cheng et al. fabricated silicon nanomembranes from p-type silicon-on-insulator (SOI) wafers and leveraged their piezoresistive effect[161]. Through heavy doping and etching, the sensors achieved enhanced conductivity and flexibility, operating stably over
Piezoelectric pressure sensors
Piezoelectric pressure sensors detect mechanical stimuli by converting stress induced lattice deformation into changes in material polarization. Applied pressure shifts the orientation of electric dipoles, generating transient charges or voltage between the electrodes[154,162]. Yuan et al. developed a skin-conformal, self-adaptive flexible ultrasound transducer integrated with piezoelectric sensors [Figure 5E] that can be adhered tightly to arterial sites for signal acquisition[163]. It exhibited a SNR of 30.1 dB, a penetration depth exceeding 30 mm, and a beam width of 2.1-4.6 mm with a focal depth of 3.3-53 mm, covering the curvature and depth of most human vessels. As shown at the bottom of Figure 5E, the system was successfully applied to blood pressure measurement and continuous monitoring across subjects with different body mass index (BMI) values. During continuous 24-h blood pressure monitoring, the signal exhibited no observable drift, with mean errors of only ±2.48 mmHg for systolic blood pressure and ±1.99 mmHg for diastolic blood pressure compared with a commercial sphygmomanometer. Kil et al. developed a piezoelectric pressure sensor integrated with a supercapacitor [Figure 5F], in which pressure-induced compression of poly(vinylidene fluoride-co-trifluoroethylene) (PVDF-TrFE) nanofibers enhances β-phase orientation and increases spontaneous polarization by 30%-50%[164]. The resulting rise in surface charge density attracts additional H+ and PO43- ions, forming a pressure-dependent electric double layer whose ion adsorption modulates the device capacitance. This hybrid design enables high-precision, self-powered monitoring of carotid pulse waveforms. The sensor exhibited a sensitivity of 0.0309 kPa-1 in the low-pressure range below 10 kPa, 4.6 times higher than that of a pure supercapacitor - and could detect subtle pressure variations as small as 0.1 N. In the higher-pressure range (> 10 kPa), the sensitivity was 0.02556 kPa-1. The sensor was subjected to 500 compression–release cycles under an applied force of 0.5 N, during which no noticeable attenuation in the voltage signal was observed. However, long-term performance was not systematically evaluated in human wearable tests. As shown at the bottom of Figure 5F, the device successfully distinguished pulse signal amplitudes measured from different body sites.
Wearable biofluid sensors
Sweat, tears, and saliva are biofluids rich in physiological information, offering valuable pathways for noninvasive health monitoring[165-167]. Sweat is continuously secreted and contains key biomarkers such as sodium ions, glucose, lactate, and cortisol, whose concentration variations often reflect electrolyte balance or metabolic fluctuations[168]. Tears are composed of health-related molecules including glucose, proteins, and cortisol, which can serve as early diagnostic indicators for ocular diseases and systemic metabolic disorders[169,170]. Saliva contains a variety of hormones, metabolites, and inflammatory factors, enabling the assessment of stress levels, metabolic imbalances, and oral health. This section reviews recent advances in noninvasive wearable sensing technologies based on sweat, tears, and saliva.
Sweat monitoring
Sweat, as a readily accessible and noninvasively collectible biofluid, is endowed with a wealth of physiological and pathological biomarkers, which mainly encompass electrolytes (e.g., sodium, potassium, chloride, ammonium, calcium), metabolites (e.g., glucose, lactate, alcohol), and trace elements (e.g., iron, zinc, copper)[171]. Conventional detection methods are plagued by inherent limitations such as bulky and costly equipment, poor portability, and intricate sample processing procedures[172]. To address these inherent limitations, the development of a wearable platform for physiological information acquisition has emerged as an imperative demand, which facilitates real-time, non-invasive, and dynamic monitoring of physiological parameters. Wearable sweat sensors typically operate based on electrochemical or colorimetric principles, and their detection strategies and target analytes are summarized in Table 4[173-180]. Electrochemical analysis is commonly employed in fluidic biosensing, detecting analytes by measuring electrical signals generated from redox reactions or ionic variations at the electrode–electrolyte interface[181]. By integrating electrodes with selective recognition materials, electrochemical sensors enable continuous monitoring of key biomarkers such as glucose, lactate, sodium, and potassium[182,183]. This approach offers high sensitivity, rapid response, and excellent compatibility with microfluidic systems for noninvasive and continuous healthcare monitoring.
Summary of representative strategies and target analytes for sweat monitoring
| Detection mechanism | Typical analytes | Representative materials or methods | Ref. |
| Electrochemical analysis | K+ | Valinomycin-selective membrane | Ma et al.[173] |
| Lactate; glucose | Lactate oxidase–modified electrode; glucose oxidase–modified electrode | Son et al.[174] | |
| pH | Polyaniline | Sun et al.[175] | |
| pH | Lactate oxidase–modified electrode | Qu et al.[176] | |
| Colorimetric analysis | Urea | Urease | Yue et al.[177] |
| Sweat rate and volume | Paper chips modified by dyes | Li et al.[178] | |
| Vitamin C | Ferric reducing/antioxidant and ascorbic acid | Kim et al.[179] | |
| Amino acids, lysine, alanine | Enzyme-coupled fluorescence reactions | Cho et al.[180] |
Integrating electrochemical sensors onto flexible microfluidic substrates such as fibers or polymers significantly enhances their stretchability and wearing comfort for wearable sweat monitoring. Ma et al. developed a monolithically integrated sweat sensor embedded in a textile wristband [Figure 6A, upper][173]. By incorporating a valinomycin-selective membrane into the sensing interface, the device achieved precise detection of K+ ions with a high sensitivity of 66 mV per decade. As shown in Figure 6A, this sensor exhibited stable potential responses to K+ solutions with varying concentrations over time. Sun et al. developed a stretchable smart-wettability sensing patch [Figure 6B][175]. This pH sensor covered a wide detection range of 3.75-8.20. By electrodepositing polyaniline (PANI) onto a multi-walled CNT (MWCNT) electrode, the device responded to H+ concentration through the protonation–deprotonation process of PANI, exhibiting a high sensitivity of 65.7 mV per decade.
Figure 6. Sensors for sweat monitoring and their performance. (A) Schematic of textile wristband for wireless sweat analysis and the response of the K+ sensor to different K+ concentrations. Reproduced with permission[173]. Copyright 2018, American Association for the Advancement of Science; (B) Schematic illustration and optical image of a sweat-sensing patch attached to the skin and the sensors show open-circuit potentials in physiologically relevant analytes in the range of 3.75 to 8.20 pH. Reproduced with permission[175]. Copyright 2024, American Chemical Society; (C) Illustration of the composition of the sweat sensor and its color and RGB responses of the colorimetric sensing units toward glucose, lactate, urea, and pH. Reproduced with permission[177]. Copyright 2022, American Chemical Society; (D) Illustrates the three-dimensional schematic structure of the flexible multifunctional microfluidic colorimetric detection device and photographs of the microfluidic detection device after the sweat reached different positions. Reproduced with permission[178]. Copyright 2025, American Chemical Society. RGB: Red, green, blue; PVB: polyvinyl Butyral; PVDF-HFP: poly(vinylidene fluoride-co-hexafluoropropylene); TSR: temperature-sensitive resistor; PDMS: polydimethylsiloxane.
Colorimetric analysis is another widely used method in sweat monitoring, which detects analytes through color changes caused by chemical or enzymatic reactions[178,184]. By using chromogenic reagents that respond to specific biomarkers such as pH, chloride, glucose, or lactate, the concentration of these analytes can be visually or optically quantified[172,185]. Yue et al. developed a low-cost and flexible colorimetric sweat sensing platform based on a PET substrate integrated with paper-based microfluidic channels [Figure 6C][177]. The system enables colorimetric detection of glucose, lactate, urea, and pH in sweat, exhibiting excellent linear correlations for multiple biomarkers: glucose (0-2 mM, R2 = 0.9615), lactate (0-30 mM, R2 = 0.9816), urea (0-10 mM, R2 = 0.9674), and pH (4-8, R2 = 0.9604).
In monitoring sweat loss and sweat rate, configuration and paper chips are typically designed to collect sweat specimens[186,187]. These paper chips are fixed at distinct positions within the channel and appropriately labeled. As sweat flows into a serpentine channel, it sequentially wets the differently colored paper chips, forming a visible color band. By observing the position of the color chip reached by the sweat front, the approximate range of sweat volume can be preliminarily determined. Based on this strategy, Li et al. developed a multifunctional microfluidic colorimetric detection device [Figure 6D][178]. This device integrated a capillary bursting valve (CBV) with a specific geometric structure and paper chips containing specialized colorimetric reagents, enabling sequential monitoring of sweat volume and sweat rate.
Tear monitoring
Although sweat sensors have shown great promise for noninvasive and continuous health monitoring, yet their performance is often affected by irregular sweat secretion, evaporation variability, and environmental factors[188,189]. To overcome these challenges and broaden noninvasive biofluid analysis, researchers have turned to tears, which contain numerous health-related biomarkers such as glucose, proteins, and cortisol[189,190]. Tear proteins, in particular, can indicate ocular health and have been identified as early diagnostic markers for diabetic retinopathy, aniridia, and dry eye syndrome. Similarly, wearable tear-based sensors operating on electrochemical or colorimetric principles have their sensing strategies and target analytes summarized in Table 5[191-195]. Consequently, developing comfortable and user-friendly wearable tear sensors is essential for routine health monitoring. Park et al. have developed a flexible intelligent contact lens, which for the first time realized the integration of “wireless power supply, tear glucose detection, and real-time display”[196]. It was verified that the glucose sensor covered a detection range of 0.1-0.9 mM, with a response time of ~1.3 s. It only required naturally secreted tears to complete the detection, without the requirement for additional sample collection. The lens was fabricated on a soft and transparent silicone elastomer substrate, where the glucose sensor, wireless circuit, and micro-LED are interconnected through stretchable transparent conductors. As shown in Figure 7A, the relative change in resistance decreased linearly with increasing glucose concentration, and the sensitivity - defined as the slope of the calibration curve - was calculated to be approximately -22.72% mM-1. Ku et al. developed a graphene field-effect transistor (FET)-based cortisol immunosensor integrated with transparent antennas and near field communication (NFC) in a flexible smart lens [Figure 7B][197]. The graphene channel was functionalized with cortisol monoclonal antibodies (C-Mab) through 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC)/N-hydroxysulfosuccinimide (NHS) coupling on a chemical vapor deposition (CVD)-grown graphene layer transferred onto a soft substrate, enabling selective and stable immunorecognition. This device detected cortisol levels from 1-40 ng/mL in artificial tears, covering physiological ranges, with a sensitivity of 1.84 ng/mL per 1% ΔR (the absolute change in resistance of the sensor induced by an external stimulus). Yin et al. developed a wearable dual-emission fluorescent eye patch for simultaneous detection of four fluoroquinolone antibiotics in human tears[198]. The patch adheres to periocular skin without eye contact, avoiding irritation, and requires only ~40 μL of tears for analysis within 5 min. This patch consists of a biocompatible hydrogel substrate embedded with dual-emission carbon dots and Eu3+ complexes, enabling stable fluorescence signals and mechanical flexibility. Results showed 3.3%-6.4% deviation from HPLC, and the quantitative response to norfloxacin concentration, as shown in Figure 7C.
Figure 7. Sensors for tear monitoring and their performance. (A) Structure and core components of the flexible smart contact lens and the response of the glucose sensor to different glucose concentrations. Reproduced with permission[196]. Copyright 2018, American Association for the Advancement of Science; (B) Structure of the smart contact lens and the response of this sensor to different concentrations of cortisol. Reproduced with permission[197]. Copyright 2020, American Association for the Advancement of Science; (C) Wearable eye patch for tear monitoring and the response of this sensor to NOR. Reproduced with permission[198]. Copyright 2024, American Chemical Society. NOR: Norfloxacin; PDMS: polydimethylsiloxane.
Summary of representative strategies and target analytes for tear monitoring
| Detection mechanism | Typical analytes | Representative materials or methods | Ref. |
| Electrochemical analysis | Glucose | Fe/Co bimetallic oxides | Zhou et al.[191] |
| Urea | Ni-Co-MOF hydrogel | Mukundan et al.[192] | |
| Cholesterol | Cholesterol oxidase | Chen et al.[193] | |
| Colorimetric analysis | Glucose | Cerium oxide nanoparticles | Park et al.[194] |
| Vitamin C | 2, 6-Dichloroindophenol | Wang et al.[195] | |
| Protein | Citric acid and tert-butyl peroxybenzoate |
Saliva monitoring
Wearable saliva sensors offer a promising noninvasive approach for real-time health monitoring, enabling detection of biomarkers such as glucose, cortisol, cytokines, and electrolytes[199-201]. Made from flexible, biocompatible materials and integrated into mouthguards or oral patches, they provide comfort and practicality. Wearable saliva-based sensors operating on electrochemical or colorimetric principles have their sensing strategies and target analytes summarized in Table 6[202-206]. Cortisol, a key stress biomarker, regulates metabolism and blood glucose; its abnormal levels can cause obesity, fatigue, and endocrine disorders, highlighting the importance of continuous monitoring[207-209]. Cho et al. developed a breathable bioelectronic mask integrating a nanonet cortisol biosensor via electrospinning [Figure 8A][210]. The mask enabled saliva collection through natural actions such as coughing or speaking, allowing noninvasive cortisol monitoring. The sensor was fabricated on an electrospun poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP) nanomesh substrate with interdigitated Au/Ag electrodes, subsequently functionalized with a cortisol monoclonal antibody through dithiobis(succinimidyl propionate) (DTSP) linkers to ensure selective molecular recognition. The sensor showed a linear response from 100 pM to 1 μM, with charge transfer resistance (Rct) increasing by ~5%-8% per 100 pM of cortisol. Kim et al. developed an intelligent bioelectronic dental guard integrating enzyme-modified electrodes and wireless electronics[211]. It continuously collects saliva samples non-invasively via direct saliva contact, enabling precise real-time monitoring of salivary uric acid. The saliva sensor demonstrated a sensitivity of 2.32 mA/mM toward uric acid detection, indicating its high electrochemical responsiveness to the target analyte, shown in Figure 8B. Physiological monitoring of newborns is extremely challenging and demands specialized platform. Lim et al. developed a thin, portable wireless bioelectronic system for salivary electrolyte monitoring [Figure 8C][212]. Microfluidic channels continuously draw saliva from the mouth for real-time electrolyte monitoring. This novel device integrated a flexible Bluetooth circuitry with polymer-coated ion-selective electrodes embedded in a PDMS-based microfluidic channel, ensuring both flexibility and biocompatibility. Results showed that this sodium sensor had a sensitivity of 52 mV/decade and the potassium sensor had 57 mV/decade, enabling accurate quantitative detection of both ions.
Figure 8. Sensors for saliva monitoring and their performance. (A) Physiological mechanism and application scenarios of smart bioelectronic nanomesh face masks for monitoring cortisol in saliva, as well as the electrochemical impedance spectroscopy plot of this sensor and the relationship between the sensor’s relative resistance change rate and cortisol concentration. Reproduced with permission[210]. Copyright 2025, American Chemical Society; (B) Mouthguard biosensor integrated with wireless amperometric circuit board and current response of this sensor to different concentrations of uric acid. Reproduced with permission[211]. Copyright 2015, Elsevier B.V; (C) Application scenarios and structural details of the smart bioelectronic pacifier, and the change of sodium - to - potassium ion ratio in saliva over time. Reproduced with permission[212]. Copyright 2022, Elsevier B.V. DTSP: Dithiobis(succinimidyl propionate); C-Mab: cortisol monoclonal antibodies; EA: ethanolamine; BSA: bovine serum albumin; PPD: polymerized o-phenylenediamine.
Summary of representative strategies and target analytes for saliva monitoring
| Detection mechanism | Typical analytes | Representative materials or methods | Ref. |
| Electrochemical analysis | Glucose | Glucose-oxidase-based enzymatic detection | García-Carmona et al.[202] |
| Glucose | Glucose-oxidase-based enzymatic detection | Sun et al.[203] | |
| Uric acid | Three-dimensional nitrogen-doped spherical carbon aerogels | Bi et al.[204] | |
| Colorimetric analysis | Glucose | Organic dyes | Bordbar et al.[205] |
| Glucose and creatinine | Polyethylene glycol | Chi et al.[206] |
Wearable ultrasound sensors
With the advancement of medical technologies, wearable ultrasound sensors have undergone rapid development[213-215]. These devices are characterized by a compact form factor that does not restrict the wearer’s mobility or interfere with daily activities. Wearable ultrasound sensors are designed to enable continuous monitoring of internal tissue structures and to provide therapeutic interventions without the need for manual operation. Dutta et al. developed a densely packed, stretchable ultrasonic array sensor based on surface charge engineering, employing a 1-3 lead zirconate titanate (PZT) composite as the core transduction unit[216]. The device leverages its excellent piezoelectric properties for efficient ultrasonic transmission and reception, while sensing is achieved through dynamic capacitance variations induced by ultrasonic interactions with the target. The system enables recognition of multiple human postures and hand gestures at distances of up to 7 cm through capacitance variations induced by ultrasonic interactions. Wang et al. developed an origami-inspired adaptive wearable ultrasonic transducer (AWUT) using projection micro-stereolithography (PμSL) 3D printing, integrating a 1-3 PZT-5H piezoelectric composite to derive arterial blood pressure from time- and frequency-domain ultrasonic echoes, enabling noninvasive, continuous, and accurate blood pressure monitoring[217]. Zou et al. developed a fully integrated conformal wearable ultrasound patch (CWUS Patch) based on a 1-3 PZT-5 piezoelectric composite, which can be stretched up to 30% strain to accommodate skin curvature[218]. When laminated onto the skin, the patch employs a printed circuit board (PCB)-driven PZT array to generate focused ultrasound (FUS) that precisely penetrates tumor tissue. The ultrasound activates hematoporphyrin monomethyl ether (HMME) within the tumor region, efficiently transferring energy to oxygen molecules to generate abundant reactive oxygen species (ROS). These ROS disrupt tumor cell structures and induce apoptosis. This work advances the clinical translation of sonodynamic therapy, providing a safer and more convenient therapeutic option for cancer treatment. Wearable ultrasound sensors represent an emerging and highly promising class of wearable technologies. By enabling non-invasive, real-time interrogation of internal biological tissues, these devices overcome the intrinsic limitations of conventional surface-based wearable sensors that are restricted to skin-level signals. Owing to their deep tissue penetration capability, high spatial resolution, and strong adaptability to complex physiological environments, wearable ultrasound sensors hold significant potential for continuous health monitoring, disease diagnosis, and therapeutic interventions in both clinical and daily-life settings.
Wearable sensing enhanced by ML
Wearable sensors have shown broad application potential in human–machine interaction (HMI)[219-221]. In particular, wearable pressure sensors that perceive and respond to external stimuli play a crucial role in advancing HMI technologies. The dexterity of human fingers enables diverse actions such as grasping and gestures, which can be converted into analyzable digital signals with the assistance of sensors, thereby enhancing the naturalness and intuitiveness of intelligent HMI systems. Guo et al. developed a drum-structured triboelectric nanogenerator (DS-TENG)-based wearable pressure sensor to detect wrist flexor digitorum superficialis tendon (FDST) vibrations induced by finger bending[219]. Figure 9A illustrates the schematic of gesture recognition and wireless real-time robotic control based on the DS-TENG wearable pressure sensor array. Different finger gestures generate distinct FDST vibration amplitudes and areas. A 2 × 2 sensor array mounted on the wrist produces analog voltage signals, which are processed using a random forest (RF) ML model to simultaneously recognize motions of five fingers with an accuracy of up to 92%. More importantly, the high-precision gesture recognition enables wireless real-time control of robotic systems, demonstrating precise and intuitive HMI. Chen et al. developed a resistive flexible pressure sensor array based on a multilevel dome structure to detect palm muscle pressure distributions induced by hand manipulation actions[222]. As shown in Figure 9B, different operational gestures (e.g., pushing a joystick or controlling a throttle) generate distinct muscle pressure patterns, and sensors at different indexed positions collect different pressure signals. The collected data for each action were preprocessed using a RF algorithm, with 70% of the data randomly selected as the training set and the remaining data used for testing. The proposed method achieved a prediction accuracy of up to 100%, demonstrating the effectiveness of the classification algorithm. By identifying muscle activation patterns in the human hand, this approach enables accurate discrimination of different hand movements and provides precise feedback on fine motor performance, thereby demonstrating its potential in medical rehabilitation applications. Meanwhile, multifunctional biomimetic sensors are gaining increasing practical importance in intelligent biomimetic robotic systems. Li et al. developed a biomimetic electronic palm system (BEPS) capable of decoupled dual-modal sensing of pressure and temperature[223]. The pressure-sensing unit employed a PVDF-HFP ionogel with a periodically layered micro-cone structure fabricated via a 3D-printed template, where pressure-induced changes in the contact area with electrodes led to capacitance variations. Temperature sensing relied on the temperature-dependent conductivity of PANI, resulting in resistance changes in the sensing layer. The decoupled bimodal signals were classified using a decision tree and convolutional neural network (CNN) algorithms. Figure 9C presents thermal images of the cup captured under different conditions. Figure 9D shows that, after the robotic arm successfully grasps the cup, the system processes the acquired temperature data and their spatial information to calculate the real-time temperature and water level of the grasped cup.
Figure 9. Applications of ML in the field of wearable sensors. (A) The schematic of gesture recognition and wireless real-time robotic control based on the DS-TENG wearable pressure sensor array. Reproduced with permission[219]. Copyright 2025, Wiley-VCH GmbH; (B) Different operational gestures generate distinct muscle pressure patterns, and sensors at different indexed positions collect different pressure signals. Reproduced with permission[222]. Copyright 2024 Wiley-VCH GmbH; (C) Thermal images of the cup captured under different conditions; and (D) Interface for water level and temperature recognition in a cup, displaying recognition results and corresponding curves for different temperatures and water levels. Reproduced with permission[223]. Copyright 2025, Wiley-VCH GmbH. ML: Machine learning; DS-TENG: drum-structured triboelectric nanogenerator; MCU: microcontroller unit.
INTEGRATION OF WIRELESS BIOSENSING TECHNOLOGIES
Wearable biosensing is an end-to-end system integration problem[224]. It couples the sensing front-end and bio-interface, the power supply/energy management, and the wireless link for data delivery. In practice, sensing elements are interfaced with modality-specific analog front-ends (e.g., amplification, filtering, and impedance matching), followed by digitization and local processing [analog-to-digital converter (ADC) and microcontroller unit (MCU)/system on chip (SoC)]. Power management (battery/energy harvesting, regulation, and duty cycling) co-determines the achievable sampling strategy and wireless budget, while the wireless transceiver and antenna/packaging deliver data to a smartphone or gateway for storage and analytics[225,226]. Chapters 2-3 review key material designs and representative sensing modalities for physiological and biofluid monitoring. Here, we focus on wireless integration and its tightly coupled enabling modules, because the wireless link often defines the practical form factor, lifetime, and user experience. The integration of wearable sensors into daily life depends on efficient and reliable data transmission[227]. While traditional wired connections restrict movement and impose psychological burdens, wireless technologies enable unobtrusive monitoring of physiological data. Bluetooth low energy (BLE) and Wi-Fi are battery-powered solutions that support continuous connectivity, with BLE prioritizing low power and Wi-Fi enabling higher throughput. NFC and radio frequency identification (RFID) are typically reader-powered in wearable-tag implementations and enable battery-free operation, with NFC favoring very short-range smartphone-based readout and RFID supporting broader coverage and multi-tag access depending on the band and infrastructure. Resonant coupling provides another battery-free route, where sensing information can be encoded in resonance characteristics, enabling minimalist circuitry and long-term operation, but often with stronger coupling and detuning constraints. Supported by Bluetooth, Wi-Fi, advanced power supplies, and flexible antennas, active wireless devices are becoming increasingly efficient, stable, and compact[228]. Meanwhile, passive sensors centered on technologies such as RFID, NFC, and resonant coupling (RC) technology demonstrate unique value in disposable and implantable medical applications[229]. Furthermore, emerging architectures such as integration of communication and sensing functions (ISAC), body area networks (BANs), and integration of multiple communication technologies (IMCTs) are further enabling the synergistic integration of sensing, power, and communication within microsystems[230]. Table 7 summarizes key trade-offs and representative application scenarios for BLE, Wi-Fi, NFC, RFID, and resonant coupling.
Comparison of representative wireless technologies for wearable biosensing
| Technology | Data ratea | Rangeb | Powerc | Key advantages | Key limitations | Application | Ref. |
| BLE | Low-moderate | Long | Moderate | Low power; mature ecosystem; continuous link | Battery-limited; body detuning; limited throughput | Continuous vitals; multi-sensor wearables; low-power telemonitoring | [231-247] |
| Wi-Fi | High | Long | High | High throughput; easy infrastructure access | High power; packaging/thermal burden | Data-intensive sensing; indoor/clinical connectivity; high-rate streaming | [248-253] |
| NFC | Low | Short | Low | Battery-free; ultra-thin; phone readout | Cm-range; alignment; on-demand | On-demand patches; sweat/wound snapshots; disposable readout | [254-264] |
| RFID | Low | Moderate-long | Low | Longer range; multi-tag; scalable | Reader needed; environment/body shadowing; small payload | Multi-tag tracking; infrastructure-based sensing; low-maintenance monitoring | [265-270] |
| Resonant coupling | Low | Very short | Low | Minimal circuitry (chipless); battery-free; long-term | Coupling-sensitive; detuning; multiplexing hard | Minimalist passive; implantable/disposable; long-term battery-free monitoring | [271-282] |
Active communication technologies
Active communication solutions for wearable sensors have become the mainstream choice for multi-functional and demanding applications due to their stable performance and strong adaptability[283,284]. Active sensor nodes with integrated power enable continuous wireless transmission, offering key advantages in range, data rate, and reliability.
Bluetooth and BLE are widely used personal area network (PAN) technologies operating in the 2.4 GHz frequency band. Designed specifically for minimal power consumption, BLE is ideal for integration into wearable electronics. The Bluetooth module is highly compatible with various power modules and terminal devices such as smartphones and computers. BLE is widely used for long-term monitoring of physiological signals such as pulse[231,232], respiration[233,234], electroencephalography (EEG)[235], and ECG[236] due to its low power consumption. As shown in Figure 10A, Li et al. developed a thin, soft, wearable system that measures arterial blood pressure[237]. Powered by a coin-sized rechargeable lithium battery (80 mAh), it can operate continuously for nearly two days when taking measurements six times per hour, each lasting 15 s [Figure 10A(ii)]. Additionally, Bluetooth technology efficiently supports simultaneous transmission from multiple sensors, making it suitable for applications requiring multi-sensor signal detection, such as multimodal sweat analysis[238-242], emotion monitoring[243], infant care[243-245], and telemedicine[246,247]. As shown in Figure 10B, Lee et al. proposed a personalized skin-integrated wireless facial interface that incorporates dual-mode strain and vibration sensors to capture facial expressions and vocal cord vibrations[243]. Multi-channel physiological signals from different sensing areas (e.g., forehead, eyes, lips, vocal cords) are transmitted in real time via BLE to a computer for ML-based emotion recognition [Figure 10B(ii)].
Figure 10. Wearable sensors based on active communication technology. (A) The wearable wireless continuous blood pressure monitoring system based on BLE. (i) The system overview, (ii) Lifetime of the rechargeable lithium-ion battery in intermittent working mode. Reproduced with permission[237]. Copyright 2023, Springer Nature Limited; (B) PSiFI system based on BLE. Reproduced with permission[243]. Copyright 2024, Springer Nature Limited; (C) A flexible smart wearable alarming system. (i) System overview schematic diagram, (ii) Optical image of flexible dual-mode sensor[248]. Copyright 2025, Springer Nature Limited; (D) Wireless, integrated system for real-time, onboard, adaptive wound diagnostics and treatment. (i) CAD model of There Heal wearable device, (ii) The Heal platform. Reproduced with permission[253]. Copyright 2025, Springer Nature Limited; (E) All-polymer aqueous sodium-ion battery with PANI as symmetric electrodes[292]. Copyright 2024, Springer Nature Limited; (F) The self-powered wearable leg movement sensing system. Reproduced with permission[302]. Copyright 2023, Wiley-VCH GmbH; (G) Photograph of the stretchable patch antenna array[309]. Copyright 2025, Wiley-VCH GmbH; (H) Direct-ink-writing printed stretchable eutectic gallium-indium antenna. Reproduced with permission[310]. Copyright 2025, Wiley-VCH GmbH. BLE: Bluetooth low energy; PSiFI: personalized skin-integrated facial interface; CAD: computer-aided design; PANI: polyaniline; TENG: triboelectric nanogenerator; CNN: convolutional neural network.
Wi-Fi’s high transmission rates (up to hundreds of Mbps or Gbps) keep it crucial for real-time, high-data-rate signals such as ultrasound images[249,250], motion trajectories[251,252], and high-resolution pictures[253]. Shown in Figure 10C, Zhou et al. developed a flexible “artificial epidermis” assembled from a three-dimensionally conductive metal-organic framework (MOF), enabling dual-mode sensing of NO2 gas and pressure[248]. To support stable long-range connectivity with cloud services and smart devices, the system uses an ESP32 Wi-Fi link to stream sensor data directly to the cloud/terminals for real-time remote monitoring and early warning. As shown in Figure 10D, Li et al. proposed an adaptive wound platform that captures high-definition wound images for ML diagnosis[253]. The large size of these image data is much greater than simple physiological parameters. This powerful data transmission capability of Wi-Fi ensures that data can be collected and transmitted every 2 h.
In wearable form factors, the performance of active radios is jointly determined by the protocol, the power source and the RF front-end/antenna. Although Bluetooth and Wi-Fi technologies ensure stable information exchange, wearable devices also remain constrained by rigid batteries with limited capacity and flexible antennas with unreliable performance during movement. These core hardware challenges drive research to improve the overall device. In terms of power supply, researchers introduced materials such as MXene[285,286], graphene[287,288], CNTs[289,290], PEDOT:PSS[291], and hydrogels[292,293] to enhance the flexibility, biocompatibility, and energy storage efficiency of batteries. Additionally, recent progress in fiber-based batteries provides mechanically compliant, textile-integrable power solutions for wearable devices, helping alleviate the form-factor and reliability constraints associated with rigid cells[294-298]. For example, Wu et al. reported an interface-engineered self-healing quasi-solid fiber battery enabled by borate-ester dynamic crosslinking[295]. The device achieved an energy density of 104 Wh/kg, a power density of 20.8 W/kg, tolerated strain exceeding 25%, and maintained thermal stability up to 200 °C, supporting mechanically robust textile-integrable power supplies. As shown in Figure 10E, Hong et al. developed an all-polymer aqueous sodium-ion battery using symmetric PANI electrodes and a polymer-aqueous electrolyte that stabilizes redox products by modulating solvation layers and forming a solid-electrolyte interphase[292]. It delivered
The antenna serves as the central element in Bluetooth and Wi-Fi communication modules, responsible for transmitting and receiving wireless signals. Unlike rigid antennas that weaken or break when bent, flexible antennas were developed to move with the body, ensuring reliable connectivity and comfort. Researchers improve their performance under dynamic bending and stretching by incorporating new substrate materials such as PDMS, Ecoflex, and textiles, along with advanced conductors such as traditional metals, AgNWs, and liquid metal[306-308]. Kim et al. developed a stretchable RF device using a “dielectric elastic” substrate, where embedded ceramic nanoparticles linearly tune the dielectric constant during stretching to counteract resonant frequency shifts, preserving wireless performance[306]. As shown in Figure 10G, Li et al. proposed a stretchable mesh metal antenna design using a serpentine mesh layout, which achieves high flexibility while maintaining stable electromagnetic performance[309]. Integrated on the surface of a balloon, it forms a directional wireless communication system with a measured gain of 7.69 dB and enables reliable multi-target communication over a distance of 100 m. As shown in Figure 10H, Guo et al. proposed a method for fabricating broadband stretchable antennas by printing thermoplastic polyurethane-modified gallium-indium eutectic (EGaIn) ink using direct-write technology[310]. These antennas maintained stable wireless communication over 50 m even after undergoing 240% stretching.
Passive communication technologies
The constraints in batteries and deformation-sensitive RF hardware motivate increasing interest in reader-powered and passive communication strategies, which are discussed next. Passive wireless sensors, which are typically based on NFC, RFID, and resonant coupling technologies, harvest energy from reader devices (e.g., smartphones) via near-field or far-field methods and communicate by backscattering sensor data[283,311]. This enables ultra-thin, flexible, and even structurally integrated form factors, free from battery constraints. It also drastically lowers the cost and complexity of each sensor node, paving the way for single-use or permanent implantable monitoring devices[229,312].
NFC is a short-range wireless technology operating at 13.56 MHz, which typically works in a reader-powered mode using electromagnetic induction to harvest energy from a reader and transmit data. Its simple antenna and miniaturized chip allow thin, flexible integration into wearables without affecting comfort, making NFC suitable for wound monitoring[254-256], breath analysis[257,258,260], sweat sensing[261,262], and glucose testing[263,264]. As shown in Figure 11A, Cho et al. proposed a battery-free, wireless multimodal sensing platform for continuous monitoring of pressure, temperature, and skin hydration in wheelchair users, where both power delivery and data readout rely on an NFC near-field link via resonant magnetic inductive coupling[259]. As presented in Figure 11B, Li et al. developed a multifunctional pathogen infection diagnosis system (PIDS) that integrates immune detection, respiration monitoring, and temperature measurement into a compact NFC circuit, enabling rapid virus screening with a smartphone tap[260]. NFC technology offers a simple and convenient way for wearable devices to monitor health, facilitating effective health management.
Figure 11. Wearable sensors based on passive communication technology. (A) Wireless, multimodal sensors for continuous measurement of pressure, temperature, and hydration of patients in wheelchairs. (i) Schematic illustration of the movable system, (ii) Schematic illustration of the battery-free, wireless sensing platform that includes crack-activated pressure sensor, temperature sensor, and GSR sensor[259]. Copyright 2023, Springer Nature; (B) NFC-based PIDS. (i) Structure, components, and multiple modules of PIDS, (ii) Block diagram of PIDS, (iii) Photograph of real-time detection. Reproduced with permission[260]. Copyright 2023, Springer Nature Limited; (C) The fully integrated passive wireless sensing system based on RFID technology. Reproduced with permission[267]. Copyright 2024, American Chemical Society; (D) Schematic illustration of a passive RFID tag that recognizes eye movement[270]. Copyright 2024, Wiley-VCH GmbH; (E) Hydrogel bio interface for implantable pressure sensors based on RC technology. (i) Exploded view, (ii) Detection principle. Reproduced with permission[271]. Copyright 2024, Wiley-VCH GmbH; (F) Wireless immunoassays for virus aerosol detection, multiplexed readout of ImmHR and temperature sensors. Reproduced with permission [314]. Copyright 2024, Springer Nature Limited. GSR: Galvanic skin response; NFC: near field communication; PIDS: pathogen infection diagnosis system; RFID: radio frequency identification; RC: resonant coupling; SOC: system on chip; fPCB: flexible printed circuit board; TBS: temperature biosensor; RBS: respiration biosensor; PI: polyimide; IBS: immuno biosensor; CPU: central processing unit; RAM: random access memory; ADC: analog-to-digital converter; MWS: multiparameter passive wireless sensor; PLGA: poly(lactic-co-glycolic acid); POC: poly(1,8-octanediol-co-citrate); SARS: severe acute respiratory syndrome; RSV: respiratory syncytial virus; IR: infrared.
Unlike NFC with its centimeter-range limitation, RFID technology provides several meters of reading range, multi-tag identification, and a miniaturized chip design, enabling broad use in human pose estimation[265,266], body temperature monitoring[267], sweat analysis[268], indoor positioning[313], and motion recognition[269]. As shown in Figure 11C, Zhou et al. developed a ultra high frequency (UHF) RFID-based battery-free multi-parameter sensor that simultaneously monitors body temperature, pressure, and humidity (e.g., from sweat or urine)[267]. By forming a BAN, it continuously tracks physiological signals such as respiration and ballistocardiogram (BCG), offering a promising solution for long-term monitoring of bedridden patients. As shown in Figure 11D, Sun et al. proposed a projection stereolithography (PSL) approach to rapidly pattern a photocurable, silver-flake hydrogel into customizable antenna geometries, and demonstrated flexible RFID tags by integrating the printed antennas with commercial RFID chips for passive, battery-free wireless sensing (e.g., eye-movement recognition via RFID readout)[270].
Based on magnetically coupled resonance, resonant coupling technology enables efficient wireless power transfer and two-way communication over medium distances. Chipless resonant sensing schemes further extend this concept by eliminating the on-device integrated circuit (IC) and encoding sensing information directly in the spectral signature of patterned resonators. It features high penetration, low power consumption, and good biocompatibility, making it suitable for various physiological monitoring applications such as wound infection tracking[271], blood monitoring[272,273], intraocular pressure sensing[274], biomarker detection in body fluids[275,276], battery-free human-computer interaction[277], and limb motion perception[278,279]. RC technology also holds great potential in implantable devices[280-282]. As presented in Figure 11E, Lin et al. realized a battery-free, biodegradable pressure sensor using a radio-frequency inductor-capacitor (LC) circuit[271]. Combined with a tough hydrogel bio-adhesive layer, the system enabled long-term stable wireless monitoring of intracranial pressure in rats and fully degraded in vivo, eliminating the need for surgical removal. As shown in Figure 11F, Li et al. developed a wireless immunoassay[314]. With a relay unit to expand vector network analyzer (VNA) multi-frequency detection, the system uses hydrogel swelling from antigen recognition to shift resonant frequency, allowing for rapid (< 10 min), ultrasensitive (fg/L level) detection of multiple airborne viruses.
System integration technologies
Building on the active and passive strategies above, system-level integration becomes the key path to next-generation wearable devices. Active and passive communication technologies offer diverse data transmission options for wearable sensors. Traditional wearable designs use a modular approach with independent sensing, power, and communication units. This often leads to bulky and inefficient systems. Deeper system integration is the key path to developing next-generation wearable devices. It goes beyond simply combining individual parts and involves a comprehensive co-design of sensing, power, and communication units from the perspective of overall efficiency. This process drives the evolution of wireless sensing systems toward more integrated, networked, and multifunctional advanced forms.
By utilizing the inherent physical properties of signals during transmission and interaction, ISAC enables a single hardware platform or signal stream to perform both sensing and communication functions simultaneously[315-317]. This integration significantly improves system simplicity, energy efficiency, and compactness. Song et al. developed a full textile-based body-coupled electrical stimulation (BCES) system that uses the human body as a medium to harvest stray electromagnetic energy and static electricity for wireless muscle activation [Figure 12A][318]. With conductive threads knitted into a sock design, the device generates electric fields to stimulate muscle fibers, combining wireless energy transmission, battery-free operation, and physiological performance enhancement in one textile structure. Xie et al. proposed a triboelectric-discharge-based self-powered wireless sensing system for smart-home applications [Figure 12B], in which low-frequency mechanical motion triggers discharge to generate high-frequency wireless signals; information is stabilized/encoded via capacitance-based multi-frequency modulation and transmitted through a coil-enabled wireless link over > 5 m[319]. As a complementary battery-free paradigm at the system level, passive surface acoustic wave (SAW) delay-line/resonator sensors can be wirelessly interrogated by an RF reader, where measurands are encoded in the echo response (e.g., time delay/phase or resonance features), highlighting a communication–sensing co-integration characteristic[320-322].
Figure 12. Novel system integration technology in wearable devices. (A) Concept of BCES for wearable bioelectronics. Reproduced with permission[318]. Copyright 2024, Springer Nature Limited; (B) Schematic illustration of the SWSS based on F-TENG[319]. Copyright 2024, OAE Publishing Inc.; (C) Wireless networking of implantable devices with wearable metamaterials. Reproduced with permission[327]. Copyright 2023, Springer Nature Limited; (D) Coaxially-shielded textile metamaterial enabled robust near-field BAN. Reproduced with permission[325]. Copyright 2024, Springer Nature Limited; (E) Continuously operating wearable device for powering and communicating with WBF NFC-capable implantable electronics. Reproduced with permission[330]. Copyright 2025, The American Association for the Advancement of Science; (F) An energy-efficient flexible multi-modal wireless sweat sensing system[331]. Copyright 2023, Licensee MDPI, Basel, Switzerland. BCES: Body-coupled electrical stimulation; SWSS: self-powered wireless sensing system; F-TENG: freestanding-mode triboelectric nanogenerator; NBR: nitrile butadiene rubber; LIG: laser-induced graphene; PTFE: polytetrafluoroethylene; BAN: body area network; WBF: wireless, battery-free; NFC: near field communication; ISAC: integration of communication and sensing functions; PFA: perfluoroalkoxy; IMCTs: integration of multiple communication technologies; BLE: Bluetooth low energy.
When multiple wearable or implantable devices need to work together within the body area, BANs become a key solution for system integration[323]. BANs are designed to create a communication network around the human body that connects these miniaturized sensor nodes, enabling data collection and centralized processing[324-326]. Tian et al. introduced metamaterial textiles that enable direct communication between implants by guiding radio frequency signals non-radiatively along the body surface [Figure 12C][327]. This approach reduces signal loss in biological tissues, improving received signal strength by over 30 dB. Zhu et al. proposed a textile BAN resilient in high-sweat and aquatic environments [Figure 12D][325]. By confining electric fields with embroidered coaxial metamaterials, it maintains spectral stability and uses magneto-inductive waves to power multiple passive sensors for continuous monitoring.
Real-world applications are complex, and no single communication technology performs optimally in all scenarios[328]. This has led to integrated strategies that combine multiple technologies for greater adaptability and robustness[329]. These systems intelligently select or switch communication methods based on transmission distance, data rate, and power requirements. As shown in Figure 12E, Kasper et al. developed a wireless “bio-symbiotic hub” that combines far-field RF (915 MHz) for self-charging, NFC (13.56 MHz) to power and communicate with battery-free implants such as bone sensors, and BLE for external data exchange[330]. By integrating far-field charging and near-field communication, this system enables months-long, continuous monitoring of bone health without user intervention. Feng et al. proposed an energy-efficient flexible multimodal sweat sensing system based on laser-induced graphene [Figure 12F], where sweat biomarker data are forwarded through a relay using NFC and BLE to enable practical wireless connectivity to the host device[331].
Application progress
Real world case studies have increasingly demonstrated the practical feasibility of wearable sensing technologies beyond controlled laboratory environments. In recent years, several representative studies have reported continuous and long-term monitoring of physiological signals in daily life scenarios, such as chronic disease management, postoperative rehabilitation, and home-based health assessment. For example, the T-Sens sEMG system developed by a wearable sensing company enables flexible electrode placement on various body parts, such as the arms or legs, to detect bioelectric potentials generated by muscle cells, thereby facilitating biomechanical analysis of human motion[332]. In addition, TAION Apparel (Nagoya, Japan) has introduced the Vital Belt, a belt-type wearable sensing device that pioneers the use of millimeter-wave sensing at the waist position[333]. This system has demonstrated the capability to detect stair usage by elderly individuals and trigger alerts when necessary, as well as to monitor fetal activity and intestinal conditions. Furthermore, Samsung Electronics (Suwon, Republic of Korea) has released the Galaxy Ring, which integrates an accelerometer, optical bio-signal sensors, and skin temperature sensors[334]. This ring enables real-time monitoring of sleep quality and nocturnal skin temperature and provides insights into cardiovascular health through continuous heart rate tracking. Although large-scale clinical validation remains limited, these real-world examples provide important evidence supporting the translational potential of wearable sensors from laboratory prototypes to practical clinical and healthcare applications.
CONCLUSION AND OUTLOOK
In summary, we reviewed recent advances in wearable sensors for health monitoring, in terms of the material properties, sensing principles, and wireless communication technologies. With the progress in stretchable materials and fabrication methods, wearable sensors have obtained excellent electrical and mechanical performance under complex human motion, while self-healing systems extend service life and enhance reliability in harsh environments. Materials with excellent biocompatibility and breathability further ensure comfort and safety during long-term continuous wear. Given the heterogeneity of sensing tasks and operating scenarios, composites that balance mechanical robustness, electrical stability, and bio-interface compatibility represent a rational pathway; nevertheless, interfacial adhesion and nonuniform component dispersion remain significant bottlenecks. Although state-of-the-art sensors achieve high sensitivity and accurate analyte detection, multimodal monitoring inherently collects heterogeneous signals, making efficient signal decoupling and cross-interference suppression more challenging. Fully integrated, multifunctional devices that fuse physical and biochemical sensing units are still scarce, and their broad commercialization will require sustained research and engineering investment. Meanwhile, wireless wearable technologies are propelling healthcare monitoring toward further miniaturization, and low-power operation. Progress in flexible circuit integration, passive sensor design, and system-level co-optimization helps balance comfort and functionality; however, practical hurdles persist, including limited communication stability, deformation-induced antenna attenuation, and enduring trade-offs between power consumption and battery life.
To address these challenges, interfacial interactions among disparate constituents can be strengthened via graft copolymerization and surface functionalization (e.g., introducing hydroxyl or amino groups), while coupling agents serve as molecular “bridges” to mitigate interfacial defects. Advanced fabrication techniques - such as in situ polymerization, electrospinning, and 3D printing - facilitate uniform dispersion and ordered arrangement of multicomponent systems, thereby preserving the substrate’s stretchability while ensuring the continuity of the conductive network. For heterogeneous-signal decoupling and crosstalk suppression in multimodal sensing, dedicated readout channels can be designed for distinct sensing units, electromagnetic shielding can be implemented to reduce interference, and both electrode architecture and transduction mechanisms can be optimized to minimize non-target responses. From the perspective of wireless communication, signal quality can be improved by optimizing flexible circuitry and antenna design; integrating energy harvesting with intelligent power management extends operational lifetime; and hybrid communication architectures that adapt to diverse scenarios can be developed to enhance robustness. Through system-level co-optimization across sensing, power, and communication, robust and long-term health monitoring can be realized, thereby advancing personalized healthcare.
The clinical demand driving the development of wearable sensors has become increasingly explicit and urgent. With the growing burden of chronic disease management, population aging, and the rise of personalized medicine, clinicians are seeking technologies capable of continuous, noninvasive, real-time, and high-precision monitoring to overcome the limitations of traditional intermittent medical assessments. For instance, long-term management of conditions such as cardiovascular disease, hypertension, diabetes, and metabolic syndrome requires monitoring at a frequency far beyond conventional outpatient visits. In parallel, in-hospital monitoring systems are expected to become more lightweight, wireless, and patient-friendly to minimize discomfort and improve compliance. Furthermore, the clinical need for multimodal physiological data - including mechanical, biochemical, and electrophysiological signals - continues to grow, enabling more comprehensive pathophysiological analysis. Consequently, the next generation of wearable sensors should be designed to address key clinical challenges in precise diagnosis, telemedicine, personalized interventions, and early-stage disease warning. This evolution will shift wearable systems from simple data acquisition tools toward integrated intelligent platforms capable of supporting clinical decision-making, thereby enabling more proactive and continuous healthcare delivery. Moreover, deep interdisciplinary collaboration among materials science, electrical engineering, and clinical medicine - guided by clinical needs to refine performance metrics - will be essential to ensure both practicality and clinical value of next-generation wearable sensors.
DECLARATIONS
Authors’ contributions
Designed the original draft: Li, S.; Liang, J. G.
Wrote the original draft: Li, S.; Kong, C.; Wu, E.
Reviewed and revised the manuscript: Kong, C.; Wu, E.; Yang, C.; Gao, Z.
Provided comments and suggestions on the revision of the manuscript: Zhou, X.; Guo, H.; Jiang, S.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant Nos. 62504023, 62504024, 62206030), the Natural Science Foundation of Jiangsu Province (Grant No. BK20230629), the Leading Innovative Talents Introduction and Cultivation Project of Changzhou (Grant No. CQ20220110), the Scientific Research Foundation of the Jiangsu Provincial Education Department (Grant No. 23KJB510001), the Research and Innovation Project of the Compound Semiconductor Innovation Consortium (Grant No. RIPCSIC202503), the Postgraduate Research & Practice Innovation Program of Jiangsu Province (Grant No. KYCX25_3377), and the Jiangsu Students’ Platform for Innovation and Entrepreneurship Training Program (Grant Nos. X202510292225, S202510292083).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Yang, X.; Chen, W.; Fan, Q.; et al. Electronic skin for health monitoring systems: properties, functions, and applications. Adv. Mater. 2024, 36, e2402542.
2. Liang, X.; Zhang, S.; Meng, S.; Tan, R.; Zhang, K.; Hu, J. A non-enzymatic flexible and wearable sensor based on thermal transfer printing technology for continuous glucose detection in sweat. Microchem. J. 2024, 207, 111690.
3. Zulkifli, N. A.; Jeong, W.; Kim, M.; et al. 3D-printed magnetic-based air pressure sensor for continuous respiration monitoring and breathing rehabilitation. Soft. Sci. 2024, 4, 20.
4. Ali, M.; Hoseyni, S. M.; Das, R.; Awais, M.; Basdogan, I.; Beker, L. A flexible and biodegradable piezoelectric-based wearable sensor for non-invasive monitoring of dynamic human motions and physiological signals. Adv. Mater. Technol. 2023, 8, 2300347.
5. Yang, Y.; Yang, S.; Xia, X.; et al. MXenes for wearable physical sensors toward smart healthcare. ACS. Nano. 2024, 18, 24705-40.
6. Zhou, Y.; Wang, Y.; Qiu, S.; et al. Microscopic response mechanism of epsilon-negative and epsilon-near-zero metacomposites. Research 2025, 8, 0556.
7. Ma, Z.; Li, S.; Wang, H.; et al. Advanced electronic skin devices for healthcare applications. J. Mater. Chem. B. 2019, 7, 173-97.
8. Jung, Y.; Kim, M.; Jeong, S.; Hong, S.; Ko, S. H. Strain-insensitive outdoor wearable electronics by thermally robust nanofibrous radiative cooler. ACS. Nano. 2024, 18, 2312-24.
9. Lu, Y.; Ma, L.; Zhang, H.; Mei, Y.; Xiong, Z.; Song, E. Soft wearable electronics for evaluation of biological tissue mechanics. Soft. Sci. 2024, 4, 36.
10. Yeo, W. H.; Kim, Y. S.; Lee, J.; et al. Multi-functional electronics: multifunctional epidermal electronics printed directly onto the skin (Adv. Mater. 20/2013). Adv. Mater. 2013, 25, 2772.
11. Li, X.; Fan, Y. J.; Li, H. Y.; et al. Ultracomfortable hierarchical nanonetwork for highly sensitive pressure sensor. ACS. Nano. 2020, 14, 9605-12.
12. Sabry, F.; Eltaras, T.; Labda, W.; Alzoubi, K.; Malluhi, Q. Machine learning for healthcare wearable devices: the big picture. J. Healthc. Eng. 2022, 2022, 4653923.
13. Ghadi, Y. Y.; Shah, S. F. A.; Waheed, W.; et al. Integration of wearable technology and artificial intelligence in digital health for remote patient care. J. Cloud. Comp. 2025, 14, 39.
14. Pan, Y.; Wang, X.; Sun, Y.; Qiu, S.; Zhou, Y.; Huang, Y. Flexible and printed electronics manufacturing in China. Flex. Print. Electron. 2025, 10, 043001.
15. Yuan, J.; Zhao, W.; Zhou, Y.; Huang, Y. Flexible sensor-driven smart vehicles: opportunities and prospects. CHAIN 2025, 2, 227-35.
16. Kang, S.; Cho, S.; Shanker, R.; et al. Transparent and conductive nanomembranes with orthogonal silver nanowire arrays for skin-attachable loudspeakers and microphones. Sci. Adv. 2018, 4, eaas8772.
17. Wang, Y.; Zhao, C.; Wang, J.; et al. Wearable plasmonic-metasurface sensor for noninvasive and universal molecular fingerprint detection on biointerfaces. Sci. Adv. 2021, 7, eabe4553.
18. Min, S.; Kim, D. H.; Joe, D. J.; et al. Clinical validation of a wearable piezoelectric blood-pressure sensor for continuous health monitoring. Adv. Mater. 2023, 35, e2301627.
19. Yang, M.; Peng, K.; Li, Z.; et al. Recent progress in flexible materials for wearable devices for body function and athletic performance monitoring. Chem. Eng. J. 2025, 505, 159659.
20. Tan, C.; Dong, Z.; Li, Y.; et al. A high performance wearable strain sensor with advanced thermal management for motion monitoring. Nat. Commun. 2020, 11, 3530.
21. Babu, R. S.; Vinodh, R.; Raja, R.; et al. Nanohybrid materials in flexible and wearable supercapacitors. In Hybrid nanostructures for advanced supercapacitors; Modern Aspects of Electrochemistry, Vol 63; Springer, Cham, 2026; pp. 215-49.
22. Yu, S.; Deng, W.; Wu, X.; et al. Fully printed organic thin-film transistors: pathways to scalable, high-performance flexible electronics. Chem. Soc. Rev. 2026, 55, 567-604.
23. Gao, W.; Huang, J.; He, J.; et al. Recent advances in ultrathin materials and their applications in e-skin. InfoMat 2023, 5, e12426.
24. Zhao, W.; Natsuki, J.; Dinh Trung, V.; et al. AgNPs/CNTs modified nonwoven fabric for PET-based flexible interdigitated electrodes in pressure sensor applications. Chem. Eng. J. 2024, 499, 156252.
25. Liu, X.; Ma, Y.; Dai, X.; Li, S.; Li, B.; Zhang, X. Flexible pressure sensor based on Pt/PI network with high sensitivity and high thermal resistance. Chem. Eng. J. 2024, 494, 152996.
26. Zhang, T.; Chai, Y.; Wang, S.; et al. Recent study advances in flexible sensors based on polyimides. Sensors 2023, 23, 9743.
27. Li, S.; Chu, H.; Li, S.; Wang, X.; Qiu, T.; Yan, H. Synthesis of a breathable and degradable silk fibroin/sodium alginate/aniline tetramer electronic skin by in situ polymerization method and for human motion monitoring. Compos. Commun. 2025, 56, 102419.
28. Liang, A.; Liu, W.; Yao, M.; et al. Bioinspired dual-mode self-powered paper sensor for sustainable wearable health monitoring via synergistic humidity gradient and triboelectric effects. Langmuir 2025, 41, 27512-24.
29. Huang, P.; Cai, Y.; Jia, Q.; Yang, Y.; Yue, T.; Zhong, S. High-performance piezoresistive sensor based on indium tin oxide nanoparticles/cellulose nanofiber composite for human activity monitoring. Compos. Commun. 2025, 59, 102544.
30. Chen, G.; Matsuhisa, N.; Liu, Z.; et al. Plasticizing silk protein for on-skin stretchable electrodes. Adv. Mater. 2018, 30, e1800129.
31. Zhang, Y.; Wang, S.; Gao, Y.; et al. Bioinspired durable mechanical-bioelectrical dual-modal sensors enabled by mixed ion-electron conduction and mechanical interlocking for multifunctional sensing. Adv. Funct. Mater. 2025, 35, 2501122.
32. Guan, S.; Xu, C.; Dong, X.; Qi, M. A highly tough, fatigue-resistant, low hysteresis hybrid hydrogel with a hierarchical cross-linked structure for wearable strain sensors. J. Mater. Chem. A. 2023, 11, 15404-15.
33. He, J.; Zhou, R.; Zhang, Y.; et al. Strain-insensitive self-powered tactile sensor arrays based on intrinsically stretchable and patternable ultrathin conformal wrinkled graphene-elastomer composite. Adv. Funct. Mater. 2022, 32, 2107281.
34. Wang, J.; Ochiai, Y.; Wu, N.; et al. Intrinsically stretchable organic photovoltaics by redistributing strain to PEDOT:PSS with enhanced stretchability and interfacial adhesion. Nat. Commun. 2024, 15, 4902.
35. Ma, Z.; Khoo, B. L. Recent advances in laser-induced-graphene-based soft skin electronics for intelligent healthcare. Soft. Sci. 2024, 4, 26.
36. Li, S.; Wu, J.; Lyu, H.; et al. An artificial spiking afferent nerve based on synaptic transistor for thermal perception. IEEE. Electron. Dev. Lett. 2024, 45, 64-7.
37. Han, S. H.; Shin, J. H.; Yang, H. J.; Nam, S.; Park, J. Y.; Choi, S. S. Optical visualization of stretchable serpentine interconnects using chiral liquid crystal elastomers. Adv. Sci. 2024, 12, e2408346.
38. Shi, Y.; Zhao, J.; Zhang, B.; et al. Freestanding serpentine silicon strips with ultrahigh stretchability over 300% for wearable electronics. Adv. Mater. 2024, 36, e2313603.
39. Zhang, M.; Shahsavan, H.; Guo, Y.; Pena-Francesch, A.; Zhang, Y.; Sitti, M. Liquid-crystal-elastomer-actuated reconfigurable microscale kirigami metastructures. Adv. Mater. 2021, 33, e2008605.
40. Hao, X. P.; Xu, Z.; Li, C. Y.; Hong, W.; Zheng, Q.; Wu, Z. L. Kirigami-design-enabled hydrogel multimorphs with application as a multistate switch. Adv. Mater. 2020, 32, e2000781.
41. Lee, S.; Song, Y.; Ko, Y.; et al. A metal-like conductive elastomer with a hierarchical wrinkled structure. Adv. Mater. 2020, 32, e1906460.
42. Zhao, Y.; Gao, W.; Dai, K.; et al. Bioinspired multifunctional photonic-electronic smart skin for ultrasensitive health monitoring, for visual and self-powered sensing. Adv. Mater. 2021, 33, e2102332.
43. Yan, Z.; Liu, Y.; Xiong, J.; et al. Hierarchical serpentine-helix combination for 3D stretchable electronics. Adv. Mater. 2023, 35, e2210238.
44. Liu, H.; Li, H.; Wang, Z.; et al. Robust and multifunctional Kirigami electronics with a tough and permeable aramid nanofiber framework. Adv. Mater. 2022, 34, e2207350.
45. Nisa, F. U.; Tahir, M.; Khalid, S.; et al. Supramolecular cross-linking enables highly stretchable and ultrasensitive polyurethane-poly(3,4-ethylenedioxythiophene) tactile sensors. ACS. Nano. 2025, 19, 35102-18.
46. Wang, S.; Rong, H.; Lu, X. High-performance solid-state ion-conductive elastomer based on multiple noncovalent interactions for flexible sensors. Chem. Eng. J. 2025, 505, 159776.
47. He, S.; Zhang, Y.; Gao, J.; Nag, A.; Rahaman, A. Integration of different graphene nanostructures with PDMS to form wearable sensors. Nanomaterials 2022, 12, 950.
48. Yu, Y.; Peng, S.; Blanloeuil, P.; Wu, S.; Wang, C. H. Wearable temperature sensors with enhanced sensitivity by engineering microcrack morphology in PEDOT:PSS-PDMS sensors. ACS. Appl. Mater. Interfaces. 2020, 12, 36578-88.
49. Zhong, L.; Tian, X.; Wang, J. Y.; et al. Calibration-free optical waveguide bending sensor for soft robots. Soft. Sci. 2025, 5, 3.
50. Masihi, S.; Panahi, M.; Maddipatla, D.; et al. Highly sensitive porous PDMS-based capacitive pressure sensors fabricated on fabric platform for wearable applications. ACS. Sens. 2021, 6, 938-49.
51. Li, S.; Fu, Q.; Pan, C. A multi-functional wearable sensor based on carbon nanomaterials reinforced TPU fiber with high sensitivity. J. Alloys. Compd. 2022, 927, 167041.
52. Rana, S. M. S.; Faruk, O.; Reza, M. S.; Islam, M. R.; Kim, H.; Park, J. Y. All porous Ecoflex and SEBS-based stretchable high-performance triboelectric nanogenerator for self-powered human activity monitoring. Chem. Eng. J. 2024, 488, 151050.
53. Shrestha, K.; Pradhan, G. B.; Asaduzzaman, M.; et al. A breathable, reliable, and flexible siloxene incorporated porous SEBS-based triboelectric nanogenerator for human–machine interactions. Adv. Energy. Mater. 2024, 14, 2302471.
54. Kim, S. H.; Chung, S.; Kim, M.; et al. Designing a length-modulated azide photocrosslinker to improve the stretchability of semiconducting polymers. Adv. Funct. Mater. 2023, 33, 2212127.
55. Xia, H.; Zhang, W.; Miao, C.; et al. Ultra-thin amphiphilic hydrogel electrolyte for flexible zinc-ion paper batteries. Energy. Environ. Sci. 2024, 17, 6507-20.
56. Qiu, Y.; Wang, Y. L.; Tang, W. J.; et al. An ultra-thin transparent multi-functional sensor based on silk hydrogel for health monitoring. J. Micromech. Microeng. 2022, 32, 084003.
57. Roy, A.; Afshari, R.; Jain, S.; et al. Advances in conducting nanocomposite hydrogels for wearable biomonitoring. Chem. Soc. Rev. 2025, 54, 2595-652.
58. Pan, B.; Su, P.; Jin, M.; et al. Ultrathin hierarchical hydrogel-carbon nanocomposite for highly stretchable fast-response water-proof wearable humidity sensors. Mater. Horiz. 2023, 10, 5263-76.
59. Wu, K.; Li, J.; Li, Y.; et al. 3D printed silk fibroin-based hydrogels with tunable adhesion and stretchability for wearable sensing. Adv. Funct. Mater. 2024, 34, 2404451.
60. Tropp, J.; Collins, C. P.; Xie, X.; et al. Conducting polymer nanoparticles with intrinsic aqueous dispersibility for conductive hydrogels. Adv. Mater. 2024, 36, e2306691.
61. He, J.; Li, A.; Wang, W.; et al. Multifunctional wearable device based on an antibacterial and hydrophobic silver nanoparticles/Ti3C2Tx MXene/thermoplastic polyurethane fibrous membrane for electromagnetic shielding and strain sensing. Ind. Eng. Chem. Res. 2023, 62, 9221-32.
62. Chen, S.; Li, S.; Zheng, Y.; et al. Highly sensitive and robust soft tri-axial tactile sensors enabled by dual inductive sensing mechanisms. Soft. Sci. 2025, 5, 6.
63. Kim, S.; Choi, H. K.; Song, Y. S.; et al. Omni-directional assembly of 2D single-crystalline metal nanosheets. Adv. Mater. 2025, 37, e2501632.
64. Singh, M.; Zakria, M.; Pannu, A. S.; et al. Defect-free, few-atomic-layer thin ZnO nanosheets with superior excitonic properties for optoelectronic devices. ACS. Nano. 2024, 18, 16947-57.
65. Gogurla, N.; Kim, Y.; Cho, S.; Kim, J.; Kim, S. Multifunctional and ultrathin electronic tattoo for on-skin diagnostic and therapeutic applications. Adv. Mater. 2021, 33, e2008308.
66. Alam, M. N.; Kumar, V.; Lee, D. J.; Choi, J. Synergistically toughened silicone rubber nanocomposites using carbon nanotubes and molybdenum disulfide for stretchable strain sensors. Compos. Part. B. Eng. 2023, 259, 110759.
67. Jo, H. S.; Kwon, H. J.; Kim, T. G.; et al. Wearable transparent thermal sensors and heaters based on metal-plated fibers and nanowires. Nanoscale 2018, 10, 19825-34.
68. Lim, T.; Kim, H. J.; Won, S.; et al. Liquid metal-based electronic textiles coated with au nanoparticles as stretchable electrode materials for healthcare monitoring. ACS. Appl. Nano. Mater. 2023, 6, 8482-94.
69. Jiang, D.; Zhu, Y.; Sun, Z.; et al. A silver nanowires@Prussian blue composite aerogel-based wearable sensor for noninvasive and dynamic monitoring of sweat uric acid. Chem. Eng. J. 2024, 486, 150220.
70. Lin, M.; Zheng, Z.; Yang, L.; et al. A high-performance, sensitive, wearable multifunctional sensor based on rubber/CNT for human motion and skin temperature detection. Adv. Mater. 2022, 34, e2107309.
71. Gao, Z.; Xiao, X.; Carlo, A. D.; et al. Advances in wearable strain sensors based on electrospun fibers. Adv. Funct. Mater. 2023, 33, 2214265.
72. Cao, Y.; Zhang, K.; Huang, Z.; Li, S. A unique and robust physically unclonable function based on bionic tunable ion gel-gated synaptic transistors. IEEE. Electron. Device. Lett. 2023, 44, 1995-8.
73. Fan, W.; Liu, T.; Wu, F.; et al. An antisweat interference and highly sensitive temperature sensor based on poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate) fiber coated with polyurethane/graphene for real-time monitoring of body temperature. ACS. Nano. 2023, 17, 21073-82.
74. Zheng, X.; Wang, S.; Ding, B.; et al. Ultrathin elastomer nanofiber-based skin-like electronic with multi-mode sensing capabilities. Small. Methods. 2025, 9, e01064.
75. Zhao, Y.; Wang, F.; Liu, J.; et al. Underwater self-healing and recyclable ionogel sensor for physiological signal monitoring. ACS. Appl. Mater. Interfaces. 2023, 15, 28664-74.
76. Zhu, M.; Xu, W.; Chen, L.; et al. Ultrathin self-healing nanofibrous membrane with a hierarchical confined structure for biomimetic epidermal electrodes. ACS. Nano. 2024, 18, 28834-48.
77. Li, S.; Lyu, H.; Zhou, Y.; et al. Artificial reflex arc: an environment-adaptive neuromorphic camouflage device. IEEE. Electron. Device. Lett. 2021, 42, 1224-7.
78. Zhang, D.; Tang, Y.; Zhang, Y.; et al. Highly stretchable, self-adhesive, biocompatible, conductive hydrogels as fully polymeric strain sensors. J. Mater. Chem. A. 2020, 8, 20474-85.
79. Shi, C.; Zou, Z.; Lei, Z.; Zhu, P.; Zhang, W.; Xiao, J. Heterogeneous integration of rigid, soft, and liquid materials for self-healable, recyclable, and reconfigurable wearable electronics. Sci. Adv. 2020, 6, eabd0202.
80. Guo, X.; Zhang, S.; Patel, S.; et al. A skin-mimicking multifunctional hydrogel via hierarchical, reversible noncovalent interactions. Sci. Adv. 2025, 11, eadv8523.
81. Li, Y.; Fang, T.; Zhang, J.; et al. Ultrasensitive and ultrastretchable electrically self-healing conductors. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2300953120.
82. He, X.; Wang, H.; Wang, J.; et al. Liquid metal/poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) composite hydrogels with multi-stage gauge factors, pressure resistance, and wide pH stability. ACS. Appl. Polym. Mater. 2025, 7, 11713-22.
83. Li, S. Y.; Zhao, K. Y.; Zan, G. T.; et al. Liquid metal-enabled energy generators for self-powered soft bioelectronics. Rare. Met. 2025, 44, 9297-335.
84. Wei, J.; Chen, H.; Pan, F.; et al. Reusable liquid metal-based hierarchical hydrogels with multifunctional sensing capability for electrophysiology electrode substitution. ACS. Nano. 2025, 19, 15554-64.
85. Wang, S.; Fan, P.; Liu, W.; et al. Research progress of flexible electronic devices based on electrospun nanofibers. ACS. Nano. 2024, 18, 31737-72.
86. Li, X.; He, L.; Li, Y.; et al. Healable, degradable, and conductive MXene nanocomposite hydrogel for multifunctional epidermal sensors. ACS. Nano. 2021, 15, 7765-73.
87. Chen, Y.; Pu, X.; Liu, M.; et al. Shape-adaptive, self-healable triboelectric nanogenerator with enhanced performances by soft solid-solid contact electrification. ACS. Nano. 2019, 13, 8936-45.
88. An, S.; Kim, T.; Jang, J.; et al. Self-bondable and strain-durable electroceuticals using self-healing and stretchable conducting nanocomposite trilayer for effective vagus nerve stimulation. ACS. Nano. 2025, 19, 33091-102.
89. Zeng, X.; Teng, L.; Wang, X.; et al. Efficient multi-phsysical crosslinked nanocomposite hydrogel for a conformal strain and self-powered tactile sensor. Nano. Energy. 2025, 135, 110669.
90. Kim, K. H.; Kim, J. H.; Ko, Y. J.; Lee, H. E. Body-attachable multifunctional electronic skins for bio-signal monitoring and therapeutic applications. Soft. Sci. 2024, 4, 24.
91. Li, S.; Lyu, H.; Li, J.; et al. Multiterminal ionic synaptic transistor with artificial blink reflex function. IEEE. Electron. Device. Lett. 2021, 42, 351-4.
92. Du, Y.; Kim, J. H.; Kong, H.; et al. Biocompatible electronic skins for cardiovascular health monitoring. Adv. Healthc. Mater. 2024, 13, e2303461.
93. Jiang, Y.; Zhang, Z.; Wang, Y. X.; et al. Topological supramolecular network enabled high-conductivity, stretchable organic bioelectronics. Science 2022, 375, 1411-7.
94. Zhang, Z.; Weng, L.; Guo, K.; Guan, L.; Wang, X.; Wu, Z. Durable and highly sensitive flexible sensors for wearable electronic devices with PDMS-MXene/TPU composite films. Ceram. Int. 2022, 48, 4977-85.
95. Amjadi, M.; Yoon, Y. J.; Park, I. Ultra-stretchable and skin-mountable strain sensors using carbon nanotubes-Ecoflex nanocomposites. Nanotechnology 2015, 26, 375501.
96. Jamatia, T.; Matyas, J.; Olejnik, R.; et al. Wearable and stretchable SEBS/CB polymer conductive strand as a piezoresistive strain sensor. Polymers 2023, 15, 1618.
97. Zhang, Q.; Zhang, H.; Liang, J.; et al. Ti3C2Tx-MXene/PET textile-based flexible pressure sensor for wearable pulse monitoring. J. Mater. Chem. C. 2023, 11, 15638-48.
98. Gao, F. L.; Min, P.; Gao, X. Z.; et al. Integrated temperature and pressure dual-mode sensors based on elastic PDMS foams decorated with thermoelectric PEDOT:PSS and carbon nanotubes for human energy harvesting and electronic-skin. J. Mater. Chem. A. 2022, 10, 18256-66.
99. Dang, X.; Fu, Y.; Wang, X. A temperature and pressure dual-responsive, stretchable, healable, adhesive, and biocompatible carboxymethyl cellulose-based conductive hydrogels for flexible wearable strain sensor. Biosens. Bioelectron. 2024, 246, 115893.
100. Chen, X.; Xia, X.; Guo, C. F. Flexible iontronic sensing: ionic materials, electrodes, and encapsulation. Adv. Funct. Mater. 2026, 36, e12920.
101. Wang, X.; Huang, J.; Liu, Y.; et al. Design of protective and high sensitivity encapsulation layers in wearable devices. Sci. China. Technol. Sci. 2023, 66, 223-32.
102. Gai, Y.; Wang, E.; Liu, M.; et al. A self-powered wearable sensor for continuous wireless sweat monitoring. Small. Methods. 2022, 6, e2200653.
103. Chung, H. U.; Rwei, A. Y.; Hourlier-Fargette, A.; et al. Skin-interfaced biosensors for advanced wireless physiological monitoring in neonatal and pediatric intensive-care units. Nat. Med. 2020, 26, 418-29.
104. Brasier, N.; Niederberger, C.; Salvatore, G. A. The sweat rate as a digital biomarker in clinical medicine beyond sports science. Soft. Sci. 2024, 4, 6.
105. Li, W.; Zou, K.; Guo, J.; et al. Integrated fibrous iontronic pressure sensors with high sensitivity and reliability for human plantar pressure and gait analysis. ACS. Nano. 2024, 18, 14672-84.
106. Chen, S.; Guo, Z.; Lu, B.; et al. A wearable device for continuous immunoassay-based monitoring of C-peptide in interstitial fluid. Sci. Adv. 2025, 11, eadw2182.
107. Tang, X.; Qi, C.; Sun, Q. Recent progress of biosensors based on thermoelectric effects for monitoring physical activity and environment monitoring. Soft. Sci. 2025, 5, 11.
108. Chung, H. U.; Kim, B. H.; Lee, J. Y.; et al. Binodal, wireless epidermal electronic systems with in-sensor analytics for neonatal intensive care. Science 2019, 363, eaau0780.
109. Li, Z.; Chen, F.; Zhu, N.; Zhang, L.; Xie, Z. Tip-enhanced sub-femtomolar steroid immunosensing via micropyramidal flexible conducting polymer electrodes for at-home monitoring of salivary sex hormones. ACS. Nano. 2023, 17, 21935-46.
110. Chen, S.; Ouyang, Q.; Meng, X.; et al. Starfish-inspired wearable bioelectronic systems for physiological signal monitoring during motion and real-time heart disease diagnosis. Sci. Adv. 2025, 11, eadv2406.
111. Choi, Y.; Jin, P.; Lee, S.; et al. All-printed chip-less wearable neuromorphic system for multimodal physicochemical health monitoring. Nat. Commun. 2025, 16, 5689.
112. Declercq, D.; Peremans, L.; Glorieus, M.; et al. Urinary sodium/creatinine ratio is a predictor for fractional sodium excretion and related to age in patients with cystic fibrosis. J. Cyst. Fibros. 2022, 21, e136-40.
113. Calvo-López, A.; Arasa-Puig, E.; Alonso-Chamarro, J.; Puyol, M. Serum/plasma potassium monitoring using potentiometric point-of-care microanalyzers with improved ion selective electrodes. Talanta 2023, 253, 124100.
114. Krishnan, S. S.; Lennox, A. T.; Welter, J.; Dozor, A. J. Relationship between sweat chloride and pulmonary function in healthy young adults - a single-center, pilot study. Respir. Med. 2025, 245, 108177.
115. Zhou, Y.; Nan, F.; Li, Z.; et al. High-performance graphdiyne oxide/Au nanoparticle electrode for electrochemical non-enzymatic glucose sensor. ACS. Nano. 2025, 19, 25262-72.
116. Wu, Z. Q.; Cao, X. Q.; Hua, Y.; Yu, C. M. A bifunctional wearable sensor based on a nanoporous membrane for simultaneous detection of sweat lactate and temperature. Anal. Chem. 2024, 96, 3087-95.
117. Song, H.; Shin, H.; Seo, H.; et al. Wireless non-invasive monitoring of cholesterol using a smart contact lens. Adv. Sci. 2022, 9, e2203597.
118. Liu, Y.; Li, X.; Yang, H.; et al. Skin-interfaced superhydrophobic insensible sweat sensors for evaluating body thermoregulation and skin barrier functions. ACS. Nano. 2023, 17, 5588-99.
119. Kang, M.; Jeong, H.; Park, S. W.; et al. Wireless graphene-based thermal patch for obtaining temperature distribution and performing thermography. Sci. Adv. 2022, 8, eabm6693.
120. Li, F.; Lin, X.; Xue, H.; et al. Ultrasensitive flexible temperature sensors based on thermal-mediated ions migration dynamics in asymmetrical polymer bilayers. ACS. Nano. 2024, 18, 7521-31.
121. Song, L.; Chen, J.; Xu, B. B.; Huang, Y. Flexible plasmonic biosensors for healthcare monitoring: progress and prospects. ACS. Nano. 2021, 15, 18822-47.
122. Zhong, S.; Lu, B.; Wang, D. C.; et al. Passive isothermal flexible sensor enabled by smart thermal-regulating aerogels. Adv. Mater. 2025, 37, e2415386.
123. Yang, Y. F.; Hao, J. M.; Zhang, W.; et al. High-precision adaptive temperature control performance of thin positive temperature coefficient materials with ultra-high resistance-temperature coefficient. Appl. Therm. Eng. 2024, 253, 123767.
124. Zhang, Z.; Li, Q.; Xu, L.; Tian, W.; Li, Z. High-performance flexible temperature sensors based on laser-irradiated Ag-MWCNTs/PEDOT:PSS. ACS. Appl. Mater. Interfaces. 2024, 16, 6078-87.
125. Li, Z.; He, B.; Li, Y.; et al. Synergizing nanosensor-enhanced wearable devices with machine learning for precision health management benefiting older adult populations. ACS. Nano. 2025, 19, 26273-95.
126. Yu, S.; Wang, S.; Zhao, L.; Shi, S.; Wang, L.; Zheng, H. Ultra-flexible transparent temperature sensor with laser-direct writing of silver nanowire electrodes. Inorg. Chem. Commun. 2024, 170, 113219.
127. Santos, M. S. C. S.; Reis, J. C. R. Surface tension of liquid mixtures and metal alloys. Positive and negative temperature coefficients in alloys with remarkably high surface density. J. Alloys. Compd. 2023, 939, 168791.
128. Choi, Y.; Kim, M.; So, H. Negative temperature coefficient effect of TPU/SWCNT/PEDOT:PSS polymer matrices for wearable temperature sensors. Polym. Test. 2024, 141, 108652.
129. Zhang, W.; Li, L.; Wang, Y.; et al. Continuous core body temperature monitoring for heatstroke alert via a wearable in-ear thermometer. ACS. Sens. 2025, 10, 1440-9.
130. Van Nguyen, D.; Song, P.; Manshaii, F.; Bell, J.; Chen, J.; Dinh, T. Advances in soft strain and pressure sensors. ACS. Nano. 2025, 19, 6663-704.
131. Zhou, Y.; Wang, S.; Yin, J.; et al. Flexible metasurfaces for multifunctional interfaces. ACS. Nano. 2024, 18, 2685-707.
132. Ha, K. H.; Huh, H.; Li, Z.; Lu, N. Soft capacitive pressure sensors: trends, challenges, and perspectives. ACS. Nano. 2022, 16, 3442-8.
133. Li, R.; Zhou, Q.; Bi, Y.; et al. Research progress of flexible capacitive pressure sensor for sensitivity enhancement approaches. Sens. Actuators. A. Phys. 2021, 321, 112425.
134. Ha, K. H.; Zhang, W.; Jang, H.; et al. Highly sensitive capacitive pressure sensors over a wide pressure range enabled by the hybrid responses of a highly porous nanocomposite. Adv. Mater. 2021, 33, e2103320.
135. Cao, M.; Su, J.; Fan, S.; Qiu, H.; Su, D.; Li, L. Wearable piezoresistive pressure sensors based on 3D graphene. Chem. Eng. J. 2021, 406, 126777.
136. Ji, F.; Sun, Z.; Hang, T.; et al. Flexible piezoresistive pressure sensors based on nanocellulose aerogels for human motion monitoring: a review. Compos. Commun. 2022, 35, 101351.
137. Kim, S. W.; Lee, J. H.; Ko, H. J.; et al. Mechanically robust and linearly sensitive soft piezoresistive pressure sensor for a wearable human-robot interaction system. ACS. Nano. 2024, 18, 3151-60.
138. Bijender; Kumar, A. Recent progress in the fabrication and applications of flexible capacitive and resistive pressure sensors. Sens. Actuators. A. Phys. 2022, 344, 113770.
139. Nassar, H.; Khandelwal, G.; Chirila, R.; et al. Fully 3D printed piezoelectric pressure sensor for dynamic tactile sensing. Addit. Manuf. 2023, 71, 103601.
140. Hou, S.; Chen, X.; Assi, D. S.; et al. Tactile-transparent wearable sensor for clinician-friendly pulse wave velocity monitoring and cardiovascular risk profiling. ACS. Nano. 2025, 19, 32822-35.
141. Shi, W.; Jang, S.; Kuss, M. A.; et al. Digital light processing 4D printing of poloxamer micelles for facile fabrication of multifunctional biocompatible hydrogels as tailored wearable sensors. ACS. Nano. 2024, 18, 7580-95.
142. Qin, J.; Yin, L. J.; Hao, Y. N.; et al. Flexible and stretchable capacitive sensors with different microstructures. Adv. Mater. 2021, 33, e2008267.
143. Han, R.; Liu, Y.; Mo, Y.; et al. High anti-jamming flexible capacitive pressure sensors based on core–shell structured AgNWs@TiO2. Adv. Funct. Mater. 2023, 33, 2305531.
144. Xing, D.; Wang, Z.; Liu, S.; et al. Microtetrahedral electrode-enhanced wearable capacitive dual-mode sensor for integrated tactile pressure and proximity detection. ACS. Sens. 2026, 11, 371-83.
145. Ruan, X.; Zhao, Y.; Zhu, X.; et al. Biomimetic flexible capacitive sensor with loop electrode and snail tentacle structure for enhanced proximity and pressure sensing. ACS. Appl. Mater. Interfaces. 2025, 17, 32936-48.
146. Xu, J.; Zeng, W.; Luo, X.; et al. Noncontact perception of thin-film transistors by the synergy of both capacitive and electrostatic induction mechanisms. ACS. Appl. Mater. Interfaces. 2024, 16, 54154-62.
147. Huang, J.; Wang, H.; Li, J.; et al. High-performance flexible capacitive proximity and pressure sensors with spiral electrodes for continuous human–machine interaction. ACS. Materials. Lett. 2022, 4, 2261-72.
148. Zhou, Z.; Yuan, W. Functionally integrated conductive organohydrogel sensor for wearable motion detection, triboelectric nanogenerator and non-contact sensing. Compos. Part. A. Appl. Sci. Manuf. 2023, 172, 107603.
149. Xiong, Y.; Han, J.; Wang, Y.; Wang, Z. L.; Sun, Q. Emerging iontronic sensing: materials, mechanisms, and applications. Research 2022, 2022, 9867378.
150. Cao, S.; Huo, Z.; Chen, Y.; Guo, L.; Huang, C.; Xiao, X. A supercapacitive iontronic sensor using woven dielectric of surface-grooved fibers. IEEE. Sens. J. 2025, 25, 6087-94.
151. Tahir, M.; He, L.; Haider, W. A.; et al. Co-electrodeposited porous PEDOT-CNT microelectrodes for integrated micro-supercapacitors with high energy density, high rate capability, and long cycling life. Nanoscale 2019, 11, 7761-70.
152. Muzaffar, A.; Ahamed, M. B.; Deshmukh, K.; Thirumalai, J. A review on recent advances in hybrid supercapacitors: design, fabrication and applications. Renew. Sustain. Energy. Rev. 2019, 101, 123-45.
153. Yoon, H.; Ko, S.; Chhetry, A.; et al. Ultra-sensitive and quick-responsive hybrid-supercapacitive iontronic pressure sensor for intuitive electronics and artificial tactile applications. Adv. Mater. Technol. 2022, 7, 2101743.
154. Jin, Y.; Xue, S.; He, Y. Flexible pressure sensors enhanced by 3D-printed microstructures. Adv. Mater. 2025, 37, e2500076.
155. Meng, K.; Xiao, X.; Wei, W.; et al. Wearable pressure sensors for pulse wave monitoring. Adv. Mater. 2022, 34, e2109357.
156. Kim, M. K.; Kantarcigil, C.; Kim, B.; et al. Flexible submental sensor patch with remote monitoring controls for management of oropharyngeal swallowing disorders. Sci. Adv. 2019, 5, eaay3210.
157. Wang, Q.; Guan, H.; Wang, C.; et al. A wireless, self-powered smart insole for gait monitoring and recognition via nonlinear synergistic pressure sensing. Sci. Adv. 2025, 11, eadu1598.
158. Huang, W.; Xu, Y.; Wang, Z.; Liao, K.; Zhang, Y.; Sun, Y. Dual nanozyme based on ultrathin 2D conductive MOF nanosheets intergraded with gold nanoparticles for electrochemical biosensing of H2O2 in cancer cells. Talanta 2022, 249, 123612.
159. Zhang, J.; Luan, Y.; Ma, Q.; et al. Large-area grown ultrathin molybdenum oxides for label-free sensitive biomarker detection. Nanoscale 2024, 16, 13061-70.
160. Yan, X.; Li, H.; Yin, T.; Jie, G.; Zhou, H. Photoelectrochemical biosensing platform based on in situ generated ultrathin covalent organic framework film and AgInS2 QDs for dual target detection of HIV and CEA. Biosens. Bioelectron. 2022, 217, 114694.
161. Cheng, L.; Hao, X.; Liu, G.; et al. A flexible pressure sensor based on silicon nanomembrane. Biosensors 2023, 13, 131.
162. Li, S.; Yang, C.; Jia, L.; et al. A bio-inspired neuromorphic pressure afferent nerve based on fully inkjet-printed synaptic transistor. Appl. Phys. Lett. 2025, 127, 063301.
163. Yuan, J.; Li, Z.; Zhao, Y.; et al. Skin-adaptive focused flexible micromachined ultrasound transducers for wearable cardiovascular health monitoring. Sci. Adv. 2025, 11, eadw7632.
164. Kil, H. J.; Park, J. W. Carotid artery monitoring patch using a supercapacitive pressure sensor with piezoelectricity. Nano. Energy. 2023, 114, 108636.
165. Choi, J.; Ghaffari, R.; Baker, L. B.; Rogers, J. A. Skin-interfaced systems for sweat collection and analytics. Sci. Adv. 2018, 4, eaar3921.
166. Sim, D.; Brothers, M. C.; Slocik, J. M.; et al. Biomarkers and detection platforms for human health and performance monitoring: a review. Adv. Sci. 2022, 9, e2104426.
167. Duan, H.; Peng, S.; He, S.; et al. Wearable electrochemical biosensors for advanced healthcare monitoring. Adv. Sci. 2025, 12, e2411433.
168. Manjakkal, L.; Yin, L.; Nathan, A.; Wang, J.; Dahiya, R. Energy autonomous sweat-based wearable systems. Adv. Mater. 2021, 33, e2100899.
169. Wang, T.; Liu, T.; Zhu, X.; et al. High-entropy Prussian blue analogues-mediated flexible field-effect transistor for wearable sweat dopamine monitoring. Biosens. Bioelectron. 2025, 289, 117889.
170. Childs, A.; Mayol, B.; Lasalde-Ramírez, J. A.; Song, Y.; Sempionatto, J. R.; Gao, W. Diving into sweat: advances, challenges, and future directions in wearable sweat sensing. ACS. Nano. 2024, 18, 24605-16.
171. Gao, F.; Liu, C.; Zhang, L.; et al. Wearable and flexible electrochemical sensors for sweat analysis: a review. Microsyst. Nanoeng. 2023, 9, 1.
172. Li, S.; Ma, Z.; Cao, Z.; Pan, L.; Shi, Y. Advanced wearable microfluidic sensors for healthcare monitoring. Small 2020, 16, e1903822.
173. Ma, X.; Wang, P.; Huang, L.; et al. A monolithically integrated in-textile wristband for wireless epidermal biosensing. Sci. Adv. 2023, 9, eadj2763.
174. Son, J.; Bae, G. Y.; Lee, S.; et al. Cactus-spine-inspired sweat-collecting patch for fast and continuous monitoring of sweat. Adv. Mater. 2021, 33, e2102740.
175. Sun, Y.; Wang, J.; Lu, Q.; et al. Stretchable and smart wettable sensing patch with guided liquid flow for multiplexed in situ perspiration analysis. ACS. Nano. 2024, 18, 2335-45.
176. Qu, M.; Shen, L.; Wang, J.; et al. Superhydrophobic, humidity-resistant, and flexible triboelectric nanogenerators for biomechanical energy harvesting and wearable self-powered sensing. ACS. Appl. Nano. Mater. 2022, 5, 9840-51.
177. Yue, X.; Xu, F.; Zhang, L.; et al. Simple, skin-attachable, and multifunctional colorimetric sweat sensor. ACS. Sens. 2022, 7, 2198-208.
178. Li, F.; Shen, N.; Zhang, L.; et al. Skin-conformal multifunctional microfluidic colorimetric detection device for multiplexed sweat analysis. ACS. Sens. 2025, 10, 5968-80.
179. Kim, J.; Wu, Y.; Luan, H.; et al. A skin-interfaced, miniaturized microfluidic analysis and delivery system for colorimetric measurements of nutrients in sweat and supply of vitamins through the skin. Adv. Sci. 2022, 9, e2103331.
180. Cho, S. H.; Cho, S.; Lv, Z.; et al. Soft, wearable, microfluidic system for fluorometric analysis of loss of amino acids through eccrine sweat. Lab. Chip. 2025, 25, 1647-55.
181. Vinoth, R.; Nakagawa, T.; Mathiyarasu, J.; Mohan, A. M. V. Fully printed wearable microfluidic devices for high-throughput sweat sampling and multiplexed electrochemical analysis. ACS. Sens. 2021, 6, 1174-86.
182. Zhang, Y.; Sun, Y.; Han, J.; Zhang, M.; Li, F.; Yang, D. High-sensitivity flexible electrochemical sensor for real-time multi-analyte sweat analysis. Talanta 2025, 287, 127644.
183. Wang, X.; He, A.; Yu, B.; et al. Uncovering the sweat biofouling components and distributions in electrochemical sensors. Anal. Chem. 2022, 94, 14402-9.
184. Choi, J.; Bandodkar, A. J.; Reeder, J. T.; et al. Soft, skin-integrated multifunctional microfluidic systems for accurate colorimetric analysis of sweat biomarkers and temperature. ACS. Sens. 2019, 4, 379-88.
185. Shi, H.; Cao, Y.; Zeng, Y.; et al. Wearable tesla valve-based sweat collection device for sweat colorimetric analysis. Talanta 2022, 240, 123208.
186. Choi, D. H.; Gonzales, M.; Kitchen, G. B.; Phan, D. T.; Searson, P. C. A capacitive sweat rate sensor for continuous and real-time monitoring of sweat loss. ACS. Sens. 2020, 5, 3821-6.
187. Zhong, B.; Jiang, K.; Wang, L.; Shen, G. Wearable sweat loss measuring devices: from the role of sweat loss to advanced mechanisms and designs. Adv. Sci. 2022, 9, e2103257.
188. Jang, J.; Kim, J.; Shin, H.; et al. Smart contact lens and transparent heat patch for remote monitoring and therapy of chronic ocular surface inflammation using mobiles. Sci. Adv. 2021, 7, eabf7194.
189. Tian, Y.; Cui, Q.; Xu, L.; et al. Alloyed AuPt nanoframes loaded on h-BN nanosheets as an ingenious ultrasensitive near-infrared photoelectrochemical biosensor for accurate monitoring glucose in human tears. Biosens. Bioelectron. 2021, 192, 113490.
190. Moreddu, R.; Elsherif, M.; Adams, H.; et al. Integration of paper microfluidic sensors into contact lenses for tear fluid analysis. Lab. Chip. 2020, 20, 3970-9.
191. Zhou, F.; Zhao, H.; Chen, K.; Cao, S.; Shi, Z.; Lan, M. Flexible electrochemical sensor with Fe/Co bimetallic oxides for sensitive analysis of glucose in human tears. Anal. Chim. Acta. 2023, 1243, 340781.
192. Mukundan, G.; Badhulika, S. Nickel-cobalt metal-organic frameworks based flexible hydrogel as a wearable contact lens for electrochemical sensing of urea in tear samples. Mikrochim. Acta. 2024, 191, 252.
193. Chen, Z.; Liu, Y.; Yu, W.; Liu, S.; Huang, Y. Machine learning-driven wearable sweat sensors with AgNW/MXene for non-invasive SERS-based cardiovascular disease detection. ACS. Appl. Nano. Mater. 2025, 8, 5602-10.
194. Park, S.; Hwang, J.; Jeon, H. J.; et al. Cerium oxide nanoparticle-containing colorimetric contact lenses for noninvasively monitoring human tear glucose. ACS. Appl. Nano. Mater. 2021, 4, 5198-210.
195. Wang, Z.; Dong, Y.; Sui, X.; et al. An artificial intelligence-assisted microfluidic colorimetric wearable sensor system for monitoring of key tear biomarkers. npj. Flex. Electron. 2024, 8, 35.
196. Park, J.; Kim, J.; Kim, S. Y.; et al. Soft, smart contact lenses with integrations of wireless circuits, glucose sensors, and displays. Sci. Adv. 2018, 4, eaap9841.
197. Ku, M.; Kim, J.; Won, J. E.; et al. Smart, soft contact lens for wireless immunosensing of cortisol. Sci. Adv. 2020, 6, eabb2891.
198. Yin, S.; Chen, X.; Li, R.; Sun, L.; Yao, C.; Li, Z. Wearable, biocompatible, and dual-emission ocular multisensor patch for continuous profiling of fluoroquinolone antibiotics in tears. ACS. Nano. 2024, 18, 18522-33.
199. Paqué, P. N.; Herz, C.; Jenzer, J. S.; et al. Microbial analysis of saliva to identify oral diseases using a point-of-care compatible qPCR assay. J. Clin. Med. 2020, 9, 2945.
200. Santonocito, R.; Cavallaro, A.; Pappalardo, A.; et al. Detection of human salivary stress biomarkers using an easy-to-use array sensor based on fluorescent organic molecules. Biosens. Bioelectron. 2025, 270, 116986.
201. Gu, M.; Zhong, Y.; Hu, J.; et al. Bio-inspired nanoengineered wood for scalable monolithic gas sensor fabrication. Adv. Mater. 2025, 37, e2507829.
202. García-Carmona, L.; Martín, A.; Sempionatto, J. R.; et al. Pacifier biosensor: toward noninvasive saliva biomarker monitoring. Anal. Chem. 2019, 91, 13883-91.
203. Sun, M.; Pei, X.; Xin, T.; et al. A flexible microfluidic chip-based universal fully integrated nanoelectronic system with point-of-care raw sweat, tears, or saliva glucose monitoring for potential noninvasive glucose management. Anal. Chem. 2022, 94, 1890-900.
204. Bi, Y.; Sun, M.; Wang, J.; et al. Universal fully integrated wearable sensor arrays for the multiple electrolyte and metabolite monitoring in raw sweat, saliva, or urine. Anal. Chem. 2023, 95, 6690-9.
205. Bordbar, M. M.; Hosseini, M. S.; Sheini, A.; et al. Monitoring saliva compositions for non-invasive detection of diabetes using a colorimetric-based multiple sensor. Sci. Rep. 2023, 13, 16174.
206. Chi, Y. J.; Ryu, B.; Ahn, S.; Koh, W. G. A colorimetric biosensor based on a biodegradable fluidic device capable of efficient saliva sampling and salivary biomarker detection. Sens. Actuators. B. Chem. 2023, 396, 134601.
207. Tu, J.; Yeom, J.; Ulloa, J. C.; et al. Stressomic: a wearable microfluidic biosensor for dynamic profiling of multiple stress hormones in sweat. Sci. Adv. 2025, 11, eadx6491.
208. Tang, W.; Yin, L.; Sempionatto, J. R.; Moon, J. M.; Teymourian, H.; Wang, J. Touch-based stressless cortisol sensing. Adv. Mater. 2021, 33, e2008465.
209. Lou, C.; Yang, H.; Hou, Y.; et al. Microfluidic platforms for real-time in situ monitoring of biomarkers for cellular processes. Adv. Mater. 2024, 36, e2307051.
210. Cho, S.; Won, C.; Kwon, C.; et al. Smart bioelectronic nanomesh face masks with permeability and flexibility for monitoring cortisol in saliva. ACS. Sens. 2025, 10, 148-58.
211. Kim, J.; Imani, S.; de Araujo, W. R.; et al. Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics. Biosens. Bioelectron. 2015, 74, 1061-8.
212. Lim, H. R.; Lee, S. M.; Park, S.; et al. Smart bioelectronic pacifier for real-time continuous monitoring of salivary electrolytes. Biosens. Bioelectron. 2022, 210, 114329.
213. Zhou, S.; Park, G.; Lin, M.; Yang, X.; Xu, S. Wearable ultrasound technology. Nat. Rev. Bioeng. 2025, 3, 835-54.
214. Huang, H.; Wu, R. S.; Lin, M.; Xu, S. Emerging wearable ultrasound technology. IEEE. Trans. Ultrason. Ferroelectr. Freq. Control. 2024, 71, 713-29.
215. Amado-Rey, A. B.; Goncalves Seabra, A. C.; Stieglitz, T. Towards ultrasound wearable technology for cardiovascular monitoring: from device development to clinical validation. IEEE. Rev. Biomed. Eng. 2025, 18, 93-112.
216. Dutta, A.; Niu, Z.; Abdullah, A. M.; et al. Closely packed stretchable ultrasound array fabricated with surface charge engineering for contactless gesture and materials detection. Adv. Sci. 2024, 11, e2303403.
217. Wang, B.; Peng, J.; Li, Y.; et al. A 3D-printed adaptive origami-structured wearable ultrasound transducer for arterial blood pressure monitoring. Measurement 2026, 260, 119815.
218. Zou, F.; Luo, Y.; Zhuang, W.; Xu, T. A fully integrated conformal wearable ultrasound patch for continuous sonodynamic therapy. Adv. Mater. 2024, 36, e2409528.
219. Guo, M.; Xia, Y.; Liu, J.; Zhang, Y.; Li, M.; Wang, X. Wearable pressure sensor based on triboelectric nanogenerator for information encoding, gesture recognition, and wireless real-time robot control. Adv. Funct. Mater. 2025, 35, 2419209.
220. Wan, Z.; Liu, P.; Fu, Y.; et al. Wearable multimodal flexible sensor based on layer-by-layer aerogel/nanofibers composite structure for real-time information encoding and gesture recognition. Chem. Eng. J. 2025, 521, 166455.
221. Zhang, S.; Zhou, H.; Tchantchane, R.; Alici, G. A wearable human–machine-interface (HMI) system based on colocated EMG-pFMG sensing for hand gesture recognition. IEEE/ASME. Trans. Mechatron. 2025, 30, 369-80.
222. Chen, X.; Luo, Y.; Chen, Y.; et al. Biomimetic contact behavior inspired tactile sensing array with programmable microdomes pattern by scalable and consistent fabrication. Adv. Sci. 2024, 11, e2408082.
223. Li, H.; Niu, H.; Yin, F.; Zhou, W.; Shen, G.; Li, Y. A labor-division cooperation electronic palm system for high-precision crosstalk-free cognition of pressure and temperature. Adv. Mater. 2026, 38, e10241.
224. Ates, H. C.; Nguyen, P. Q.; Gonzalez-Macia, L.; et al. End-to-end design of wearable sensors. Nat. Rev. Mater. 2022, 7, 887-907.
225. Mu, G.; Zhang, Y.; Yan, Z.; Yu, Q.; Wang, Q. Recent advancements in wearable sensors: integration with machine learning for human-machine interaction. RSC. Adv. 2025, 15, 7844-54.
226. Ma, C. B.; Shang, X.; Sun, M.; et al. Emerging multifunctional wearable sensors: integrating multimodal sweat analysis and advanced material technologies for next-generation health monitoring. ACS. Sens. 2025, 10, 2388-408.
227. Kong, L.; Li, W.; Zhang, T.; et al. Wireless technologies in flexible and wearable sensing: from materials design, system integration to applications. Adv. Mater. 2024, 36, e2400333.
228. Cao, Q.; Deng, R.; Pan, Y.; et al. Robotic wireless capsule endoscopy: recent advances and upcoming technologies. Nat. Commun. 2024, 15, 4597.
229. Yue, W.; Guo, Y.; Lee, J. C.; et al. Advancements in passive wireless sensing systems in monitoring harsh environment and healthcare applications. Nanomicro. Lett. 2025, 17, 106.
230. Gutruf, P. Towards a digitally connected body for holistic and continuous health insight. Commun. Mater. 2024, 5, 2.
231. Zhang, X.; Chen, J.; Zheng, Z.; et al. Flexible temperature sensor with high reproducibility and wireless closed-loop system for decoupled multimodal health monitoring and personalized thermoregulation. Adv. Mater. 2024, 36, e2407859.
232. Guo, Y.; Yin, F.; Li, Y.; Shen, G.; Lee, J. C. Incorporating wireless strategies to wearable devices enabled by a photocurable hydrogel for monitoring pressure information. Adv. Mater. 2023, 35, e2300855.
233. Dong, H.; Li, X.; Liu, Y.; et al. Wearable, breathable, and wireless gas sensor enables highly selective exhaled ammonia detection and real-time noninvasive illness diagnosis. ACS. Sens. 2025, 10, 3964-75.
234. Yan, M.; Hao, Q.; Diao, S.; et al. Smart home sleep respiratory monitoring system based on a breath-responsive covalent organic framework. ACS. Nano. 2024, 18, 728-37.
235. Kaveh, R.; Schwendeman, C.; Pu, L.; Arias, A. C.; Muller, R. Wireless ear EEG to monitor drowsiness. Nat. Commun. 2024, 15, 6520.
236. Wang, S.; Dong, M.; He, J.; et al. A multi-user wearable waistband system for real-time health monitoring of respiration, ECG, and body temperature. Microelectron. Eng. 2025, 299, 112346.
237. Li, J.; Jia, H.; Zhou, J.; et al. Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure. Nat. Commun. 2023, 14, 5009.
238. Song, Y.; Min, J.; Yu, Y.; et al. Wireless battery-free wearable sweat sensor powered by human motion. Sci. Adv. 2020, 6, eaay9842.
239. Zhao, H.; Bai, J.; Zhang, X.; et al. A fully integrated wearable sweat sensing patch for online analysis of multiple Parkinson’s disease-related biomarkers. Adv. Mater. 2025, 37, e2504534.
240. Alam, M. S.; Kim, J. K.; Choi, J. Multifunctional wearable system for mapping body temperature and analyzing sweat. ACS. Sens. 2023, 8, 1980-8.
241. Jiang, D.; Liu, X.; Zhan, W.; et al. Skin-interfaced wearable sensor for long-term reliable monitoring of uric acid and pH in sweat. Nano. Lett. 2025, 25, 1427-35.
242. Xu, C.; Song, Y.; Sempionatto, J. R.; et al. A physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 2024, 7, 168-79.
243. Lee, J. P.; Jang, H.; Jang, Y.; et al. Encoding of multi-modal emotional information via personalized skin-integrated wireless facial interface. Nat. Commun. 2024, 15, 530.
244. Yoo, J. Y.; Oh, S.; Shalish, W.; et al. Wireless broadband acousto-mechanical sensing system for continuous physiological monitoring. Nat. Med. 2023, 29, 3137-48.
245. Xu, K.; Fujita, Y.; Lu, Y.; et al. A wearable body condition sensor system with wireless feedback alarm functions. Adv. Mater. 2021, 33, e2008701.
246. Li, M.; Li, G.; Xu, T.; et al. Multi-biosensing hairband for emergency health assessment. Nat. Commun. 2025, 16, 7224.
247. Jin, T.; Wang, H.; Ullah, I.; et al. A wireless operated flexible bioelectronic microneedle patch for actively controlled transdermal drug delivery. Adv. Mater. 2025, 37, e2417136.
248. Zhou, Q.; Ding, Q.; Geng, Z.; et al. A flexible smart healthcare platform conjugated with artificial epidermis assembled by three-dimensionally conductive MOF network for gas and pressure sensing. Nanomicro. Lett. 2024, 17, 50.
249. Lin, M.; Zhang, Z.; Gao, X.; et al. A fully integrated wearable ultrasound system to monitor deep tissues in moving subjects. Nat. Biotechnol. 2024, 42, 448-57.
250. Gao, X.; Chen, X.; Lin, M.; et al. A wearable echomyography system based on a single transducer. Nat. Electron. 2024, 7, 1035-46.
251. Zhang, H.; Jin, P.; Pang, K.; Jiang, Y. A wearable, highly sensitive piezoresistive pressure sensor based on MXene/NiMoO4 @CMF and mimicking the hedgehog skin spike structure for its real-time monitoring of human movement. ACS. Appl. Electron. Mater. 2024, 6, 7503-11.
252. Aliyana, A. K.; Yang, D.; Tangsirinaruenart, O.; Stylios, G. K. A garment-integrated textile stitch-based strain sensor device, IoT-Enabled for enhanced wearable sportswear applications. Results. Eng. 2024, 23, 102794.
253. Li, H.; Yang, H.; Lu, F.; et al. Towards adaptive bioelectronic wound therapy with integrated real-time diagnostics and machine learning–driven closed-loop control. npj. Biomed. Innov. 2025, 2, 31.
254. Choi, M.; Lee, S.; Kim, S. W.; et al. Battery‐free, wireless multi‐sensing platform for comprehensive management of pressure injury and hygiene. Adv. Funct. Mater. 2025, 35, e06774.
255. Ge, Z.; Guo, W.; Tao, Y.; et al. Wireless and closed-loop smart dressing for exudate management and on-demand treatment of chronic wounds. Adv. Mater. 2023, 35, e2304005.
256. Cho, S.; Ha, J.; Ahn, J.; et al. Wireless, battery‐free, optoelectronic diagnostic sensor integrated colorimetric dressing for advanced wound care. Adv. Funct. Mater. 2024, 34, 2316196.
257. Escobedo, P.; Fernández-Ramos, M. D.; López-Ruiz, N.; et al. Smart facemask for wireless CO2 monitoring. Nat. Commun. 2022, 13, 72.
258. Xu, L.; Chen, W.; Li, Q.; et al. Printing nacre‐mimetic MXene‐based E‐textile devices for sensing and breathing‐pattern recognition using machine learning. Adv. Funct. Mater. 2025, 35, e08370.
259. Cho, S.; Han, H.; Park, H.; et al. Wireless, multimodal sensors for continuous measurement of pressure, temperature, and hydration of patients in wheelchair. npj. Flex. Electron. 2023, 7, 8.
260. Li, H.; Gong, H.; Wong, T. H.; et al. Wireless, battery-free, multifunctional integrated bioelectronics for respiratory pathogens monitoring and severity evaluation. Nat. Commun. 2023, 14, 7539.
261. Shi, Z.; Deng, P.; Zhou, L. A.; et al. Wireless and battery-free wearable biosensing of riboflavin in sweat for precision nutrition. Biosens. Bioelectron. 2024, 251, 116136.
262. Mirzajani, H.; Abbasiasl, T.; Mirlou, F.; et al. An ultra-compact and wireless tag for battery-free sweat glucose monitoring. Biosens. Bioelectron. 2022, 213, 114450.
263. Park, W.; Seo, H.; Kim, J.; et al. In-depth correlation analysis between tear glucose and blood glucose using a wireless smart contact lens. Nat. Commun. 2024, 15, 2828.
264. Sailapu, S. K.; Liébana, S.; Merino-Jimenez, I.; Esquivel, J. P.; Sabaté, N. Towards a REASSURED reality: a less-is-more electronic design strategy for self-powered glucose test. Biosens. Bioelectron. 2024, 243, 115708.
265. Zhao, H.; Esfahani, M. I. M.; Krishnan, S.; Abari, O. A passive back posture monitoring system using orthogonal RFID design. IEEE. Sens. J. 2024, 24, 35610-20.
266. Liu, J.; Xia, Z.; Liu, J.; Guo, S. RF-care: RFID-based human pose estimation for nursing-care applications under visual occlusion. IEEE. Sens. J. 2024, 24, 35359-66.
267. Zhou, Z.; Wu, H.; Fu, J.; et al. Fully integrated passive wireless sensor with mechanical-electrical double-gradient for multifunctional healthcare monitoring. Nano. Lett. 2024, 24, 14781-9.
268. López-Soriano, S.; Vilajosana, X.; Melià-Seguí, J. A battery-less and low-cost RFID sensor for unassisted multilevel detection of sweat loss. IEEE. Sens. J. 2024, 24, 42028-37.
269. Zhang, H.; Wang, L.; Pei, J.; Lyu, F.; Li, M.; Liu, C. RF-sign: position-independent sign language recognition using passive RFID tags. IEEE. Internet. Things. J. 2024, 11, 9056-71.
270. Sun, Y.; Cui, J.; Feng, S.; et al. Projection stereolithography 3D printing high-conductive hydrogel for flexible passive wireless sensing. Adv. Mater. 2024, 36, e2400103.
271. Lin, J.; Chen, X.; Zhang, P.; et al. Wireless bioelectronics for in vivo pressure monitoring with mechanically-compliant hydrogel biointerfaces. Adv. Mater. 2024, 36, e2400181.
272. Xu, Y.; Ye, Z.; Zhao, G.; et al. Phase-separated porous nanocomposite with ultralow percolation threshold for wireless bioelectronics. Nat. Nanotechnol. 2024, 19, 1158-67.
273. Kwon, K.; Kim, J. U.; Won, S. M.; et al. A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature. Nat. Biomed. Eng. 2023, 7, 1215-28.
274. Yang, C.; Wu, Q.; Liu, J.; et al. Intelligent wireless theranostic contact lens for electrical sensing and regulation of intraocular pressure. Nat. Commun. 2022, 13, 2556.
275. Liu, T. L.; Dong, Y.; Chen, S.; Zhou, J.; Ma, Z.; Li, J. Battery-free, tuning circuit-inspired wireless sensor systems for detection of multiple biomarkers in bodily fluids. Sci. Adv. 2022, 8, eabo7049.
276. Kim, J.; Kim, M.; Lee, M. S.; et al. Wearable smart sensor systems integrated on soft contact lenses for wireless ocular diagnostics. Nat. Commun. 2017, 8, 14997.
277. Zhu, H.; Yang, H.; Xu, S.; et al. Frequency-encoded eye tracking smart contact lens for human-machine interaction. Nat. Commun. 2024, 15, 3588.
278. Wu, E.; Liang, J.; Kim, N.; et al. Wireless passive flexible radio frequency tactile sensor for material recognition. Nano. Lett. 2025, 25, 7960-8.
279. Galli, V.; Sailapu, S. K.; Cuthbert, T. J.; Ahmadizadeh, C.; Hannigan, B. C.; Menon, C. Passive and wireless all-textile wearable sensor system. Adv. Sci. 2023, 10, e2206665.
280. Yousefi Darestani, M. R.; Lange, D.; Chew, B. H.; Takahata, K. Electromechanically functionalized ureteral stents for wireless obstruction monitoring. ACS. Biomater. Sci. Eng. 2023, 9, 4392-403.
281. Nan, K.; Wong, K.; Li, D.; et al. An ingestible, battery-free, tissue-adhering robotic interface for non-invasive and chronic electrostimulation of the gut. Nat. Commun. 2024, 15, 6749.
282. Woods, J. E.; Alrashdan, F.; Chen, E. C.; et al. Distributed battery-free bioelectronic implants with improved network power transfer efficiency via magnetoelectrics. Nat. Biomed. Eng. 2025.
283. Kim, H.; Rigo, B.; Wong, G.; Lee, Y. J.; Yeo, W. H. Advances in wireless, batteryless, implantable electronics for real-time, continuous physiological monitoring. Nanomicro. Lett. 2023, 16, 52.
284. Kim, C. Y.; Lee, J.; Jeong, E. Y.; et al. Wireless technologies for wearable electronics: a review. Adv. Elect. Mater. 2025, 11, 2400884.
285. Inman, A.; Parker, T.; Zhang, Y.; Saraf, M.; Gogotsi, Y. Obtaining a practical wearable supercapacitor power supply. Adv. Energy. Mater. 2024, 14, 2402367.
286. Zhao, D.; Li, Z.; Xu, D.; Yang, Z. Multiple redox‐active cyano‐substituted organic compound integrated with MXene nanosheets for high‐performance flexible aqueous Zn‐ion battery. Adv. Funct. Mater. 2024, 34, 2316182.
287. Yang, L.; Feng, B.; Zhang, Y.; et al. Single wire capacitive wireless power transfer system for wearable biomedical sensors based on flexible graphene film material. IEEE. Trans. Biomed. Circuits. Syst. 2022, 16, 1337-47.
288. Park, W. Y.; Han, J.; Moon, J.; et al. Mechanically robust self-organized crack-free nanocellular graphene with outstanding electrochemical properties in sodium ion battery. Adv. Mater. 2024, 36, e2311792.
289. Liang, X.; Lin, Z.; Lin, Z.; Luo, Q.; Liang, W.; Chen, C. A sulfur-infiltrated mesoporous silica/CNT composite-based functional interlayer for enhanced Li–S battery performance. Appl. Phys. Lett. 2024, 125, 053903.
290. Han, X.; Cao, Y.; Liu, Y. Y.; et al. Liquid gallium-assisted pyrolysis of MOF affording CNT non-hollow frameworks in high yields for high-performance sodium-ion battery anode. Adv. Mater. 2024, 36, e2407274.
291. Manjakkal, L.; Pullanchiyodan, A.; Yogeswaran, N.; Hosseini, E. S.; Dahiya, R. A wearable supercapacitor based on conductive PEDOT:PSS-coated cloth and a sweat electrolyte. Adv. Mater. 2020, 32, e1907254.
292. Hong, Y.; Jia, K.; Zhang, Y.; et al. Energetic and durable all-polymer aqueous battery for sustainable, flexible power. Nat. Commun. 2024, 15, 9539.
293. Li, W.; Lin, K.; Chen, L.; Yang, D.; Ge, Q.; Wang, Z. Self-powered wireless flexible ionogel wearable devices. ACS. Appl. Mater. Interfaces. 2023, 15, 14768-76.
294. Zhao, C.; Wang, R.; Liang, H.; et al. Autonomous self-healing strategy for flexible fiber lithium-ion battery with ultra-high mechanical properties and volumetric energy densities. Chem. Eng. J. 2024, 496, 154153.
295. Wu, R.; Wang, R.; Liu, S.; Wang, M.; Mo, R. Interface-engineered self-healing quasi-solid fiber battery with high energy density and robust mechanical properties. Adv. Fiber. Mater. 2025, 7, 1998-2012.
296. Wang, R.; Wu, R.; Fang, B.; Liang, H.; Mo, R. Interface‐engineered strategy on carbon nanotubes to chemical stabilize graphene as a self‐healing fiber electrode for superior capacitive deionization. Energy. Environ. Mater. 2025, e70141.
297. Li, C.; Ye, Q.; Wang, J.; et al. Ultrathin and capacity-tunable lithium metal wires for lithium-based fiber batteries. Natl. Sci. Rev. 2025, 12, nwae480.
298. Wu, R.; Wei, Y.; Wang, R.; Liu, S.; Mo, R. Nano-interfacial design of self-healable high-strength electrodes with high desalination efficiency and cycling stability. Chem. Eng. J. 2025, 525, 170242.
299. Yan, W.; Ma, C.; Cai, X.; Sun, Y.; Zhang, G.; Song, W. Self-powered and wireless physiological monitoring system with integrated power supply and sensors. Nano. Energy. 2023, 108, 108203.
300. Zhao, T.; Fu, Y.; Sun, C.; et al. Wearable biosensors for real-time sweat analysis and body motion capture based on stretchable fiber-based triboelectric nanogenerators. Biosens. Bioelectron. 2022, 205, 114115.
301. Liang, L.; Liu, X.; Li, P.; et al. A wearable multimodal health monitoring bracelet powered by high-power-density flexible thermoelectric generators. Device 2025, 3, 100748.
302. Yuan, J.; Zhang, Y.; Wei, C.; Zhu, R. A fully self-powered wearable leg movement sensing system for human health monitoring. Adv. Sci. 2023, 10, e2303114.
303. Ding, S.; Saha, T.; Yin, L.; et al. A fingertip-wearable microgrid system for autonomous energy management and metabolic monitoring. Nat. Electron. 2024, 7, 788-99.
304. Chen, Y.; Wan, X.; Li, G.; Ye, J.; Gao, J.; Wen, D. Metal hydrogel‐based integrated wearable biofuel cell for self‐powered epidermal sweat biomarker monitoring. Adv. Funct. Mater. 2024, 34, 2404329.
305. Dong, L.; Zhao, C.; Han, C.; Yang, Y.; Yang, F. Advancement of AI-assisted self-powered healthcare sensing systems. MedMat 2025, 2, 55-77.
306. Kim, S. H.; Basir, A.; Avila, R.; et al. Strain-invariant stretchable radio-frequency electronics. Nature 2024, 629, 1047-54.
307. Shen, Q.; Jiang, M.; Wang, R.; et al. Liquid metal-based soft, hermetic, and wireless-communicable seals for stretchable systems. Science 2023, 379, 488-93.
308. Liu, Y.; De Mulatier, S.; Matsuhisa, N. Unperceivable designs of wearable electronics. Adv. Mater. 2025, 37, e2502727.
309. Li, W.; Li, F.; Tang, W.; et al. Stretchable antenna arrays‐enabled multi‐target balloon wireless communication system. Adv. Funct. Mater. 2025, 36, e10521.
310. Guo, X.; Liu, Y.; Zhou, Z.; et al. Direct-ink-writing printed stretchable eutectic gallium-indium antenna for robust wireless communication. Adv. Sci. 2025, 12, e2414285.
311. Wang, Y. Emerging epidermal electrodes towards digital health and on-skin digitalization. Soft. Sci. 2024, 4, 5.
312. Bhatia, A.; Hanna, J.; Stuart, T.; Kasper, K. A.; Clausen, D. M.; Gutruf, P. Wireless battery-free and fully implantable organ interfaces. Chem. Rev. 2024, 124, 2205-80.
313. Pillai, A. S.; Sudhakar, S.; Benny, S.; Sahoo, S.; Chandran, A.; Peethambharan, S. K. Perforated PVP encapsulated AgNWs for high mass loading in silver nanowire inks for printed RFID integrated wearable smart bands. J. Mater. Chem. C. 2024, 12, 575-92.
314. Li, X.; Sun, R.; Pan, J.; et al. Rapid and on-site wireless immunoassay of respiratory virus aerosols via hydrogel-modulated resonators. Nat. Commun. 2024, 15, 4035.
315. Gu, H.; Lu, B.; Gao, Z.; et al. A battery‐free wireless tactile sensor for multimodal force perception. Adv. Funct. Mater. 2024, 34, 2410661.
316. Wang, H.; Xia, X.; Fu, J.; et al. A general self-powered wireless sensing solution based on triboelectric-discharge effect. Nano. Energy. 2023, 105, 107982.
317. Si, J.; Yang, J.; Sun, D.; et al. Breakdown discharge effect enabled self-powered multi-mechanism wireless sensing scheme. Nano. Energy. 2025, 135, 110671.
318. Song, M.; Moon, J.; Yong, H.; et al. Full textile-based body-coupled electrical stimulation for wireless, battery-free, and wearable bioelectronics. npj. Flex. Electron. 2024, 8, 76.
319. Xie, B.; Lin, S.; Guo, Y.; et al. Triboelectric discharge based self-powered wireless sensing system for smart home application. Soft. Sci. 2025, 5, 54.
320. Pan, Y.; Yan, C.; Gao, X.; et al. A passive wireless surface acoustic wave (SAW) sensor system for detecting warfare agents based on fluoroalcohol polysiloxane film. Microsyst. Nanoeng. 2024, 10, 4.
321. Gao, X.; Xue, X.; Hu, F.; et al. Wireless and passive SAW-based humidity sensor employing SiO2 thin film. IEEE. Sens. J. 2023, 23, 9936-42.
322. Liang, X.; Zhang, L.; Tan, Q.; et al. Temperature, pressure, and humidity SAW sensor based on coplanar integrated LGS. Microsyst. Nanoeng. 2023, 9, 110.
323. Niu, S.; Matsuhisa, N.; Beker, L.; et al. A wireless body area sensor network based on stretchable passive tags. Nat. Electron. 2019, 2, 361-8.
324. Ye, F.; Hajiaghajani, A.; Zargari, A.; et al. Passive wireless body joint‐monitoring networks with textile‐integrated, strongly coupled magnetic resonators. Adv. Elect. Mater. 2025, 11, 2400450.
325. Zhu, X.; Wu, K.; Xie, X.; Anderson, S. W.; Zhang, X. A robust near-field body area network based on coaxially-shielded textile metamaterial. Nat. Commun. 2024, 15, 6589.
326. Mirlou, F.; Bardakci, E.; Abbasiasl, T.; Beker, L. A wireless reader for detecting frequency shift of passive sensors. Biosens. Bioelectron. X. 2025, 26, 100668.
327. Tian, X.; Zeng, Q.; Kurt, S. A.; et al. Implant-to-implant wireless networking with metamaterial textiles. Nat. Commun. 2023, 14, 4335.
328. Alqaderi, A. I. J.; Ramakrishnan, N. Carbon-based flexible strain sensors: recent advances and performance insights in human motion detection. Chem. Eng. J. 2025, 513, 162609.
329. Sim, J. H.; Chae, H.; Kim, S. B.; et al. Wireless organic light-emitting diode contact lenses for on-eye wearable light sources and their application to personalized health monitoring. ACS. Nano. 2025, 19, 17478-89.
330. Kasper, K. A.; Romero, G. F.; Perez, D. L.; et al. Continuous operation of battery-free implants enables advanced fracture recovery monitoring. Sci. Adv. 2025, 11, eadt7488.
331. Feng, J.; Jiang, Y.; Wang, K.; et al. An energy-efficient flexible multi-modal wireless sweat sensing system based on laser induced graphene. Sensors 2023, 23, 4818.
332. Wearable Sensing. T-Sens: surface electromyogram (sEMG). https://wearablesensing.com/t-sens-semg/. (accessed 2026-03-03).
333. TRENDHUNTER. Belt-based wearable sensors: vital belt. https://www.trendhunter.com/trends/vital-belt. (accessed 2026-03-03).
334. Samsung. Explore Galaxy Ring. https://www.samsung.com/us/rings/galaxy-ring/. (accessed 2026-03-03).
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






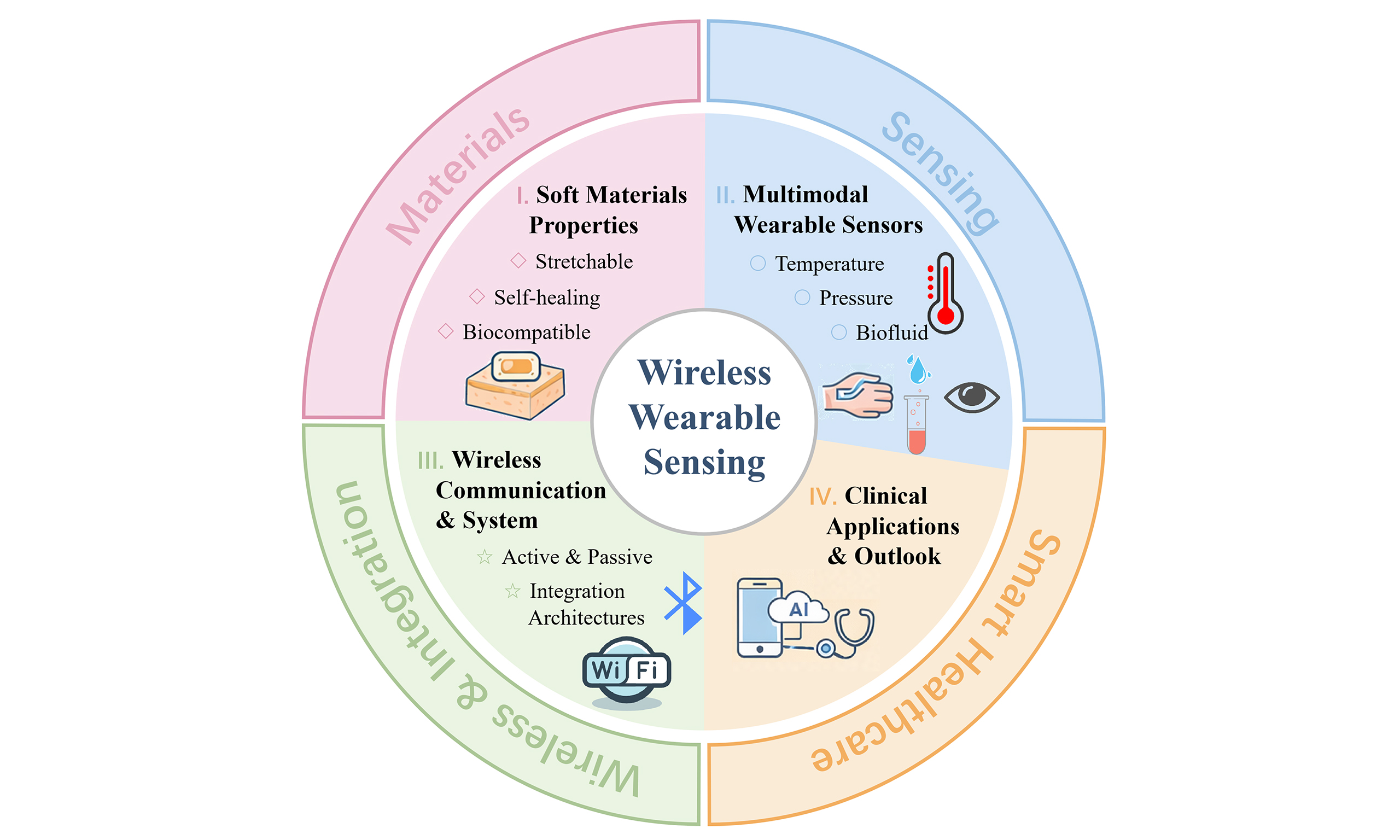
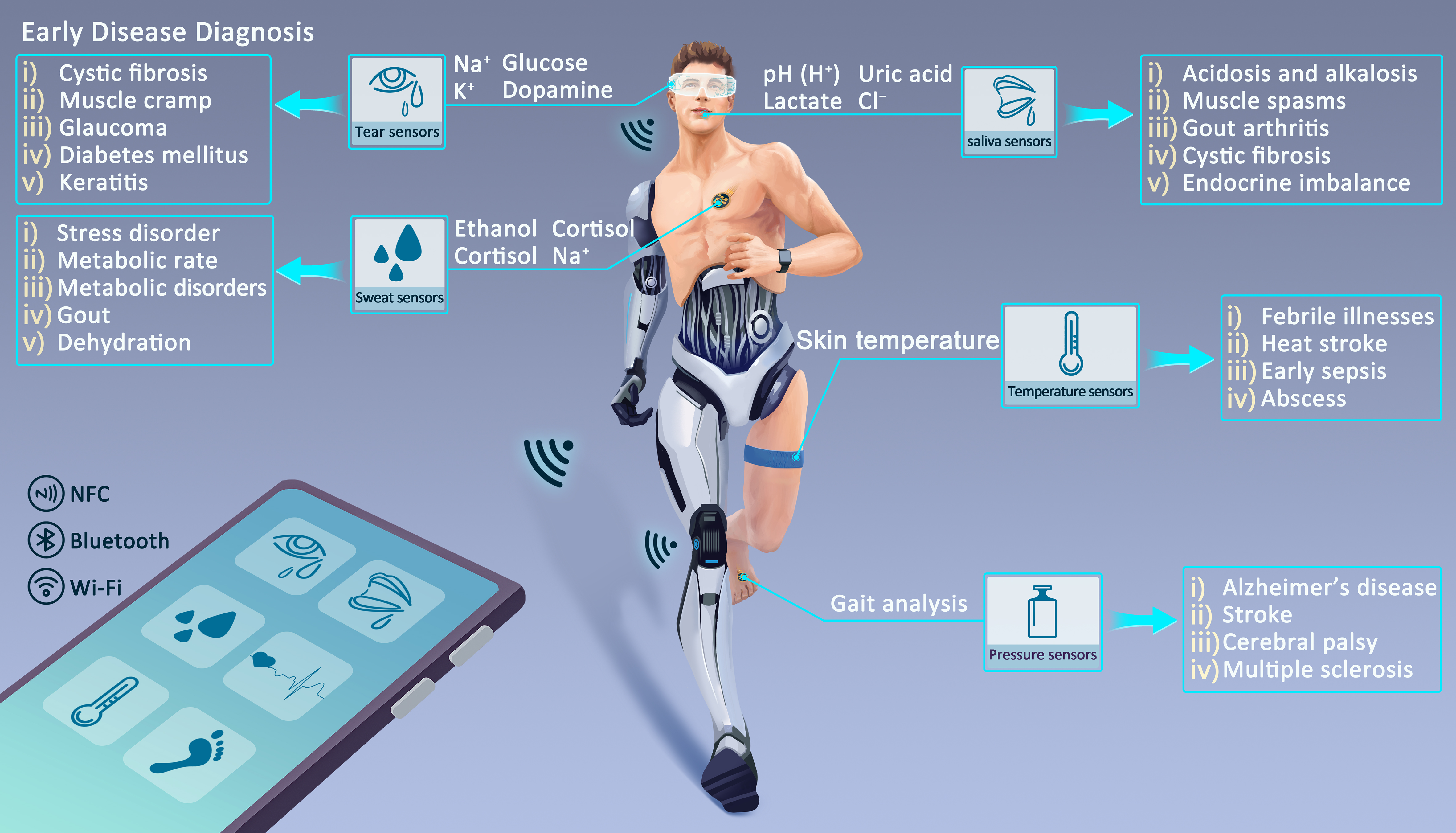
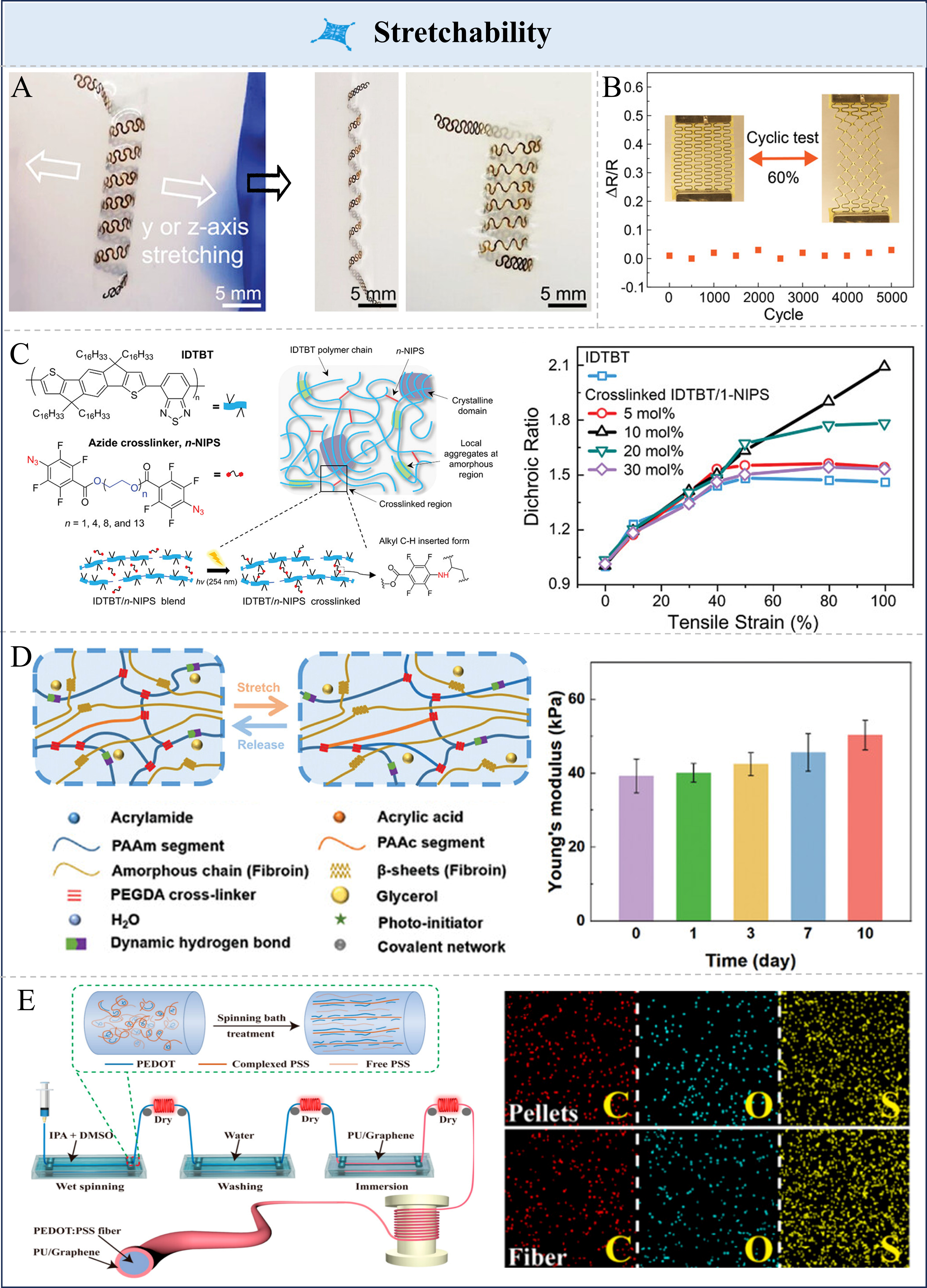
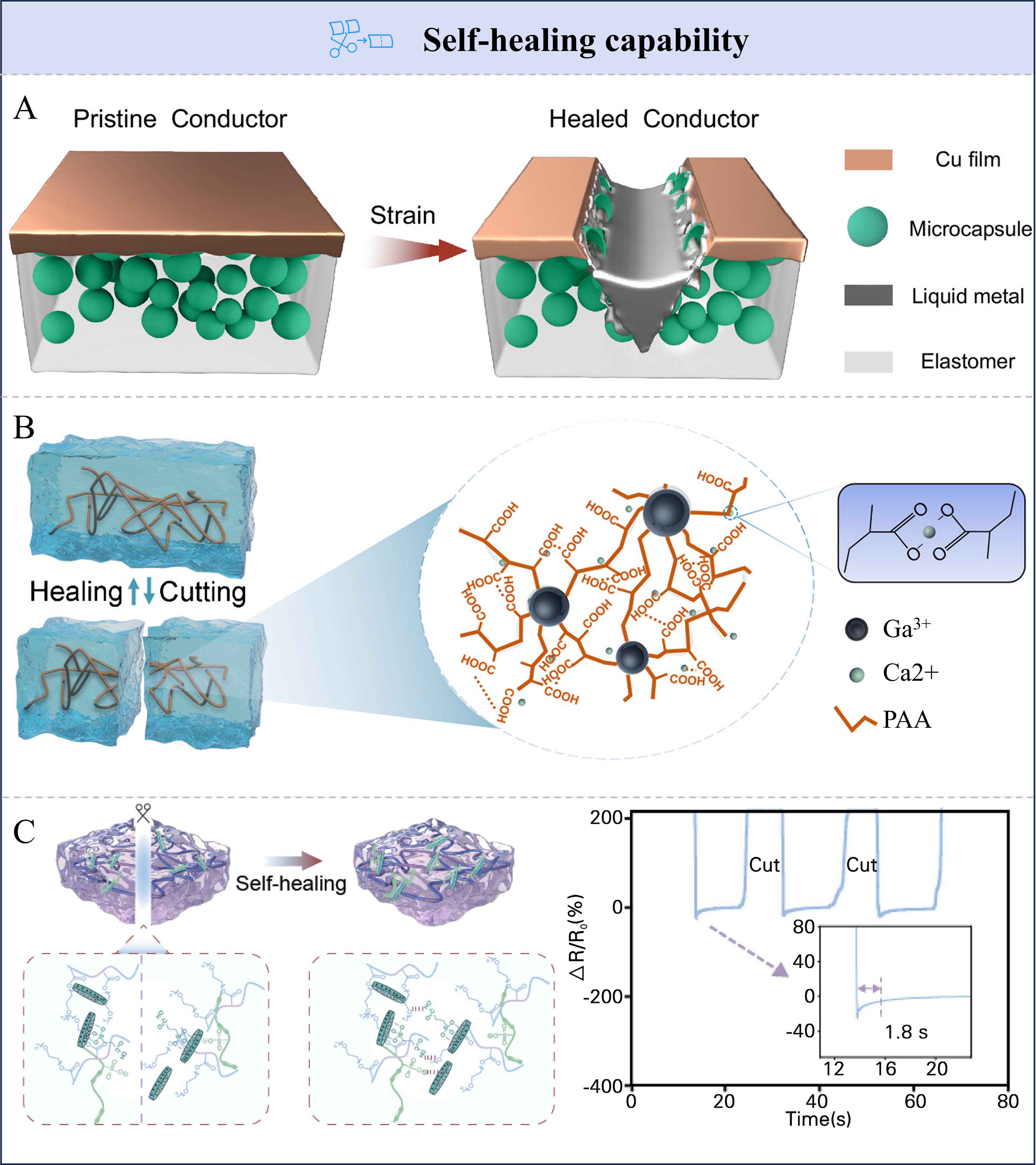
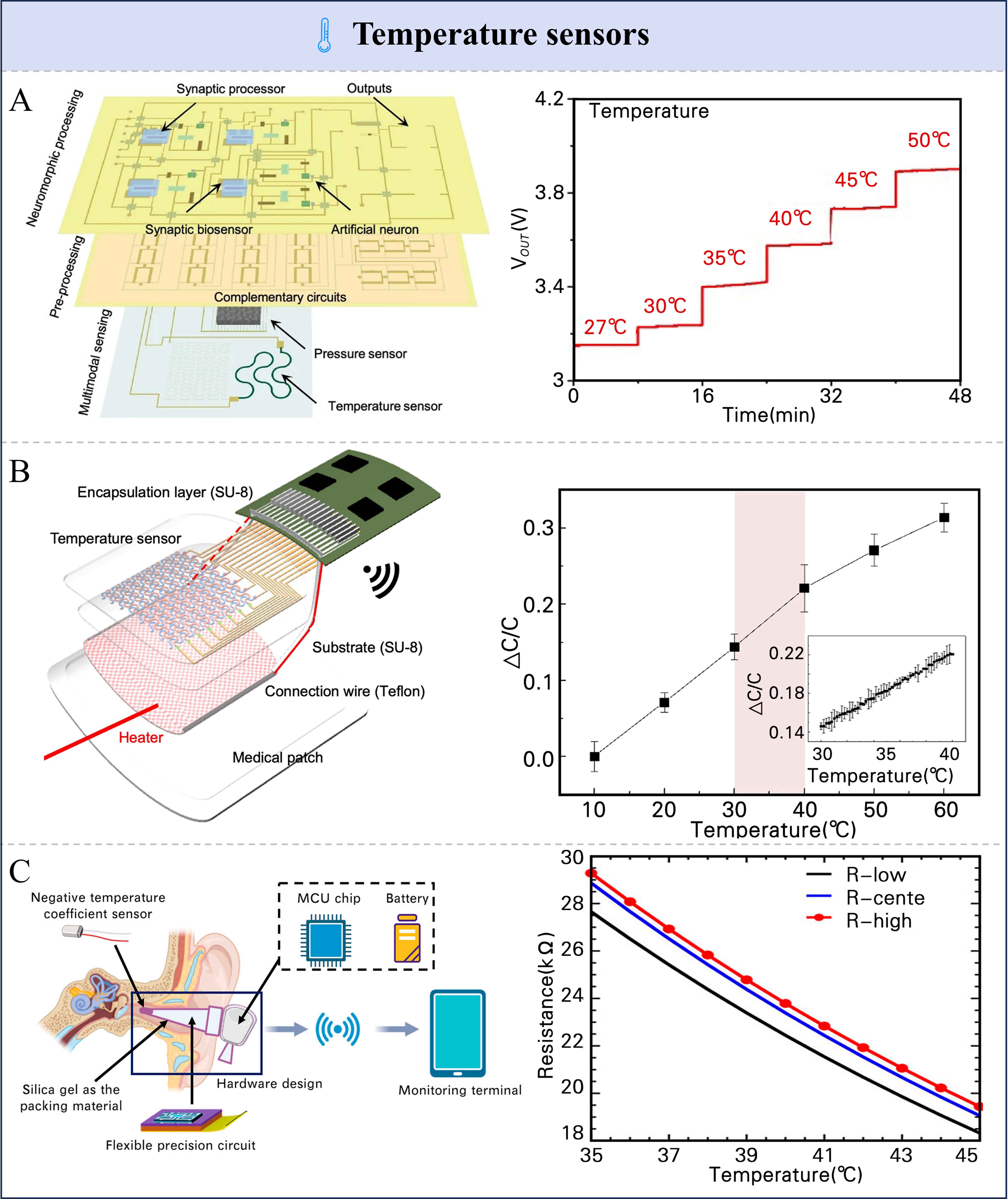
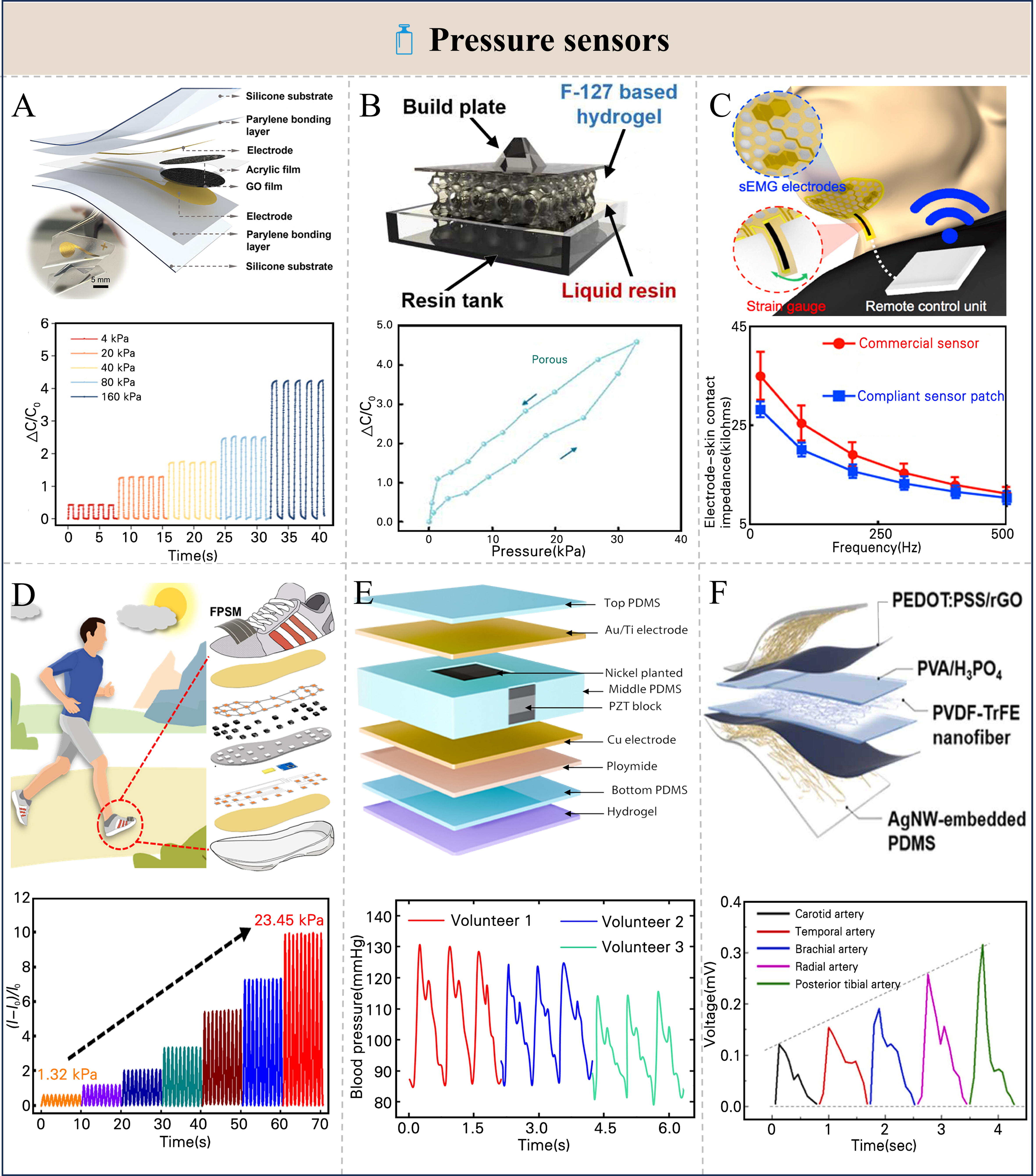
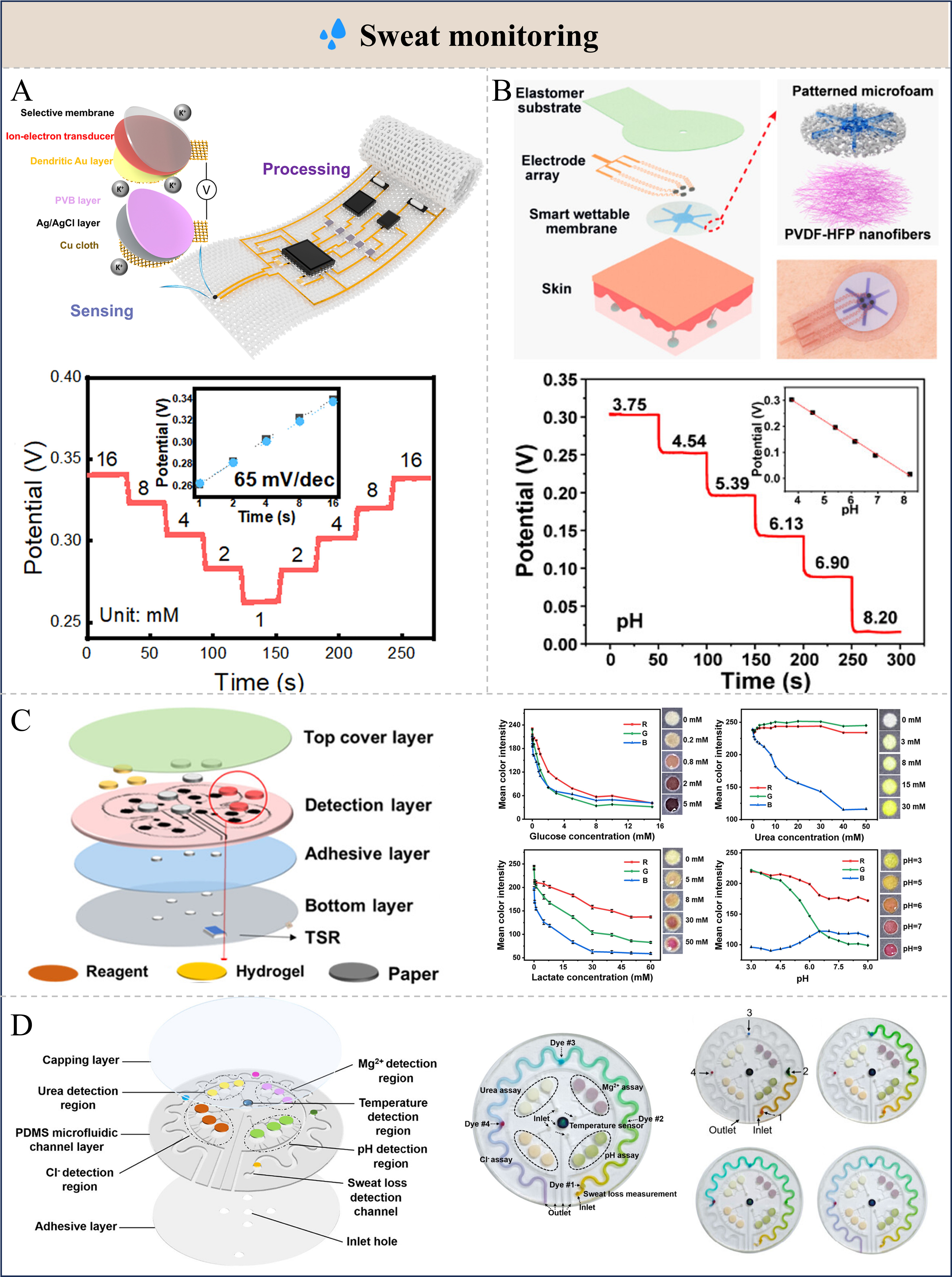
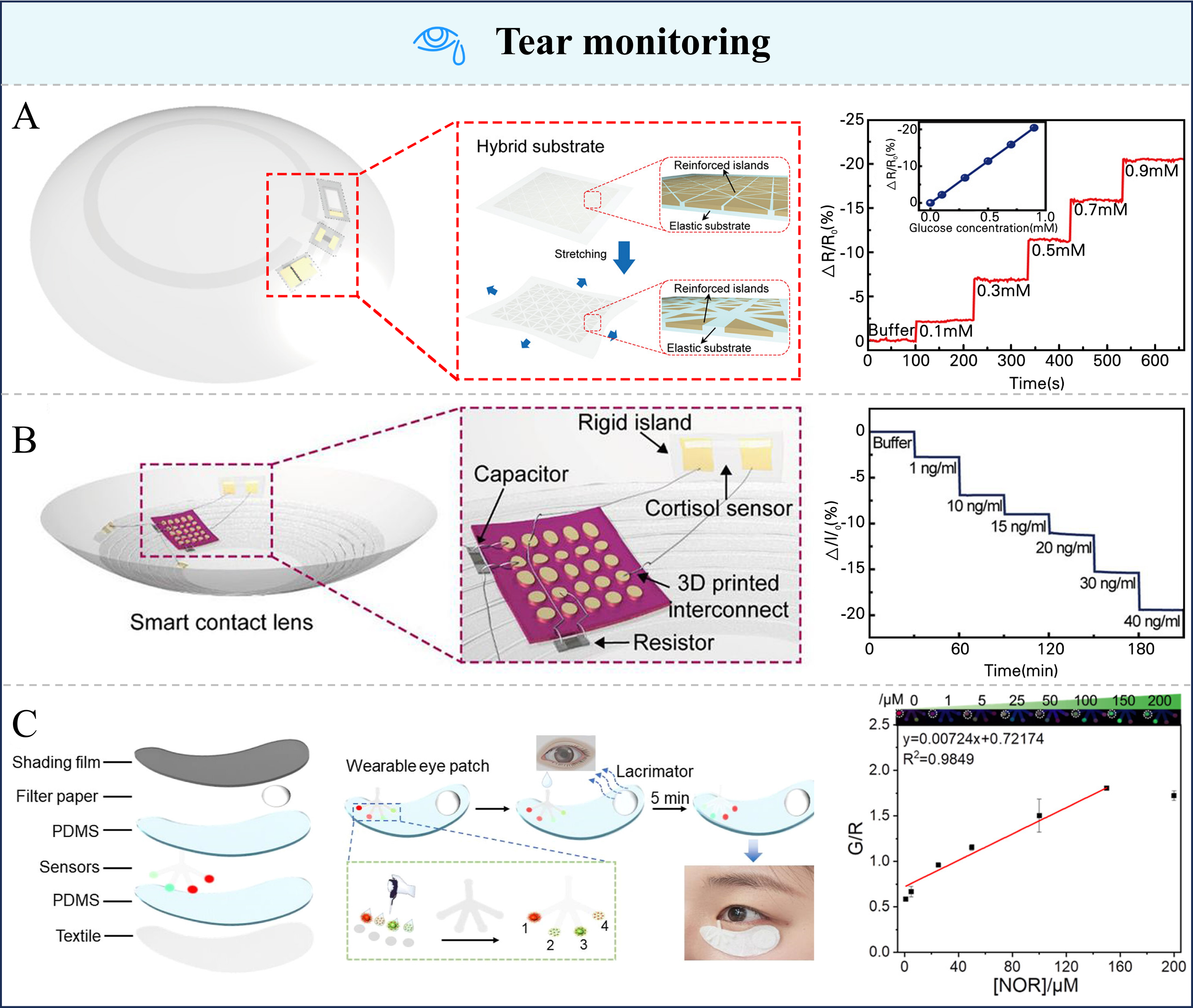
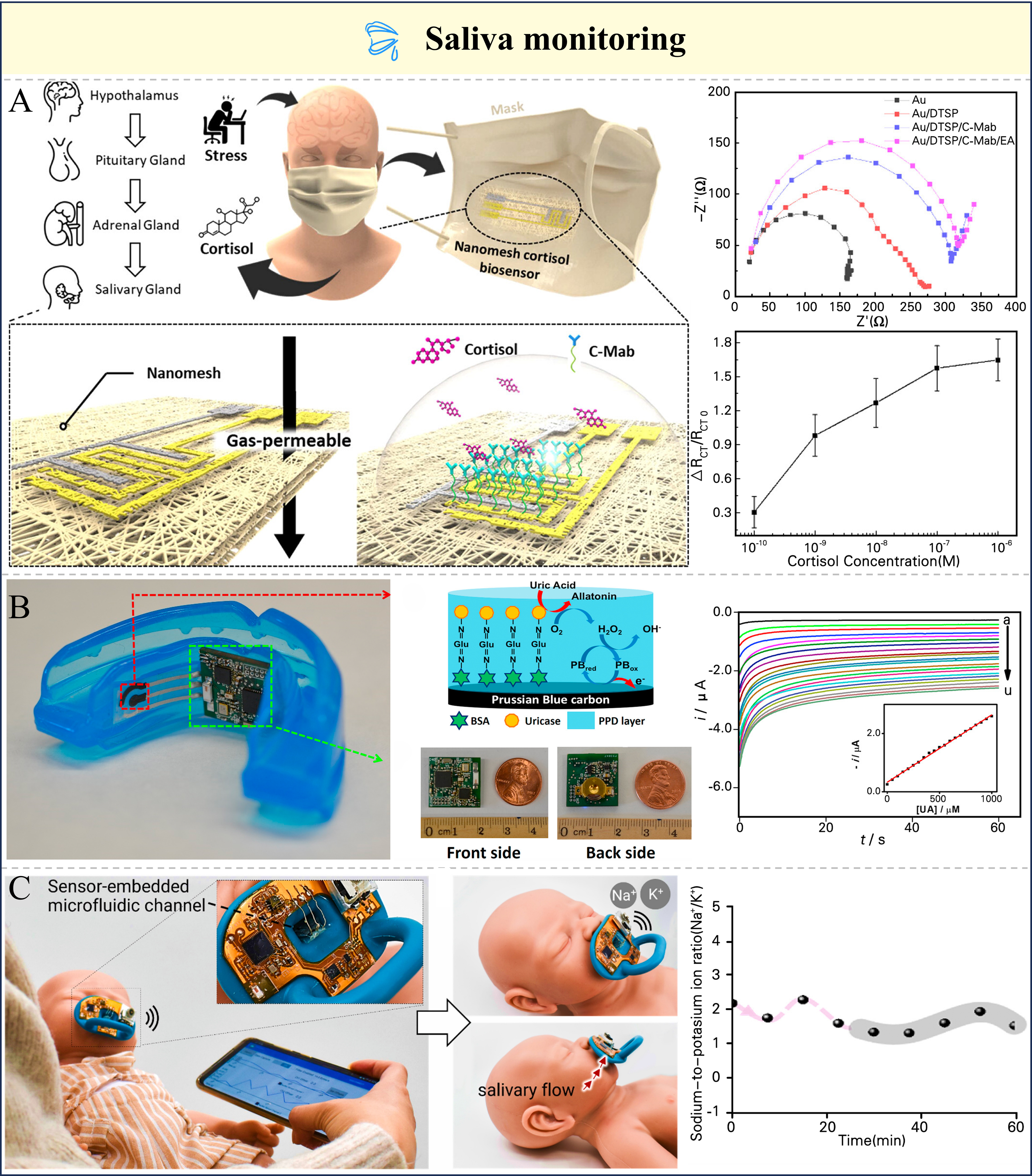
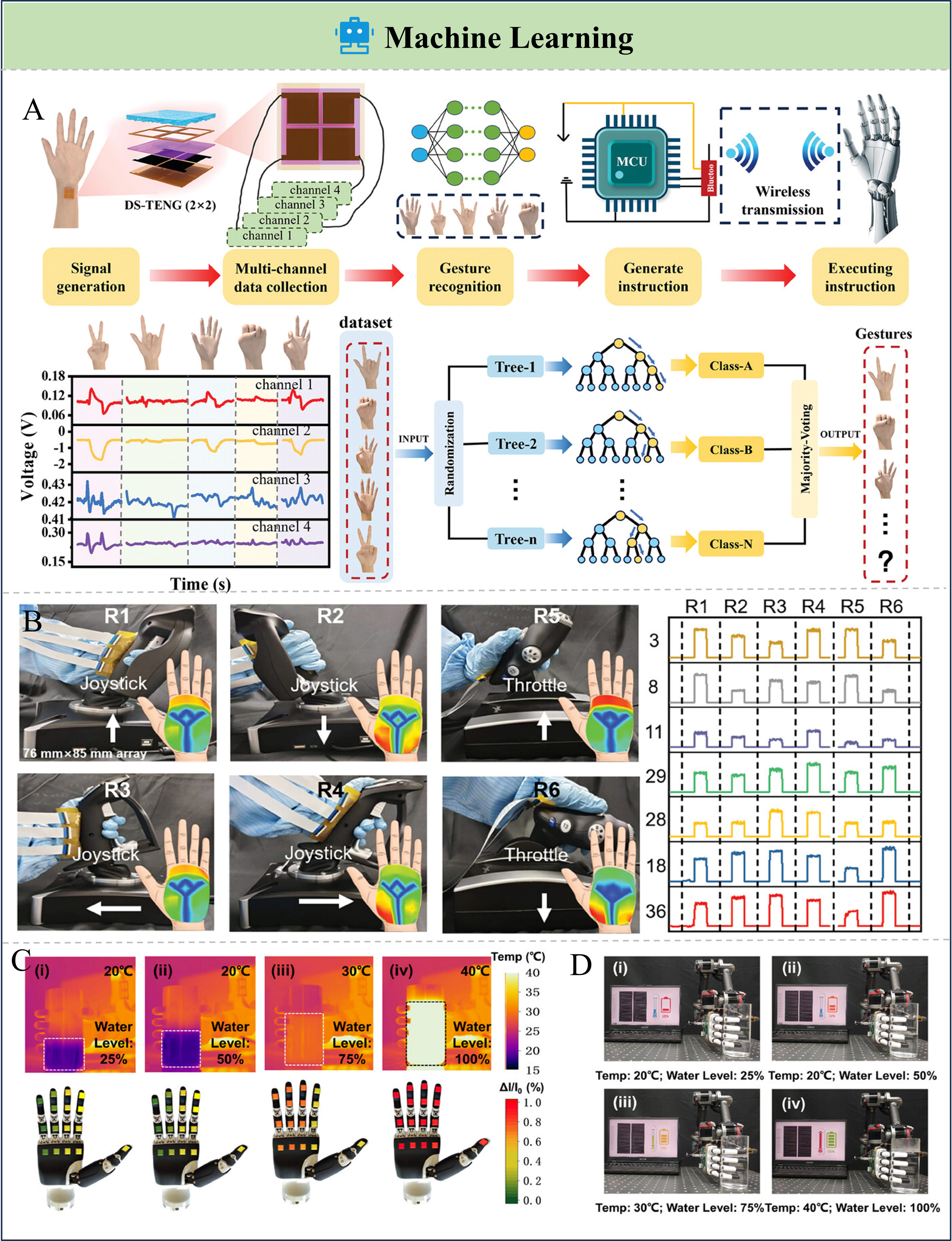
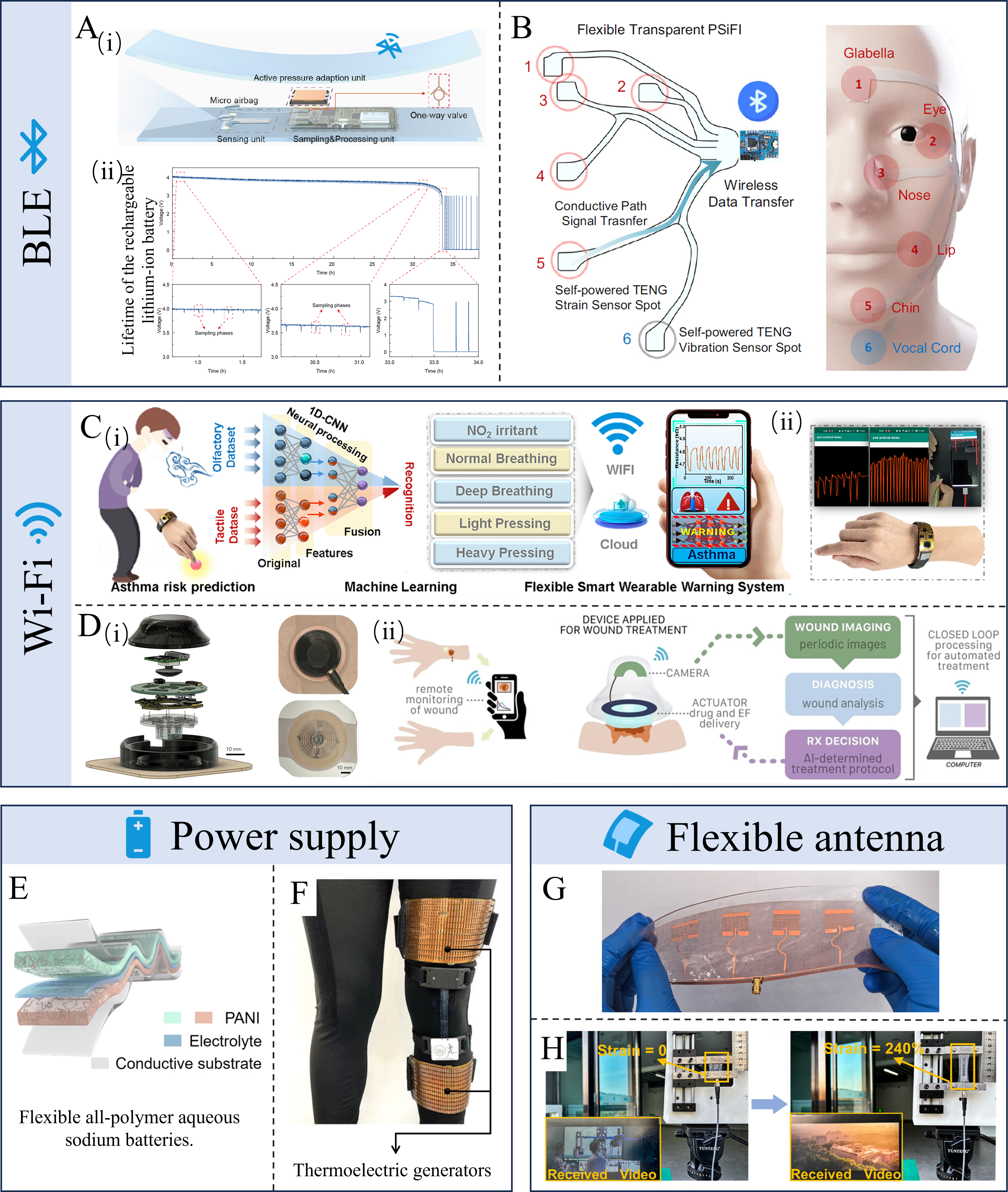
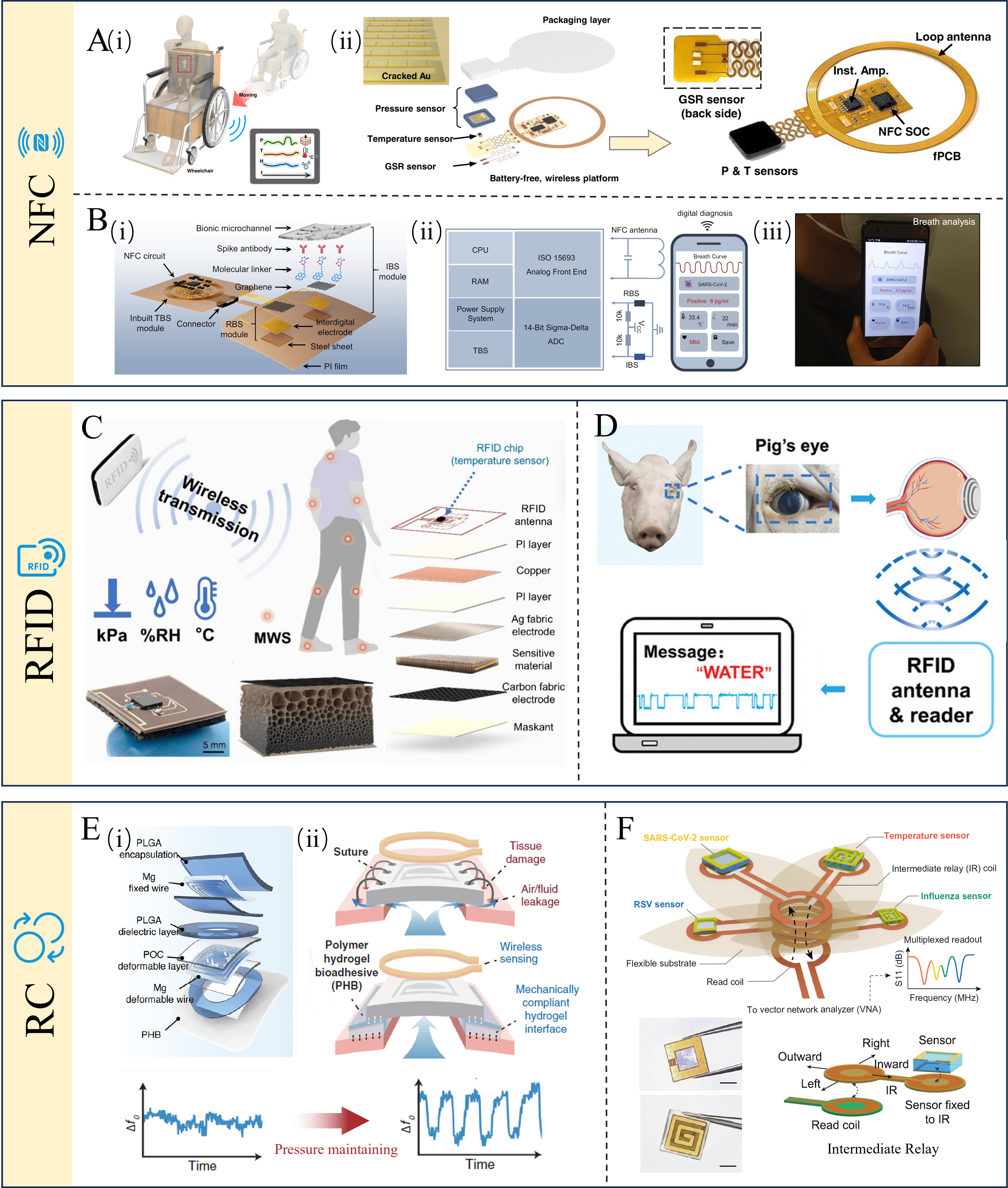
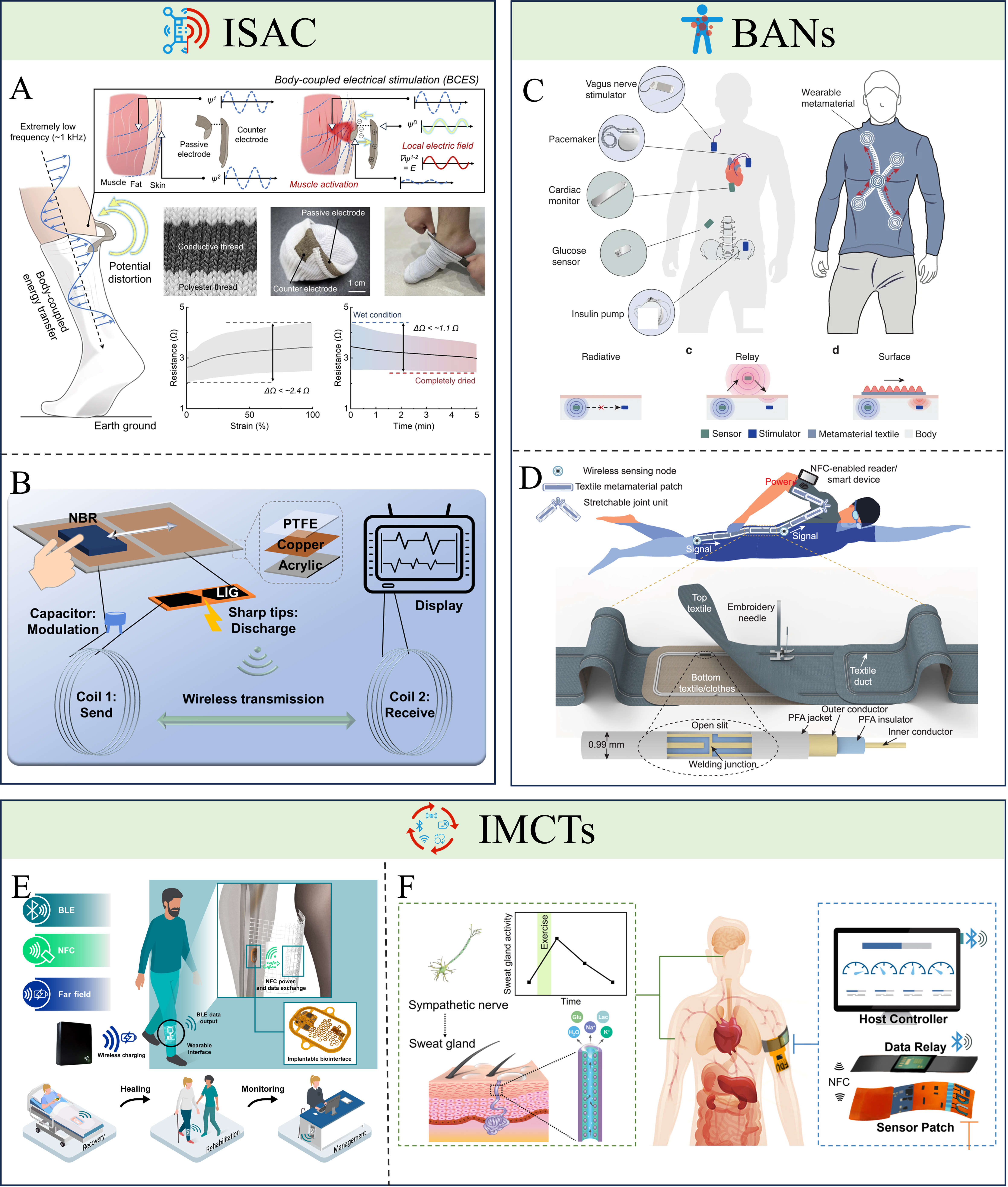












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].