Effects of once-weekly semaglutide on regional body composition in overweight or obese adults
Abstract
Aim: To explore the impact of once-weekly semaglutide-induced weight loss on regional body composition in overweight or obese adults.
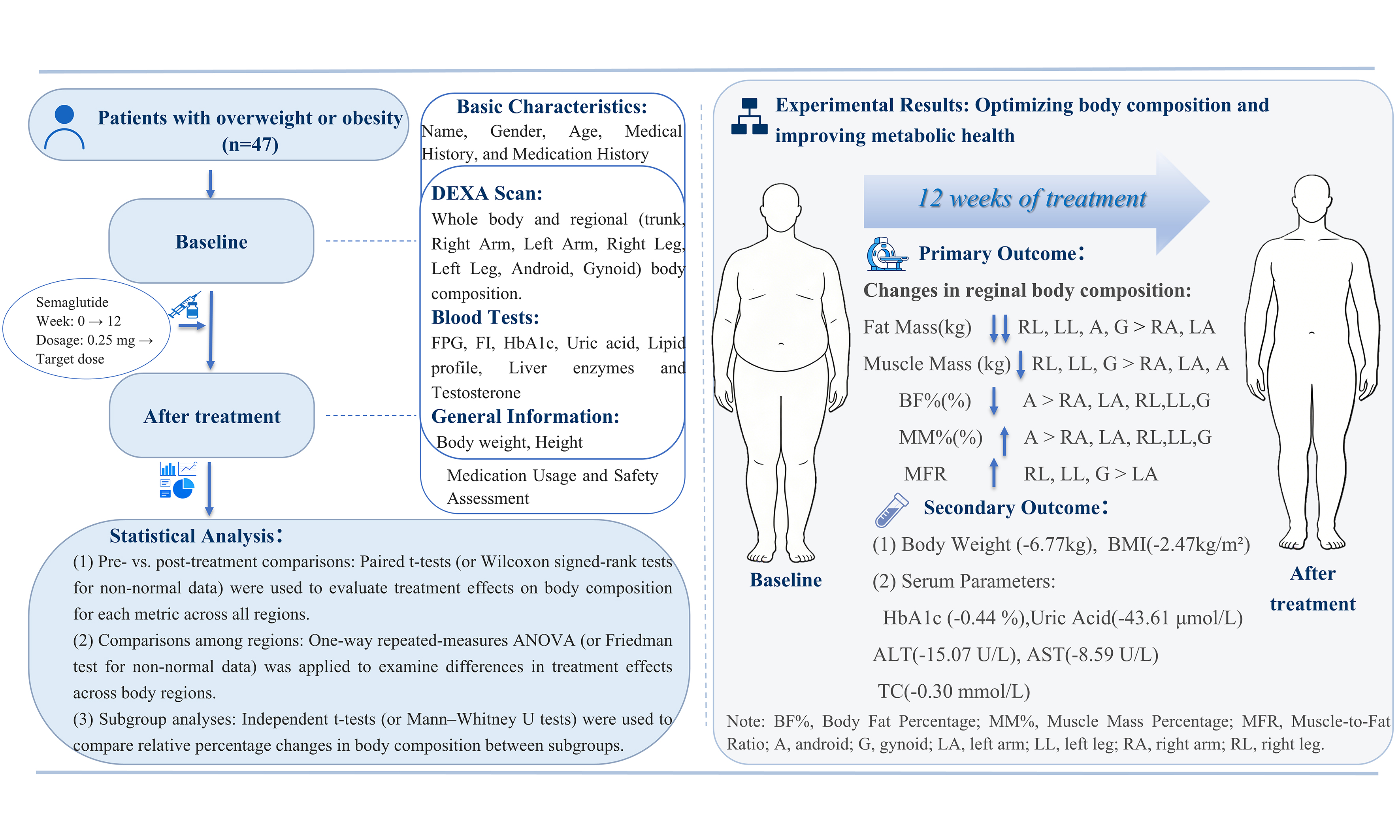
Methods: This retrospective cohort study enrolled 47 overweight or obese adults (body mass index ≥ 30 kg/m2, or ≥ 27 kg/m2 with ≥ 1 weight-related comorbidity). Participants received once-weekly semaglutide (initiated at 0.25 mg and gradually escalated to 2.0 mg) for 12 weeks, along with lifestyle intervention. The primary outcome was the change in regional body composition from baseline to week 12, which was measured using dual-energy X-ray absorptiometry.
Results: Semaglutide combined with lifestyle intervention resulted in a significant reduction in body weight (-6.77 ± 4.85 kg, P < 0.001), comprising a 4.75 kg (70.16%) loss of fat mass and a 2.02 kg (29.84%) reduction in muscle mass. The rate of visceral fat reduction was 5.16 percentage points greater than that of subcutaneous fat (21.25% vs. 16.09%; P = 0.009). With the exception of muscle mass in the right arm, both fat mass and muscle mass decreased in all other regions (all P < 0.008). The absolute reductions in fat mass, listed in descending order, were as follows: left leg (-0.74 kg), right leg (-0.71 kg), gynoid (-0.72 kg), android (-0.63 kg), left arm (-0.26 kg), and right arm (-0.19 kg). Similarly, the absolute reductions in muscle mass, in descending order, were: left leg (-0.54 kg), right leg (-0.48 kg), gynoid (-0.43 kg), android (-0.19 kg), left arm (-0.19 kg), and right arm (-0.08 kg). The muscle-to-fat ratio increased in all regions (all P < 0.008). Furthermore, multiple metabolic parameters improved concurrently.
Conclusion: Semaglutide, combined with lifestyle intervention, significantly reduces body weight, improves body composition, and enhances metabolic profiles. Future studies will further validate these findings by increasing the sample size, including parallel control groups, and extending the treatment and follow-up periods.
Keywords
INTRODUCTION
Recent data indicate that the global population of obese individuals has reached one billion[1]. Obesity has emerged as a severe and growing public health challenge worldwide. It is closely associated with an increased risk of hypertension, type 2 diabetes (T2DM), dyslipidemia, metabolic dysfunction-associated fatty liver disease (MAFLD), chronic kidney disease, osteoarthritis, sleep apnea, and various cancers[2-4]. Research indicates that obesity is associated with a 32% increase in all-cause mortality[5].
Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist and novel anti-obesity medication, demonstrates weight loss efficacy comparable to metabolic bariatric surgery. It also exhibits a more favorable safety profile, establishing it as a valuable option for weight management[4,6]. In the SUSTAIN China trial involving patients with obesity and T2DM, once-weekly semaglutide (1 mg) produced a significantly greater reduction in body weight compared with sitagliptin (-4.0 kg vs. -0.5 kg; P < 0.01)[7]. Wilding et al. reported in the STEP-1 trial (NCT03548935) that among overweight or obese adults (without diabetes), the semaglutide group achieved a superior mean weight reduction of 14.9% from baseline, compared to 2.4% in the placebo group (P < 0.001)[8].
However, weight loss involves more than a reduction in overall body mass; it involves complex alterations in specific body components, such as adipose tissue and skeletal muscle, where alterations in tissue quality and distribution are critical determinants of health outcomes. Research has demonstrated the differential effects of regional body composition on overall health. Studies have found a significant inverse association between leg muscle mass and the insulin resistance index. In particular, lower leg muscle mass is associated with greater insulin resistance. A large-scale study found that an elevated fat-to-muscle ratio in the limbs, particularly in the legs, is independently associated with an increased risk of osteoarthritis[9]. In addition, studies have shown that sarcopenia and central obesity are associated with an increased risk of cardiovascular mortality and all-cause mortality[10]. Visceral fat poses a significant threat to health and serves as an independent biomarker for morbidity and mortality from cardiovascular and metabolic diseases[11-13]. Furthermore, research has shown that the accumulation of abdominal fat is associated with brain atrophy, particularly reduced volumes in brain regions involved in cognitive function, which may contribute causally to cognitive decline[14,15]. Studies also indicated that individuals with abdominal obesity have a higher all-cause mortality risk, whereas those with leg and hip adiposity exhibit a lower risk. Furthermore, abdominal fat accumulation is associated with accelerated cardiovascular aging in men, whereas gluteofemoral fat accumulation is linked to its deceleration in women[16,17]. A longitudinal study of older Americans with diabetes found that a higher fat mass in the arms was associated with an increased risk of all-cause and non-cardiovascular mortality; conversely, a greater arm muscle mass was associated with reduced all-cause, cardiovascular, and non-cardiovascular mortality[18].
The exploratory Analysis of the STEP 1 Study (n = 140) involving dual-energy X-ray absorptiometry (DEXA) scans indicated that semaglutide-mediated weight loss was achieved mainly through a reduction in fat mass, specifically visceral adipose tissue. Furthermore, the loss of fat-free mass was proportional to the overall weight reduction, suggesting that semaglutide induces a favorable change in body composition[8]. Current research on the impact of semaglutide-induced weight loss on body composition has largely focused on overall assessments, lacking analysis of specific regional compartments such as the abdomen, hips, and individual limbs. Consequently, whether semaglutide exerts distinct effects on regional body composition remains unclear. Moreover, studies focusing on Chinese populations remain limited.
The present study employs a self-controlled design, utilizing DEXA to measure changes in regional body composition in overweight or obese adults before and after 12 weeks of semaglutide treatment. It aims to analyze the impact of semaglutide-induced weight loss on regional body composition and assess for significant differences across various body compartments.
METHODS
Study design and ethics
This study is a retrospective cohort analysis of data collected through a prospective, standardized clinical follow-up pathway. It was conducted at the Fifth Affiliated Hospital of Sun Yat-sen University, Guangdong, China. The study protocol was approved by the Institutional Review Board (IRB) of the Fifth Affiliated Hospital of Sun Yat-Sen University (Approval No: K287-1; approved in November 2025) and was conducted in accordance with the Declaration of Helsinki.
Participants
This study retrospectively analyzed data from adults with overweight or obesity who underwent a weight-loss treatment with subcutaneous injection of semaglutide combined with lifestyle intervention at the Endocrinology Department of the Fifth Affiliated Hospital of Sun Yat-sen University between April 2024 and November 2025.
Inclusion criteria: (1) 18 years old and above; and (2) body mass index (BMI) ≥ 30 kg/m2 or 27-30 kg/m2 and at least one weight-related comorbidity (e.g., hypertension, hyperglycemia, dyslipidemia, obstructive sleep apnea syndrome, cardiovascular disease).
Exclusion criteria: (1) Patients with various types of diabetes, end-stage renal disease, severe liver dysfunction, and medullary thyroid carcinoma; (2) Patients with obesity secondary to pathological factors, such as Cushing’s syndrome and hypothyroidism; (3) Patients with obesity induced by medications, including glucocorticoids; (4) Patients with triglycerides (TG) > 5.6 mmol/L; (5) Patients with a previous history of bariatric surgery; (6) Patients with contraindications to semaglutide or other GLP-1 receptor agonists (GLP-1 RAs); and (7) Patients unable to adhere to medication for three months, or those with other conditions deemed unsuitable for inclusion by the researchers.
Intervention
Each study participant received weekly subcutaneous injections of semaglutide (Ozempic®) at 1.0-2.0 mg (starting at 0.25 mg and gradually increasing to the target dose 2.0 mg based on patient tolerance). All participants also received standardized lifestyle counseling at baseline and every 4 weeks throughout the 12-week study period (detailed lifestyle guidance can be found in Supplementary Material 1).
Outcome measures
Primary Outcome: Changes in regional body composition from baseline to week 12, measured by DEXA.
Secondary Outcomes: Changes from baseline to week 12 in body weight, BMI, fasting blood glucose, fasting insulin (FI), glycated hemoglobin (HbA1c), blood lipids, uric acid, liver function, and testosterone in study participants.
Measurement indicators
Body composition measurement and index calculation
Regional body composition was measured using a DEXA instrument (GE Lunar iDXA, ME+212950)
Regional analysis: Fat distribution analysis was performed using the GE Lunar enCORE V18 software. The android fat region, defined according to the manufacturer’s algorithm, primarily corresponds to the upper abdominal area, whereas the gynoid fat region mainly represents the gluteal and upper thigh areas. The specific delineation rules are as follows: (1) Android region of interest (ROI): The inferior boundary is the pelvic tangent, and the superior boundary is located at 20% of the distance between the pelvic tangent and the neck tangent. The lateral boundaries are the arm tangents; (2) Gynoid ROI: The superior boundary is located 1.5 times the height of the android ROI below the pelvic tangent. The height of the gynoid ROI is equal to twice the height of the android ROI. The lateral boundaries are the outer leg tangents; (3) Trunk: This region was defined as the area bounded horizontally by the inferior margin of the chin line and the superior margin of the iliac crest, and separated from the arms by vertical lines descending from the medial aspects of the glenohumeral joints. It included the thoracic and lumbar vertebrae, ribs, sternum, pelvis, and overlying soft tissues; (4) Limbs: These were separated from the trunk by the aforementioned vertical lines. The lower limbs were defined as the area below the iliac crest horizontal line down to the toes, with the two legs separated by a vertical line through the midpoint of the pubic symphysis.
Definition of Muscle Mass: The muscle mass reported in this study was a composite measure derived from DEXA, representing the total mass of all non-fat, non-bone soft tissues. Thus, in addition to skeletal muscle, it includes visceral organs, body fluids, connective tissues, and other components. It is calculated as Muscle mass = Lean soft tissue mass - Bone mineral content.
Derived Indices: The following regional indices were calculated: (1) Body fat percentage = (Regional fat mass/Regional total mass) × 100%; (2) Muscle percentage = (Regional muscle mass/Regional total mass) × 100%; and (3) Muscle-to-fat ratio = Regional muscle mass/Regional fat mass.
Scanning protocol: Prior to scanning, subjects were required to fast overnight, wear light clothing, and remove shoes and socks. They were advised to avoid drinking water, eating, or engaging in strenuous activities for at least 30 min before the test and to empty their bladder. Subjects were positioned on the scanning table, centered horizontally, with the thumb facing up and the palm towards the leg. The arms were placed along the body without the hands touching the leg. The head was positioned about 3 cm below the scanning pad horizontally. Velcro straps were used to fix the patient’s knees and feet. After the staff entered the subject’s name, gender, age, height and other information, the start button was pressed to begin the scan, which lasted approximately 2 to 5 min. All scans were performed and analyzed by the same certified radiologist.
Anthropometric measurements
Weight measurement: Body weight was measured using an electronic scale. Subjects wore light clothing, removed their shoes, stood naturally in the center of the scale platform, and remained still during measurement. Weight was recorded in kilograms with an accuracy of two decimal places.
Height measurement: A mechanical height gauge was used. The subject stood barefoot on the base plate of the height scale, facing away from the column. The trunk was naturally straight, the head was upright, and both eyes were looking straight ahead (the lowest point of the upper edge of the tragus and the lower edge of the eye socket was at the same level). The upper limbs hang naturally, the heels were together, the toes were apart at about 60 degrees, and the sacral part of the heels and the space between the two shoulder blades were in contact with the column, forming a “three points in a line” standing posture. The recorded data was in centimeters and accurate to two decimal places.
Measurement of metabolic indicators
All blood samples were tested by the Department of Laboratory Medicine of the Fifth Affiliated Hospital of Sun Yat-sen University using standard clinical laboratory methods. Fasting blood glucose, blood lipid profiles, uric acid, and liver function indicators were detected using a Roche cobas c701 instrument. The FI and testosterone levels were determined by electrochemiluminescence immunoassay. HbA1c was determined by high performance liquid chromatography.
Bias
To minimize bias, we consecutively enrolled all eligible patients based on predefined criteria. All anthropometric measurements were performed by trained professionals following standardized protocols with calibrated equipment. Body composition was assessed using the same device under consistent conditions for all participants.
Study size
Based on data from a pilot study, the required sample size was calculated to be 30 participants to achieve 80% power at a two-sided significance level (α) of 0.05.
Statistical analysis
Statistical analyses were performed using SPSS 27.0. Missing data for laboratory parameters were handled using multiple imputation [Supplementary Material 1], while DEXA-derived regional body composition data were complete. Normally distributed data were presented as mean ± standard deviation (SD), and non-normally distributed data as median [interquartile range (IQR)]. (1) Pre- vs. post-treatment comparisons: To evaluate treatment effects on body composition, five metrics (fat mass, muscle mass, body fat percentage, muscle percentage, and muscle-to-fat ratio) were assessed across six regions (right/left arm, right/left leg, android, gynoid). Paired t tests (or Wilcoxon signed-rank tests for non-normal data) were performed for each region per metric. To control for multiple comparisons, a Bonferroni correction was applied within each metric group (six tests per metric), setting the significance threshold at α = 0.05/6 = 0.0083; (2) Comparisons among regions: To examine regional differences in treatment response, absolute and percentage changes were calculated for each metric. One-way repeated-measures ANOVA (or Friedman test for non-normal data) was performed with “body region” as the within-subject factor. Sphericity was assessed using Mauchly’s test, with Greenhouse-Geisser correction applied when violated. If the main effect was significant (P < 0.05), post-hoc pairwise comparisons with Bonferroni correction were conducted; (3) Subgroup analyses: Exploratory stratified analyses were performed by sex, baseline comorbidity status, and baseline visceral fat area (dichotomized by the sample mean). Relative percentage changes in body composition were compared between subgroups using independent t tests or Mann-Whitney U tests, as appropriate. A Bonferroni correction (α = 0.05/18 ≈ 0.0028) was applied to control for Type I error rate.
RESULTS
Study participants, treatment delivery and safety
This study included 47 overweight or obese subjects who completed the full 12-week treatment course (patients who discontinued treatment for personal reasons were not included in this primary analysis). The cohort consisted of 17 males (36.2%) and 30 females (63.8%). The median age of the cohort was 31 years (IQR 26-45), with a median BMI of 32.00 kg/m2 (IQR 29.5-35.2). Baseline comorbidities were as follows: hyperlipidemia (n = 16, 34.0%), hyperuricemia (n = 12, 25.5%), hypertension (n = 8, 17.0%), abnormal liver function (n = 8, 17.0%), family history of obesity (n = 8, 17.0%), polycystic ovary syndrome (n = 7, 14.9%), and sleep apnea syndrome (n = 3, 6.4%). The final maintenance dose of semaglutide had a median of 1.0 mg (IQR 1.0-1.5) with 4 patients (8.5%) successfully titrating to the target maintenance dose of 2.0 mg weekly. During the study period, the most common adverse events were mild gastrointestinal reactions, with incidences of constipation (62.5%), nausea (31%), and diarrhea (12.5%). Additionally, one case each of dizziness and depressed mood was observed. These events were transient and did not require discontinuation of treatment. No serious adverse events related to semaglutide were reported in this study (See Supplementary Tables 1 and 2 in Supplementary Material 2 for details).
Changes in whole-body composition
As shown in Table 1, after treatment, participants exhibited a notable reduction in total body weight
Changes in total body composition before and after treatment
| Body composition | Baseline | After treatment | Difference | P value | t | 95%CI | Cohen’s d |
| TBW (kg) | 89.81 ± 16.99 | 83.03 ± 16.39 | -6.77 ± 4.85 | < 0.001* | -9.58 | (-8.20,-5.35) | -1.40 |
| TFM (kg) | 37.37 ± 8.76 | 32.62 ± 8.53 | -4.75 ± 3.39 | < 0.001* | -9.60 | (-5.75,-3.75) | -1.40 |
| TMM (kg) | 49.87 ± 10.86 | 47.85 ± 10.31 | -2.02 ± 2.06 | < 0.001* | -6.73 | (-2.63,-1.42) | -0.98 |
| BF% (%) | 41.66 ± 5.26 | 39.21 ± 5.42 | - 2.45 ± 2.09 | < 0.001* | -7.99 | (-3.06,-1.83) | -1.17 |
| MM% (%) | 55.45 ± 5.13 | 57.65 ± 5.24 | 2.21 ± 1.97 | < 0.001* | 7.68 | (1.63,2.78) | 1.12 |
| MFR | 1.37 ± 0.30 | 1.52 ± 0.35 | 0.15 ± 0.14 | < 0.001* | 7.17 | (0.11,0.19) | 1.05 |
Changes in visceral and subcutaneous fat after semaglutide combined with lifestyle therapy
As shown in Table 2, the combined therapy significantly reduced both visceral and subcutaneous fat. Specifically, visceral fat mass decreased by 0.36 kg and visceral fat area by 40.03 cm2, while subcutaneous fat mass decreased by 0.37 kg and subcutaneous fat area by 41.60 cm2. All reductions remained statistically significant after the Bonferroni correction (all P < 0.001). Further comparisons revealed that the absolute reductions in visceral and subcutaneous fat were comparable, with no statistically significant differences (mass difference: 0.01 kg, P = 0.836; area difference: 1.57 cm2, P = 0.753). However, the relative reduction in visceral fat was significantly greater than that in subcutaneous fat (5.16 percentage points, P < 0.05).
Adipose tissue distribution at baseline and after intervention
| Δ (Absolute change from baseline) | P value1 (vs. baseline) | Cohen’s d1 | P value2 (VAT vs. SAT) | Cohen’s d2 | Δ% (Relative changes) | P value3 (VAT vs. SAT) | Cohen’s d 3 | |
| VAT mass (kg) | -0.36 ± 0.35 | < 0.001* | -1.03 | 0.836 | 0.03 | -21.25 ± 2.22 | 0.009* | -0.40 |
| SAT mass (kg) | -0.37 ± 0.30 | < 0.001* | -1.21 | -16.09 ± 12.39 | ||||
| VAT area (cm2) | -40.03 ± 36.32 | < 0.001* | -1.10 | 0.753 | 0.05 | -20.99 ± 2.17 | 0.008* | -0.41 |
| SAT area (cm2) | -41.60 ± 32.87 | < 0.001* | -1.27 | -15.83 ± 1.75 |
Changes in regional body composition after treatment
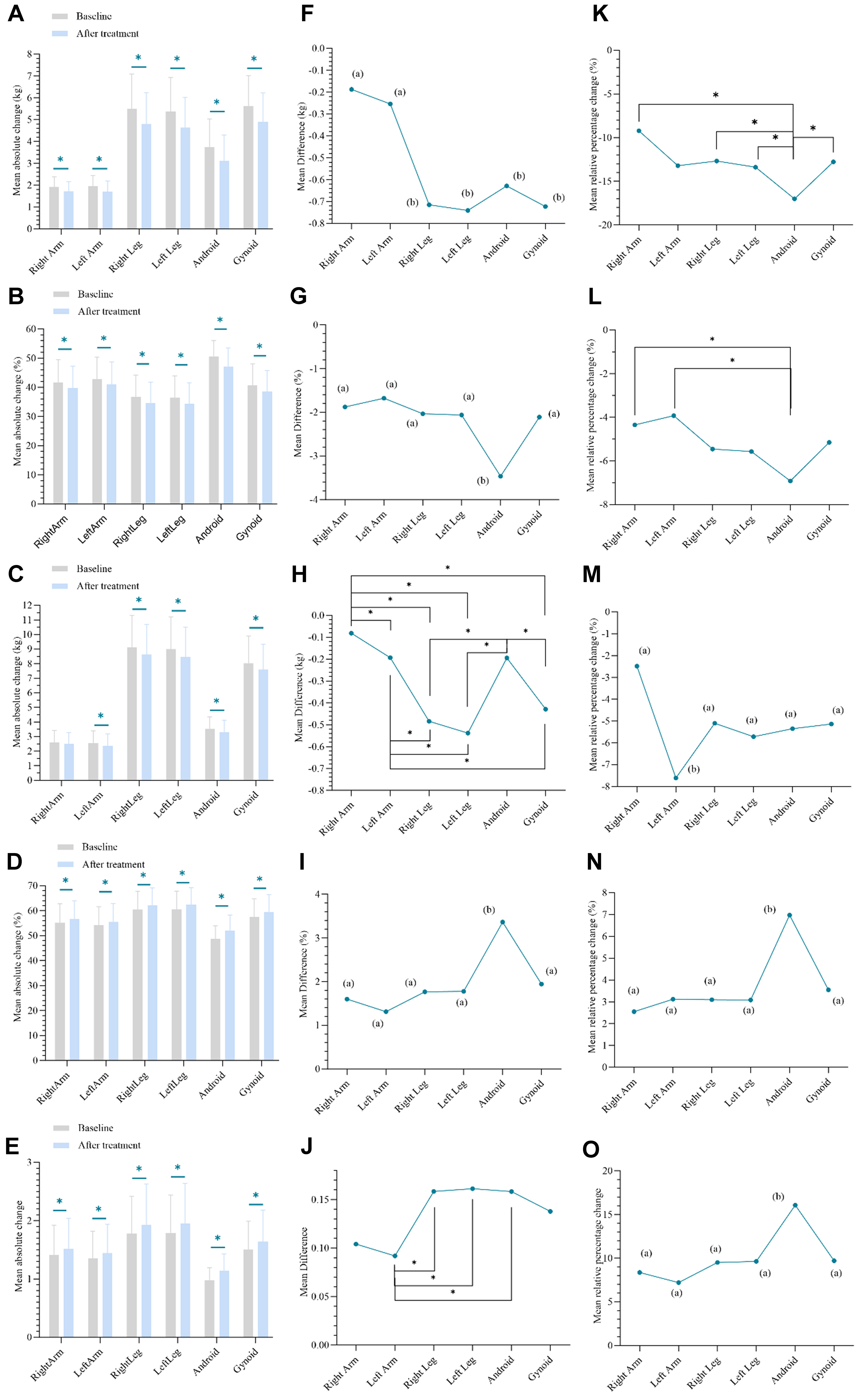
As shown in Figure 1, following treatment, both fat mass and body fat percentage decreased significantly across all regions (paired t tests with Bonferroni correction, all P < 0.0083). One-way repeated-measures ANOVA with post-hoc comparisons revealed a significant main effect of body region on the absolute reduction in fat mass, F (2.386, 109.740) = 32.855, P < 0.001, partial η2 = 0.417 (Greenhouse-Geisser corrected, ε = 0.477). Here, F represents the F-statistic used in ANOVA to test for differences between group means; partial η2 quantifies the proportion of variance in the dependent variable explained by the independent variable; and ε denotes the Greenhouse-Geisser correction factor, which adjusts the degrees of freedom when sphericity is violated, yielding a more rigorous F test. The absolute reductions in fat mass (ΔM ± SD) were as follows: left leg (-0.74 ± 0.60 kg), right leg (-0.71 ± 0.60 kg), gynoid (-0.72 ± 0.57 kg) and android (-0.63 ± 0.49 kg) > right arm (-0.19 ± 0.24 kg) and left arm (-0.26 ± 0.24 kg). Likewise, statistically significant differences were observed across body regions for percent change in fat mass, absolute change in body fat percentage, and relative change in body fat percentage. All measures exhibited a consistent order of reduction: android > right arm, left arm, right leg, left leg, gynoid (see Supplementary Tables 5-12 in Supplementary Material 2 for details).
Figure 1. Effect of semaglutide combined with lifestyle intervention on regional body composition. (A-E) Pre- to post-treatment changes in regional body composition: (A) FM; (B) FM%; (C) MM; (D) MM%; and (E) MFR; (F-J) Regional variation in absolute changes from baseline: (F) FM; (G) FM%; (H) MM; (I) MM%; and (J) MFR; (K-O) Regional variation in the percentage change from baseline: (K) FM; (L) FM%; (M) MM; (N) MM%; and (O) MFR. Data are presented as means. Regions sharing a common letter do not differ significantly (P ≥ Bonferroni-adjusted α); regions assigned different letters are significantly different (P < Bonferroni-adjusted α). Asterisks (*) indicate P < 0.05 for pairwise comparisons after Bonferroni-adjusted. FM: Fat mass; FM%: fat mass percentage; MM: muscle mass; MM%: muscle mass percentage; MFR: muscle-to-fat ratio.
Following treatment, muscle mass decreased in all regions except the right arm (all P < 0.001 for regions showing a significant decrease). The results from the one-way repeated-measures analysis of variance (ANOVA) indicated a significant main effect of body region on the absolute reduction in muscle mass, F (2.456, 112.962) = 19.479, P < 0.001, partial η2 = 0.297 (Greenhouse-Geisser corrected, ε = 0.491). Based on the integration of descriptive statistics and post hoc analysis, the results indicated an order of absolute muscle mass reduction (ΔM ± SD) as follows: left leg (-0.54 ± 0.54 kg), right leg (-0.48 ± 0.56 kg), gynoid (-0.43 ± 0.43 kg) > android (-0.19 ± 0.23 kg), left arm (-0.19 ± 0.26 kg) and right arm (-0.08 ± 0.21 kg). Regarding the percentage change in muscle mass from baseline, the left arm showed a significantly greater reduction (-7.61% ± 9.88%) compared with the other regions. In contrast, the changes in the right arm, right leg, left leg, android, and gynoid regions did not differ significantly from each other. Notably, a consistently greater reduction was observed in the left arm compared to the right arm, both in terms of absolute muscle mass loss and the percentage change from baseline, with statistically significant differences (absolute change: -0.19 kg vs. -0.08 kg; percent change: -7.61% vs. -2.49%) (see Supplementary Tables 13-17 in Supplementary Material 2 for details).
Despite a decline in muscle mass across all regions, both the muscle mass percentage and the muscle-to-fat ratio increased. For the muscle mass percentage, the increase was most pronounced in the android, with an absolute change of 3.36% and a relative change of 6.98% (all P < 0.05). In contrast, no significant changes were detected in the right arm, left arm, right leg, left leg, or gynoid. Regarding the muscle-to-fat ratio, the regional order of significant absolute changes (largest to smallest) was: right leg (0.16 ± 0.17), left leg (0.16 ± 0.16), android (0.16 ± 0.17) > left arm (0.09 ± 0.12). In terms of the percentage change, the order was: android (16.08% ± 16.76%) > right arm, left arm, right leg, left leg, gynoid (see Supplementary Tables 18-27 in Supplementary Material 2 for details).
Exploratory subgroup analysis
Results showed that at the corrected significance level, treatment-induced percentage changes in body composition did not differ significantly across subgroups based on sex, comorbidity status, or visceral fat level (all P > 0.0028). This suggests a lack of pronounced subgroup heterogeneity in the treatment effect within this study sample (Details in Supplementary Table 28 in Supplementary Material 2).
Changes in clinical indicators
As shown in Table 3, following treatment with semaglutide combined with lifestyle intervention, BMI was reduced by 2.47 ± 1.70 kg/m2, and HbA1c also showed a significant reduction (-0.44% ± 0.36%). Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) decreased by means of -15.07 ± 26.40 U/L and -8.59 ± 17.55 U/L, respectively. Concurrently, serum uric acid levels decreased by a mean of -43.61 ±
Comparison of clinical indicators before and after treatment
| Baseline | After treatment | Difference | P value | 95%CI | Cohen’s d | |
| BMI (kg/m2) | 32.67 ± 3.94 | 30.19 ± 4.18 | -2.47 ± 1.70 | < 0.001* | (-2.97,-1.98) | -1.46 |
| FPG (mmol/L) | 5.59 ± 0.66 | 5.44 ± 0.47 | -0.15 ± 0.65 | 0.118 | (-0.34,0.04) | -0.23 |
| FI (μU/ml) | 25.34 ± 13.30 | 26.50 ± 23.42 | 1.08 ± 22.95 | 0.749 | (-5.66, 7.81) | 0.05 |
| HbA1c (%) | 5.84 ± 0.40 | 5.40 ± 0.30 | -0.44 ± 0.36 | < 0.001* | (-0.54, -0.33) | -1.20 |
| Uric acid (μmol/L) | 407.38 ± 91.43 | 363.77 ± 92.19 | -43.61 ± 86.80 | 0.001* | (-69.09,-18.12) | -0.50 |
| ALT (U/L) | 49.67 ± 43.36 | 34.60 ± 37.22 | -15.07 ± 26.40 | < 0.001* | (-22.82, -7.31) | -0.57 |
| AST (U/L) | 34.72 ± 22.10 | 26.13 ± 19.45 | -8.59 ± 17.55 | 0.002* | (-13.74, -3.43) | -0.49 |
| TG (mmol/L) | 1.77 ± 0.87 | 1.51 ± 1.11 | -0.26 ± 0.78 | 0.027 | (-0.49, -0.30) | -0.33 |
| TC (mmol/L) | 5.06 ± 1.01 | 4.75 ± 1.06 | -0.30 ± 0.57 | < 0.001* | (-0.47, -0.13) | -0.53 |
| HDL-C (mmol/L) | 1.21 ± 0.31 | 1.19 ± 0.34 | -0.02 ± 0.25 | 0.618 | (-0.09, 0.06) | -0.07 |
| LDL-C (mmol/L) | 3.34 ± 0.81 | 3.13 ± 0.84 | -0.22 ± 0.58 | < 0.014 | (-0.38,-0.05) | -0.37 |
| Testosterone (ng/ml) | ||||||
| Male | 3.59 ± 1.80 | 4.19 ± 2.44 | 0.60 ± 0.98 | 0.022 | (0.10,1.10) | 0.62 |
| Female | 0.44 ± 0.51 | 0.47 ± 0.53 | 0.02 ± 0.14 | 0.402 | (-0.03,0.08) | 0.16 |
| HOMA-IR | 6.41 ± 3.64 | 6.64 ± 7.03 | 0.23 ± 6.76 | 0.817 | (-1.75, 2.21) | 0.03 |
DISCUSSION
This study utilized DEXA-measured body composition to systematically evaluate the impact of semaglutide combined with lifestyle intervention on the regional body composition of overweight or obese individuals. Semaglutide combined with lifestyle significantly reduced body weight, and this weight loss primarily resulted from the decrease in fat mass, which is consistent with the core conclusions of current studies on GLP-1 RAs[8,19-22]. The weight loss process shows significant regional heterogeneity, especially with the highest relative reduction in central fat. While treatment was accompanied by a decrease in absolute muscle mass, the muscle percentage and muscle-to-fat ratio increased across all areas, indicating that relative muscle was preserved. Within the sample of this study, no pronounced subgroup heterogeneity in treatment effect was observed. In addition, it brought a wide range of metabolic benefits. This study addresses the gap in previous semaglutide research regarding insufficient attention to the regional heterogeneity of body composition in Chinese overweight or obese populations.
Regarding changes in regional fat distribution, the proportional reduction of visceral fat was significantly greater than that of subcutaneous fat. In terms of changes in fat mass, the decreases in right arm, left arm, android, and gynoid were all greater than those in the right leg and left leg, a difference that may be related to variations in baseline fat mass across regions. Analysis of the relative percentage change in fat mass revealed that abdominal fat decreased most significantly. Furthermore, both the android body fat percentage and the magnitude of its change were significantly larger than those in other regions. These findings indicate that the fat-reducing effect of semaglutide combined with lifestyle interventions is not uniform, as it leads to a greater reduction in abdominal and visceral fat depots, which pose the greatest risk to metabolic health. This suggests that it may provide additional metabolic protection beyond weight loss alone. The underlying mechanisms may be related to the higher metabolic activity, richer blood supply, or greater sensitivity to GLP-1 receptor-mediated signaling within these fat depots[23-29]. Further research is needed to elucidate the molecular basis behind this specificity.
This study observed a reduction in muscle mass across all regions following treatment except the right arm. The decrease in muscle mass was more pronounced in the lower limbs and gynoid compared to the upper limbs and android, which may be related to their higher baseline muscle mass or adaptive changes associated with weight loss, as these areas are major fat depots. A noteworthy finding was the statistically significant difference in muscle reduction between the left and right upper limbs. In terms of relative change, muscle loss in the left arm was the most significant, while the magnitude of change in the right arm, lower limbs, android, and gynoid was relatively similar. Given that all subjects were right-handed, this disparity may reflect higher susceptibility of the non-dominant upper limb to muscle loss during energy deficit due to insufficient daily mechanical loading. This suggests that incorporating resistance exercise could help mitigate muscle loss during treatment. Additionally, factors such as minor postural deviations during measurement or technical errors in image segmentation may have influenced the results. These findings require further validation through more rigorously designed studies in the future.
From the perspective of body composition proportions, although the absolute muscle mass decreased across all regions, both the muscle percentage (muscle rate) and the muscle-to-fat ratio increased, indicating an optimization of body composition. The most pronounced improvement was observed in the abdominal region: both the absolute change in muscle rate and its percentage change from baseline were significantly higher than those observed in other regions, while the percentage change in the muscle-to-fat ratio from baseline was also markedly superior to that of other areas. In contrast, the trends in muscle rate changes in the upper limbs, lower limbs, and buttocks were relatively consistent. These findings suggest that the treatment regimen centered on semaglutide most effectively optimizes abdominal body composition, achieving a substantial reduction in metabolically harmful fat while relatively preserving muscle proportion. This provides body composition-level evidence for the specific benefits of the treatment in addressing central obesity and related metabolic abnormalities. Future studies may further explore the dynamic relationship between this regional body composition optimization and improvements in glucolipid metabolic indicators.
Semaglutide combined with lifestyle intervention significantly improved multiple metabolic parameters. After treatment, HbA1c, uric acid, AST, ALT, and TC were significantly reduced, indicating clear benefits of this regimen for glycemic control, liver function improvement, and partial lipid regulation. These improvements are consistent with most previous studies[30,31]. Changes in some parameters did not reach the significance threshold after multiple corrections: although triglyceride and low-density lipoprotein levels showed a decreasing trend, the differences were not statistically significant following Bonferroni correction. Additionally, a decrease in testosterone levels was observed in male participants, the clinical significance of which requires further investigation in relation to gonadal function and changes in body composition. These findings may be limited by the relatively small sample size and inter-individual metabolic heterogeneity, underscoring the need for validation in larger populations. Furthermore, no significant changes were observed in fasting blood glucose, FI, or the HOMA-IR. This is consistent with the known glucose-lowering mechanism of semaglutide, which primarily involves delayed gastric emptying and enhanced postprandial GLP-1 secretion, suggesting that its glycemic benefits are likely mediated mainly through postprandial glucose control. Changes in testosterone levels among female participants did not reach statistical significance, potentially due to greater individual variability (e.g., menstrual cycle, age, and ovarian functional status) and possible confounding effects of the lifestyle intervention.
This study has several limitations. First, the relatively small sample size may have allowed inter-individual heterogeneity in metabolism and body composition to influence the stability of the findings. Second, the single-arm, non-controlled design limits the ability to distinguish natural variation from intervention effects, thereby limiting the ability to draw causal inference. Third, the short observation period did not cover the weight-loss plateau phase or long-term adaptive changes in body composition; extended follow-up is necessary to clarify these dynamic trajectories. Furthermore, although a lifestyle intervention was implemented, dietary and physical activity adherence was not strictly quantified, which may have introduced confounding bias. Moreover, because all participants received the combined intervention, it was not possible to disentangle the independent contributions of semaglutide and lifestyle modifications. The observed benefits should therefore be attributed to the integrated management approach. Finally, it should be noted that the muscle mass measured by DEXA in this study is derived from the difference between lean body mass and bone mineral content. Its changes reflect overall trends in muscle tissue rather than precise measurements of pure muscle tissue.
In conclusion, semaglutide combined with lifestyle intervention not only reduces body weight but also effectively improves body composition and associated metabolic abnormalities in obese patients. This study provides multidimensional evidence regarding body composition and metabolic outcomes to inform the clinical management of obesity and related metabolic disorders. Future research with larger sample sizes, longer follow-up periods, and controlled study designs is warranted to delineate the independent contributions of different interventions (medication vs. lifestyle) and to investigate the mechanistic links between changes in body composition and metabolic benefits.
DECLARATIONS
Authors’ contributions
Made substantial contributions to conception and design of the study and performed data acquisition, analysis and interpretation: Hu F
Writing-original draft preparation: Ma Y
Performed data analysis: Ma Y, Tang B
Revised the manuscript: Hu F, Zeng Y, Cong L
Provided technical support [dual-energy X-ray absorptiometry (DEXA) scanner operation]: Zhan Y
Availability of data and materials
All data generated or analyzed during this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
The study protocol was approved by the Institutional Review Board (IRB) of the Fifth Affiliated Hospital of Sun Yat-Sen University, Guangzhou, China (Approval No: K287-1; approved in November 2025) and was conducted in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403:1027-50.
2. Janić M, Janež A, El-Tanani M, Rizzo M. Obesity: recent advances and future perspectives. Biomedicines. 2025;13:368.
3. Xu Y, Jia W, Niu X, et al. Prevalence and comorbidity network analysis of obesity and related complications: a real-world study based on 233,004 individuals. Diabetes Metab Syndr Obes. 2025;18:3297-314.
5. Kivimäki M, Strandberg T, Pentti J, et al. Body-mass index and risk of obesity-related complex multimorbidity: an observational multicohort study. Lancet Diabetes Endocrinol. 2022;10:253-63.
6. Thomsen RW, Mailhac A, Løhde JB, Pottegård A. Real-world evidence on the utilization, clinical and comparative effectiveness, and adverse effects of newer GLP-1RA-based weight-loss therapies. Diabetes Obes Metab. 2025;27:66-88.
7. Ji L, Dong X, Li Y, et al. Efficacy and safety of once-weekly semaglutide versus once-daily sitagliptin as add-on to metformin in patients with type 2 diabetes in SUSTAIN China: a 30-week, double-blind, phase 3a, randomized trial. Diabetes Obes Metab. 2021;23:404-14.
8. Wilding JPH, Batterham RL, Calanna S, et al.; STEP 1 Study Group. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989-1002.
9. Luo J, Xiang Q, Lin T, et al. Associations between total and regional fat-to-muscle mass ratio and osteoarthritis incidence: a prospective cohort study. Osteoarthritis Cartilage. 2025;33:1023-32.
10. Benz E, Pinel A, Guillet C, et al. Sarcopenia and sarcopenic obesity and mortality among older people. JAMA Netw Open. 2024;7:e243604.
11. Liu Z, Huang Q, Deng B, et al. Elevated Chinese visceral adiposity index increases the risk of stroke in Chinese patients with metabolic syndrome. Front Endocrinol. 2023;14:1218905.
12. Zhang Z, Zhao L, Lu Y, Meng X, Zhou X. Association between Chinese visceral adiposity index and risk of stroke incidence in middle-aged and elderly Chinese population: evidence from a large national cohort study. J Transl Med. 2023;21:518.
13. Neeland IJ, Ross R, Després JP, et al.; International Atherosclerosis Society, International Chair on Cardiometabolic Risk Working Group on Visceral Obesity. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. 2019;7:715-25.
14. Mina T, Yew YW, Ng HK, et al. Adiposity impacts cognitive function in Asian populations: an epidemiological and Mendelian Randomization study. Lancet Reg Health West Pac. 2023;33:100710.
15. Raji CA, Meysami S, Hashemi S, et al. Visceral and subcutaneous abdominal fat predict brain volume loss at midlife in 10,001 individuals. Aging Dis. 2024;15:1831-42.
16. Losev V, Lu C, Tahasildar S, et al. Sex-specific body fat distribution predicts cardiovascular ageing. Eur Heart J. 2025;46:5076-88.
17. Jayedi A, Soltani S, Zargar MS, Khan TA, Shab-Bidar S. Central fatness and risk of all cause mortality: systematic review and dose-response meta-analysis of 72 prospective cohort studies. BMJ. 2020;370:m3324.
18. Guo J, Wei Y, Heiland EG, Marseglia A. Differential impacts of fat and muscle mass on cardiovascular and non-cardiovascular mortality in individuals with type 2 diabetes. J Cachexia Sarcopenia Muscle. 2024;15:1930-41.
19. Chen S, Wang X, Jin Y, et al. Assessment of changes in body composition after 3 months of dulaglutide treatment. Diabetes Metab Syndr Obes. 2024;17:1301-8.
20. McCrimmon RJ, Catarig AM, Frias JP, et al. Effects of once-weekly semaglutide vs once-daily canagliflozin on body composition in type 2 diabetes: a substudy of the SUSTAIN 8 randomised controlled clinical trial. Diabetologia. 2020;63:473-85.
21. Uchiyama S, Sada Y, Mihara S, Sasaki Y, Sone M, Tanaka Y. Oral semaglutide induces loss of body fat mass without affecting muscle mass in patients with type 2 diabetes. J Clin Med Res. 2023;15:377-83.
22. Alissou M, Demangeat T, Folope V, et al. Impact of semaglutide on fat mass, lean mass and muscle function in patients with obesity: the SEMALEAN study. Diabetes Obes Metab. 2026;28:112-21.
23. Bu T, Sun Z, Pan Y, Deng X, Yuan G. Glucagon-like peptide-1: new regulator in lipid metabolism. Diabetes Metab J. 2024;48:354-72.
24. Hropot T, Herman R, Janez A, Lezaic L, Jensterle M. Brown adipose tissue: a new potential target for glucagon-like peptide 1 receptor agonists in the treatment of obesity. Int J Mol Sci. 2023;24:8592.
25. Martins FF, Marinho TS, Cardoso LEM, et al. Semaglutide (GLP-1 receptor agonist) stimulates browning on subcutaneous fat adipocytes and mitigates inflammation and endoplasmic reticulum stress in visceral fat adipocytes of obese mice. Cell Biochem Funct. 2022;40:903-13.
26. Papakonstantinou I, Tsioufis K, Katsi V. Spotlight on the mechanism of action of semaglutide. Curr Issues Mol Biol. 2024;46:14514-41.
27. Tamayo-Trujillo R, Ruiz-Pozo VA, Cadena-Ullauri S, et al. Molecular mechanisms of semaglutide and liraglutide as a therapeutic option for obesity. Front Nutr. 2024;11:1398059.
28. Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367.
29. Xiang J, Qin L, Zhong J, Xia N, Liang Y. GLP-1RA liraglutide and semaglutide improves obesity-induced muscle atrophy via SIRT1 pathway. Diabetes Metab Syndr Obes. 2023;16:2433-46.
30. Yao H, Zhang A, Li D, et al. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ. 2024;384:e076410.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].