Synergistic mechanical nanostructuring and peroxymonosulfate activation enable superior piezocatalysis
Abstract
Piezocatalysis facilitates the transduction of mechanical energy into chemical redox processes, but its practical application is hindered by intrinsically low catalytic efficiency and complex catalyst fabrication. Herein, we employ high-energy ball milling (HBM) to convert bulk lead-free Sr0.5Ba0.5Nb2O6 (SBN) ceramics into nanoscale piezocatalysts (SBN-HBM) with enhanced activity, and integrate them with peroxymonosulfate (PMS) activation to promote reactive oxygen species generation, thereby boosting overall catalytic performance. HBM refines grain size from the microscale to ~240 nm and introduces abundant oxygen vacancies, enhancing both piezoelectric polarization and surface reactivity. Under mechanical excitation, the integrated SBN-HBM/PMS system triggers synergistic oxidation featuring hydroxyl radicals (•OH), sulfate radicals (SO4•-), and piezo-induced holes, resulting in markedly accelerated degradation kinetics (e.g., k =
Keywords
INTRODUCTION
Energy-autonomous oxidation platforms that operate efficiently and robustly in complex aqueous environments remain a grand challenge in water remediation[1]. In this context, piezocatalysis, which exploits the piezoelectric polarization of non-centrosymmetric materials under mechanical stress to drive charge separation, offers a unique solution by directly converting ubiquitous mechanical energy into redox reactivity[2,3]. This technology holds promise for autonomous catalytic activity in pollutant degradation and pathogen control in decentralized or off-grid settings[4,5].
To date, various piezocatalysts have been developed, including piezoelectric semiconductors (e.g., ZnO)[6,7], layered materials (e.g., MoS2, WS2)[8-10], perovskite-type ferroelectric oxides [e.g., BaTiO3, BiFeO3, (K, Na)NbO3][11-15], and tetragonal tungsten bronze structural piezoceramics [e.g., Sr0.5Ba0.5Nb2O6 (SBN)][16,17]. Typically, these piezoelectric materials can generate a built-in electric field under mechanical stimuli to promote electron-hole separation, thereby facilitating the formation of reactive oxygen species (ROS) such as hydroxyl radicals (•OH) and superoxide radicals (O2•-) for pollutant degradation and disinfection[18]. However, most pristine piezocatalysts often suffer from intrinsically low carrier density, rapid charge recombination, and limited active sites, which restrict ROS yields and catalytic reaction efficiency[19,20]. To overcome these limitations, engineering strategies such as defect modulation, morphology control, and heterojunction construction have been proposed to improve performance[21-23]. Yet, most of these approaches typically require complex synthesis processes, precise doping protocols, or expensive instrumentation, hampering scalability and practical application.
Recent advances have revealed that piezocatalysts can effectively activate peroxymonosulfate (PMS) to amplify ROS generation beyond the capabilities of conventional piezocatalysis[24,25]. By serving as a robust electron acceptor, PMS leverages piezo-induced charges to initiate complex oxidative cascades, yielding highly reactive species such as •OH and sulfate radicals (SO4•-)[26,27]. Importantly, PMS activation encompasses both radical and non-radical pathways (e.g., singlet oxygen (1O2), high-valent metal species), thereby diversifying the oxidative mechanisms and overcoming intrinsic limitations of electron-hole recombination in piezocatalysts[28,29]. Despite these advantages, existing piezocatalytic PMS activation systems predominantly utilize two-dimensional materials or perovskite oxides synthesized through elaborate and low-throughput methods, limiting their economic viability and scalability[26,30].
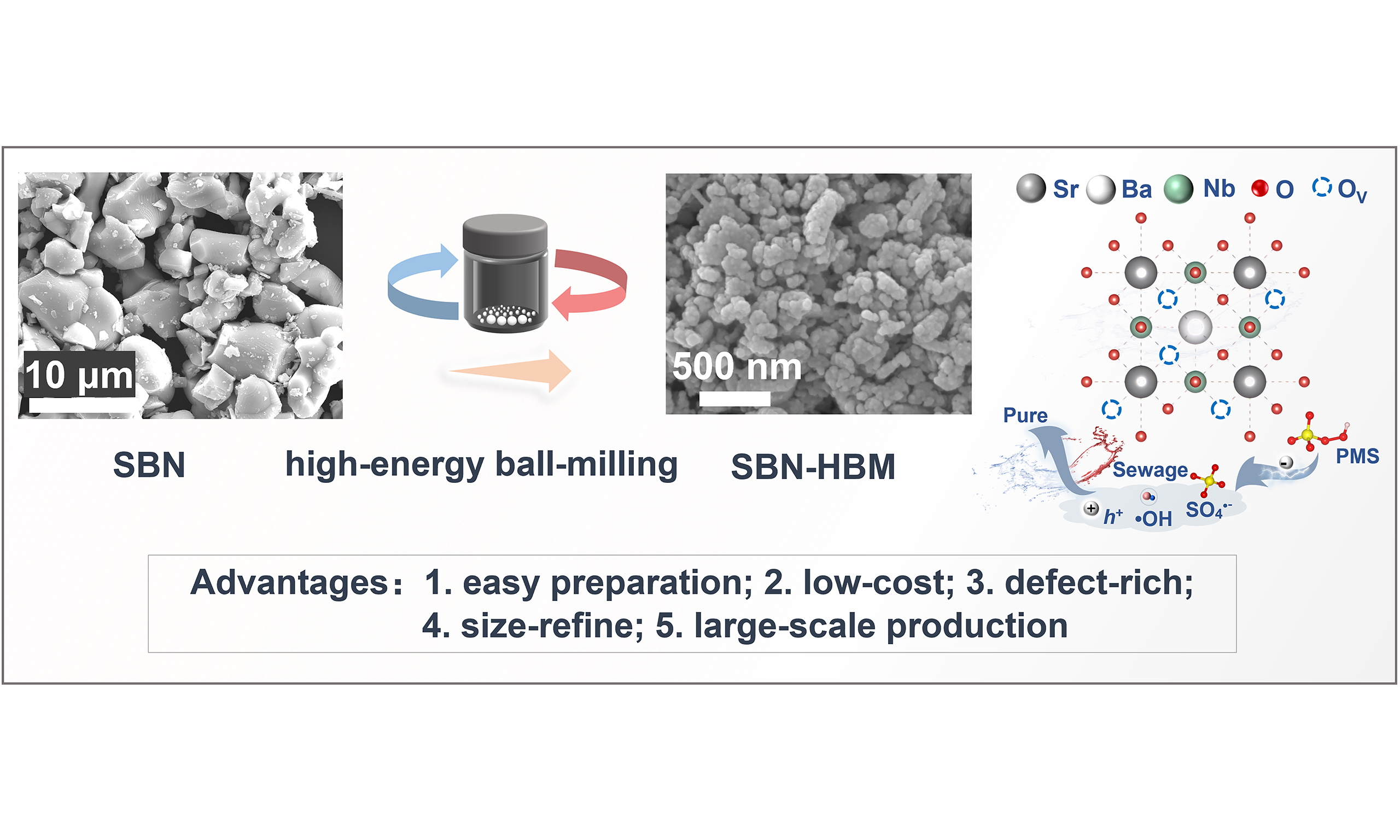
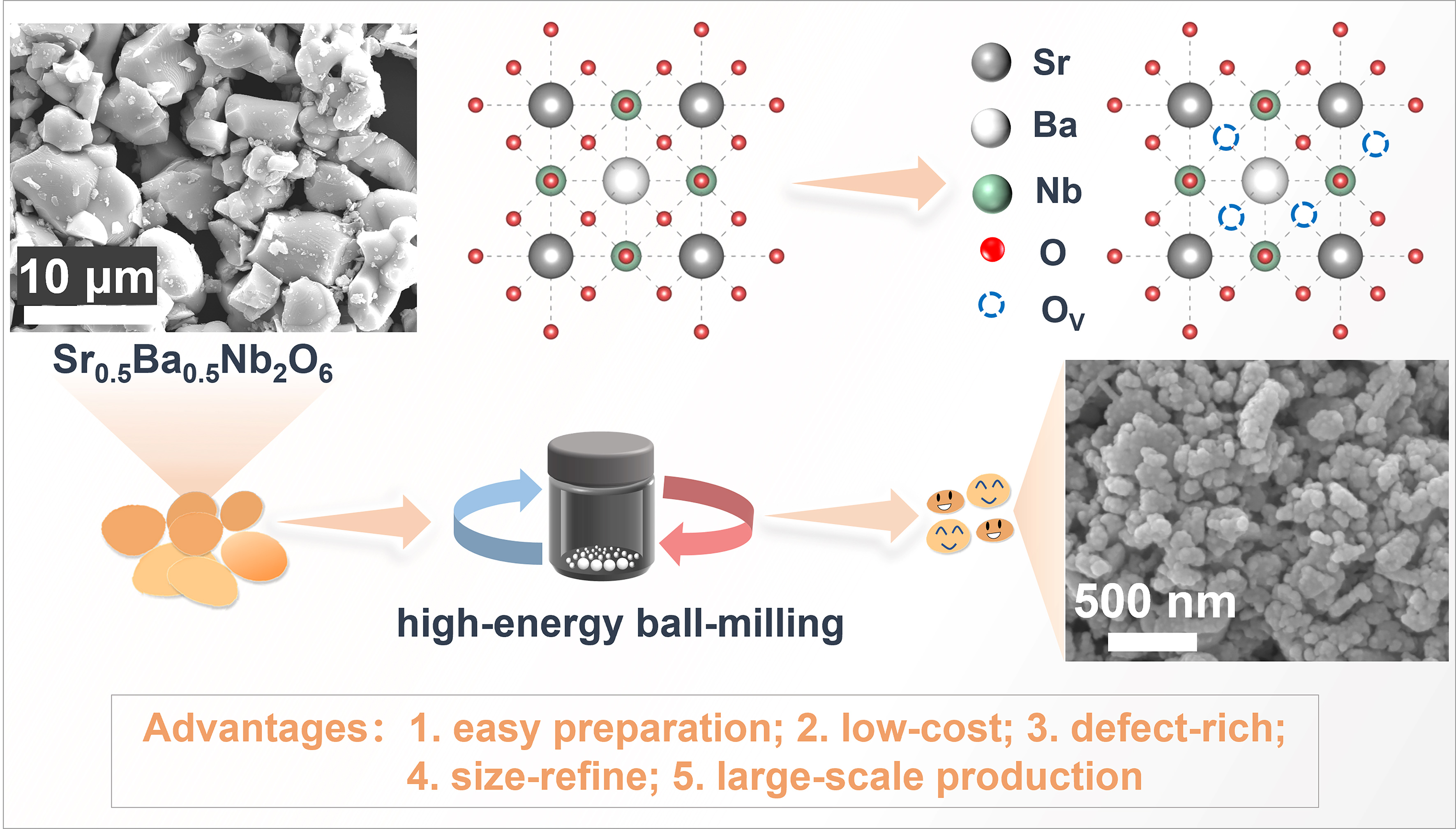
Herein, we present a facile mechanical nanostructuring strategy to transform bulk piezoceramics into high-performance piezocatalysts capable of efficiently activating PMS. Lead-free SBN piezoceramic, characterized by its high spontaneous polarization and chemical robustness, was selected as the model material. As shown in Figure 1, the micron-sized SBN powders were converted into defect-rich nanoscale piezocatalysts via high-energy ball milling (HBM), simultaneously enhancing polarization intensity and surface reactivity. Beyond the conventional hole-dominated piezocatalytic pathway, the nanostructured SBN effectively activated PMS under mechanical excitation, triggering a hybrid oxidation process in which defect-mediated charge dynamics promoted the simultaneous generation of •OH and SO4•-. The markedly enhanced catalytic performance of this integrated HBM-treated SBN piezocatalytic PMS activation system over pristine SBN demonstrates the efficacy of this straightforward and scalable strategy to convert bulk ceramics into functional catalysts, paving the way for practical and economical water treatment solutions.
MATERIALS AND METHODS
Chemicals
SrCO3 (99.95%), BaCO3 (99.95%), Nb2O5 (99.99%), potassium peroxymonosulfate (PMS, 2KHSO5·KHSO4·K2SO4, ≥ 42% KHSO5 basis), dimethyl sulfoxide (DMSO, ≥ 99.8%), methanol (MeOH,
Sample preparation
Strontium barium niobate (Sr0.5Ba0.5Nb2O6, SBN) piezoceramic powders were synthesized via a conventional solid-state reaction. Stoichiometric SrCO3, BaCO3, and Nb2O5 were homogenized by grinding for 30 min, followed by calcination at 1,300 °C for 4 h and natural cooling to room temperature to obtain SBN. The resulting powders were then subjected to high-energy wet ball milling with deionized water in a high-energy ball mill (Pulverisette 7, Fritsch & Co., Ltd., Germany) at 500 rpm in an intermittent mode (10 min milling followed by 10 min pause, repeated for a total milling duration of 12 h), yielding the high-energy ball-milled sample (SBN-HBM).
Characterizations
High-resolution X-ray diffraction (XRD, Empyrean, PANalytical, Netherlands) was employed to analyze the crystal structures of the synthesized SBN samples. The specific surface areas were determined from nitrogen adsorption-desorption isotherms at 300 K (ASAP 2460, Micromeritics, USA) using the Brunauer-Emmett-Teller (BET) method. Surface morphologies, lattice fringes, and elemental distributions were visualized by field-emission scanning electron microscopy (FE-SEM, Zeiss Merlin, Germany) and high-resolution transmission electron microscopy (HR-TEM, Tecnai F30, Thermo Fisher Scientific, USA). Local piezoelectric responses were assessed by piezoresponse force microscopy (PFM, Dimension Icon, Bruker, USA). A Pt-Ir scanning probe (tip radius ~25 nm; resonance frequency ~300 kHz; spring constant ~2.8 N·m-1) was used for PFM measurements on SBN-500 °C samples mounted on a Pt-coated silicon substrate, and a DC bias was swept from -10 to +10 V to acquire local piezoresponse hysteresis loops. Surface chemical compositions and oxidation states were analyzed by X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi, Thermo Fisher Scientific, USA). Reactive oxygen species and oxygen vacancies were identified by electron paramagnetic resonance (EPR, EMXPlus, Bruker, USA). The optical band gaps were estimated from UV-vis diffuse reflectance spectra (UV-3600 Plus, Hitachi, Japan) using BaSO4 as the reflectance reference. Piezoelectric current measurements were conducted on a CHI 760E electrochemical workstation (Shanghai Chenhua, China) in a three-electrode configuration, with ITO-coated SBN as the working electrode, Ag/AgCl as the reference electrode, and platinum as the counter electrode in 0.1 M Na2SO4 electrolyte. Briefly, 10 mg of SBN powder was dispersed in 1 mL of ethanol under ultrasonication to form a homogeneous suspension, which was then uniformly drop-cast onto a 25 × 40 mm ITO glass substrate with a fixed conductive area defined by conductive adhesive. The coated electrode was dried naturally in air for 12 h prior to measurements. A 45 kHz ultrasonic (US) generator (KQ-100VDE, Kunshan Ultrasonic Instrument Co., Ltd., China) was applied to provide mechanical excitation during the electrochemical measurements.
Catalytic activity tests
RhB was employed as the model pollutant, with all experiments conducted at 25 °C unless otherwise specified. Typically, 40 mg of catalyst was dispersed into 100 mL of RhB solution (5 mg/L) in a 200 mL beaker and magnetically stirred at 300 rpm for 30 min to establish adsorption-desorption equilibrium, and the adsorption kinetics curve was shown in Supplementary Figure 1. Subsequently, PMS (1 mM) was added to the suspension. The catalytic reaction was then initiated by US irradiation using a KQ-100VDE ultrasonic cleaner (Kunshan Ultrasonic Instrument Co., Ltd.) operating at 45 kHz frequency and 100 W power. At predetermined intervals, 5 mL aliquots were withdrawn and filtered through a 0.22 μm PES membrane to remove catalyst particles. The filtrate (2 mL, excluding negligible adsorption contributions) was immediately quenched with 100 μL of DMSO to terminate the reaction. Concentrations of dyes - including RhB, MO, and MB - were measured by UV-vis spectrophotometry (L9, Shanghai Yidian Analytical Instrument Co., Ltd.), while TC levels were quantified via high-performance liquid chromatography (HPLC, Beijing Jitian Instrument Co., Ltd.). Degradation kinetics were evaluated using the pseudo-first-order model, ln(C/C0) = -kt, where C, C0, and k denote residual concentration, initial concentration, and apparent rate constant, respectively. All experiments were conducted in duplicate to ensure the reliability and reproducibility of the results, with results reported as mean ± standard deviation (s.d.). All experiments followed identical procedures unless otherwise specified.
Radical scavenging experiments and EPR measurements
To identify the dominant reactive species in the SBN-HBM/PMS system, radical scavenging tests and EPR analyses were conducted. For quenching experiments, methanol (MeOH, 200 mM) was applied to simultaneously capture •OH and SO4•-, TBA (200 mM) was used as a selective •OH quencher, and EDTA
In vitro antibacterial experiments
The antibacterial activity against Escherichia coli (E. coli, ATCC 25922) was evaluated in three groups: Control (US only), SBN-HBM/US, and SBN-HBM/PMS/US. Bacterial suspensions were prepared in sterile Luria-Bertani (LB) medium with an initial concentration of 105 CFU/mL and subjected to US treatment
RESULTS AND DISCUSSION
Structural, morphological, and piezoresponse analysis
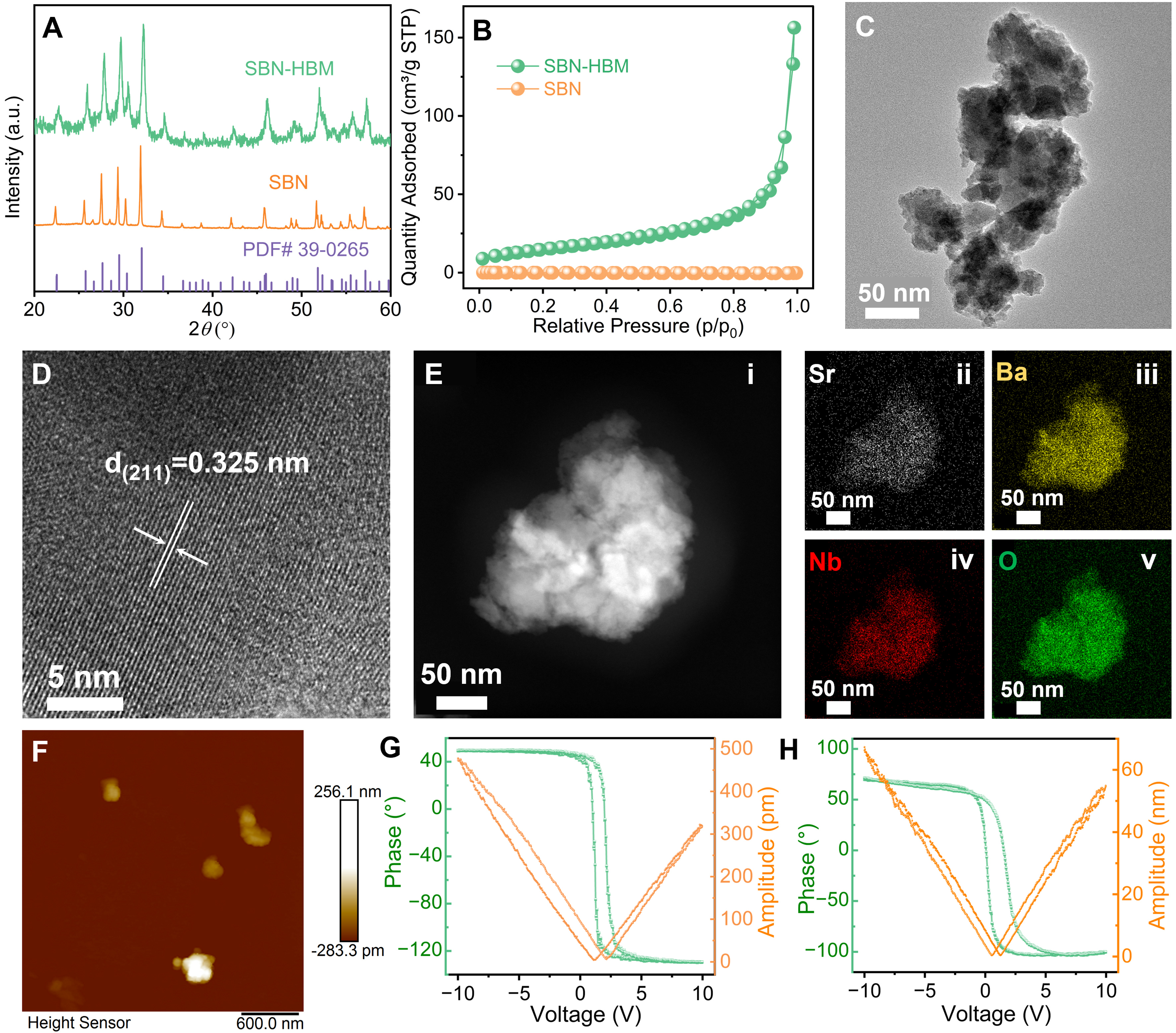
The SBN piezoceramic powders were synthesized via a conventional solid-state reaction, followed by HBM treatment to obtain the SBN-HBM sample. XRD patterns of pristine SBN and SBN-HBM are shown in Figure 2A. After HBM treatment, a noticeable peak broadening is observed, indicative of a reduced crystal size and decreased crystallinity, while both samples retain the crystal structure consistent with the JCPDS 39‑0265 phase[15,17]. The BET surface area analysis reveals a dramatic increase in specific surface area (SSA) from 0.2111 m2/g for pristine SBN to 54.2322 m2/g for SBN-HBM [Figure 2B], confirming the significant surface modification induced by HBM. SEM images [Figure 1] and particle size distribution analysis [Supplementary Figure 2] reveal that the mean grain size decreases from ~6.5 μm in pristine SBN to
Figure 2. (A) XRD patterns and (B) N2 adsorption-desorption isotherms of SBN and SBN-HBM; (C) TEM image, (D) HR-TEM image, (E) HADDF image, EDS elemental mapping, and (F) PFM topography image of SBN-HBM. Local phase-voltage hysteresis loop and amplitude-voltage butterfly loop of (G) SBN and (H) SBN-HBM. XRD: X-ray diffraction; SBN: Sr0.5Ba0.5Nb2O6; HBM: high-energy ball milling; TEM: transmission electron microscopy; HR: high-resolution; HADDF: high-angle annular dark-field; EDS: energy-dispersive X-ray spectroscopy; PFM: piezoresponse force microscopy; PDF: powder diffraction file.
Evaluation of piezocatalytic activity
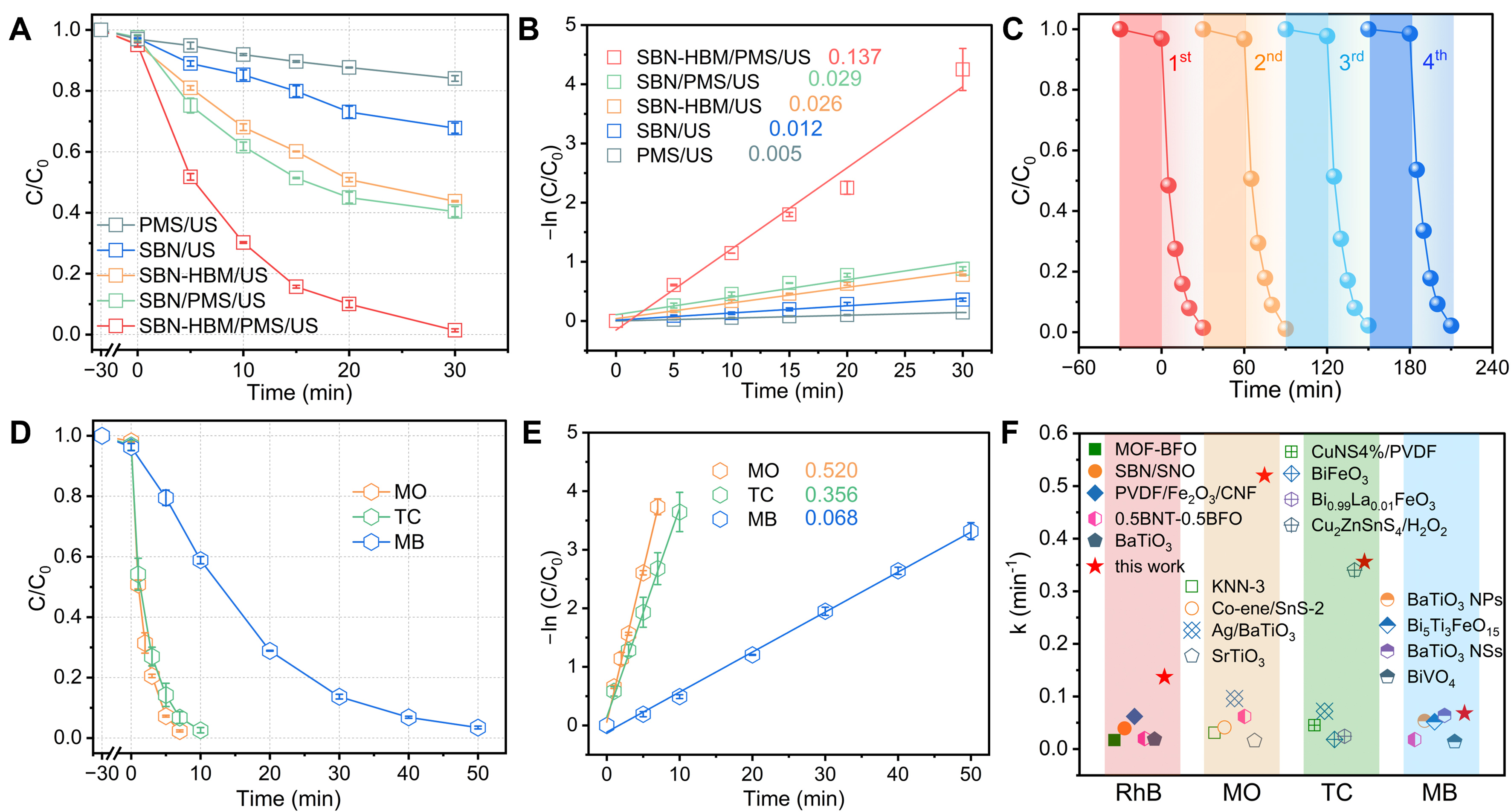
The piezocatalytic activity of the SBN samples was systematically evaluated through the degradation of various organic pollutants, including RhB, MO, MB, and TC. As illustrated in Figure 3A and B, RhB was selected as the model contaminant to evaluate the degradation efficiency under distinct catalytic scenarios, accompanied by kinetic analyses. In the absence of PMS, both pristine SBN and SBN-HBM displayed effective RhB removal under US irradiation, underscoring their inherent piezocatalytic activity. Quantitatively, pristine SBN facilitated 33% RhB degradation within 30 min, whereas SBN-HBM achieved a markedly higher removal of 56% under identical conditions. Correspondingly, the apparent rate constant (k) of SBN-HBM reached 0.026 min-1, nearly twice that of pristine SBN, thereby substantiating that HBM treatment effectively improves the piezocatalytic efficiency. In contrast, PMS alone achieves merely 15% degradation within 30 min due to mechanically induced PMS activation, corresponding to a k value of
Figure 3. (A) RhB degradation curves and (B) corresponding k values across various catalytic systems; (C) Recycling ability tests of the SBN-HBM/PMS system for RhB degradation; (D) Various organic pollutants degradation curves and (E) corresponding k values with SBN-HBM/PMS; (F) Comparison of k values from this study with previously reported piezocatalytic systems. Results are reported as the average ± SD (n = 3). RhB: Rhodamine B; SBN: Sr0.5Ba0.5Nb2O6; HBM: high-energy ball milling; PMS: peroxymonosulfate; SD: standard deviation; US: ultrasonic; MO: methyl orange; TC: tetracycline; MB: methylene blue; MOF: metal-organic framework; BFO: BiFeO3; SNO: Sr2Nb2O7; PVDF: poly(vinylidene fluoride); CNF: carbon nanofibers; BNT: Bi0.5Na0.5TiO3; KNN-3: (K, Na)NbO3-3; NPs: nanoparticles; NSs: nanosheets.
To further substantiate the applicability of the SBN-HBM/PMS system, its catalytic versatility was evaluated using a suite of organic pollutants with distinct molecular architectures, namely TC, MO, and MB [Figure 3D]. Despite their disparate electronic structures, the system exhibited rapid and nearly complete removal of each contaminant, achieving 98% degradation of MO within 7 min, 98% of TC within 10 min, and 97% of MB within 50 min. Meanwhile, quantitative kinetic analyses [Figure 3E] revealed apparent rate constants of 0.520 min-1 for MO, 0.356 min-1 for TC, and 0.068 min-1 for MB, corroborating its broad-spectrum catalytic capability and exceptional reaction kinetics. Benchmarking against state-of-the-art piezocatalytic systems [Figure 3F; detailed parameters in Supplementary Table 1] revealed that SBN-HBM/PMS outperforms most reported counterparts. This superior performance underscores the efficacy of the defect-polarization-coupled PMS activation strategy, highlighting its potential as a robust and scalable route for high-performance piezocatalytic remediation.
Mechanistic investigation
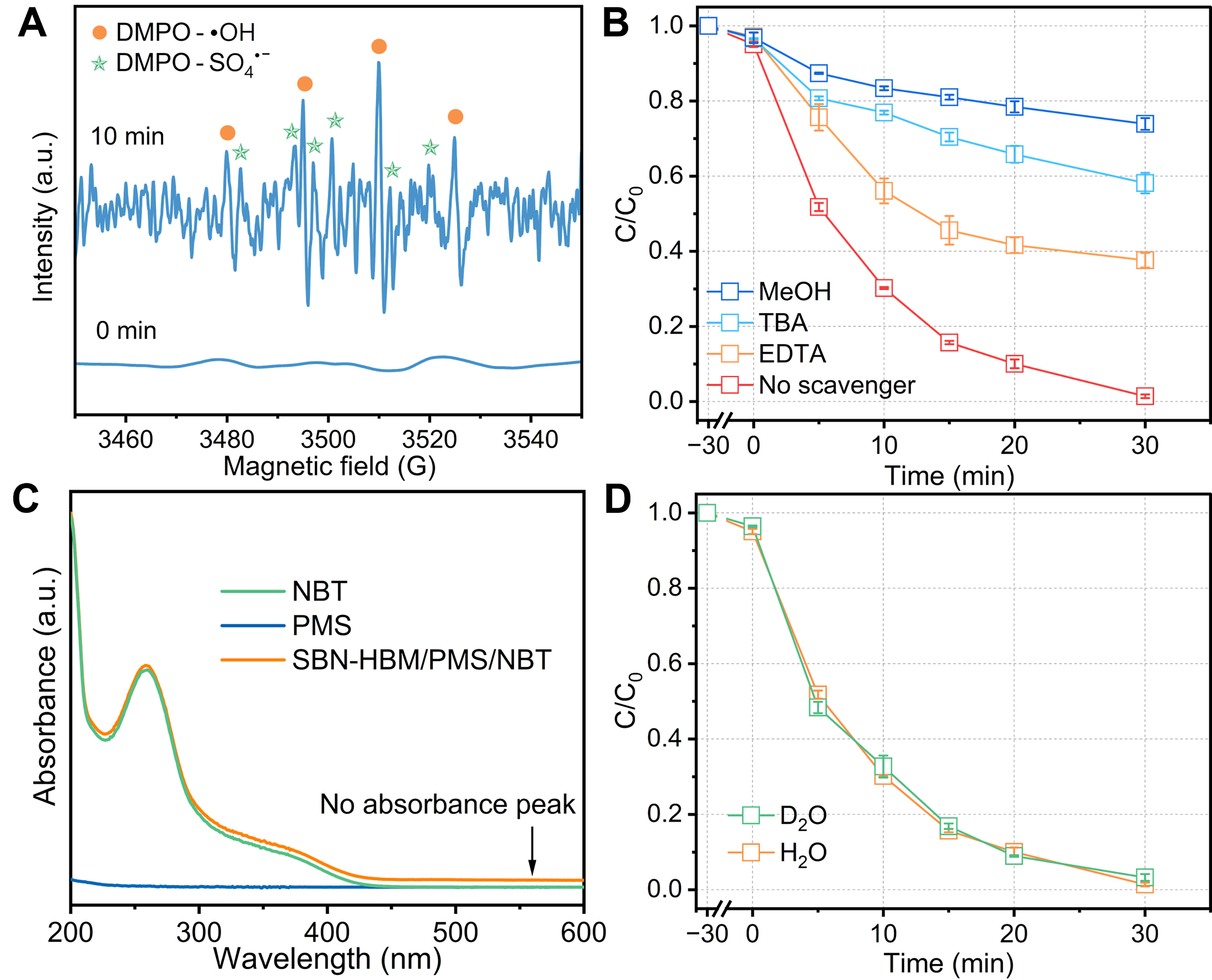
Unraveling the mechanism of the SBN-HBM/PMS system hinges on identifying the reactive species responsible for pollutant degradation. EPR spectroscopy, using DMPO as the spin-trapping agent, detected no radical signatures in the absence of US excitation [Figure 4A]. Upon US activation, distinct DMPO-•OH and DMPO-SO4•- adduct signals emerged within 10 min, which confirmed the generation of •OH and SO4•- radicals and the indispensable role of mechanical excitation for radical generation in this system[24]. The functional roles of these species were further elucidated through quenching experiments [Figure 4B]. Inhibition of RhB degradation by EDTA (37%, h+ scavenger), TBA (58%, •OH scavenger), and MeOH (74%, •OH/SO4•- scavenger) indicated that •OH dominates the oxidative pathway, with SO4•- and h+ as auxiliary participants[34]. In addition, the NBT assay exhibited no characteristic formazan absorption peaks in the SBN-HBM/PMS/NBT system [Figure 4C], excluding O2•- as a kinetically significant intermediate[35,36]. Similarly, the contribution of 1O2 was investigated through D2O/H2O solvent exchange, leveraging its prolonged lifetime in D2O[37]. The negligible variation in degradation kinetics [Figure 4D] confirmed that 1O2 plays no substantive role[38].
Figure 4. (A) EPR signal of •OH/SO4•- over SBN-HBM/PMS system; (B) RhB degradation curves of the SBN-HBM/PMS system in the presence of different scavengers; (C) UV-vis spectra of NBT systems; (D) RhB degradation curves in H2O and D2O. Results are reported as the average ± SD (n = 3). EPR: Electron paramagnetic resonance; SBN: Sr0.5Ba0.5Nb2O6; HBM: high-energy ball milling; PMS: peroxymonosulfate; RhB: Rhodamine B; UV-vis: ultraviolet-visible; NBT: 4-nitro blue tetrazolium chloride; D2O: deuterium oxide; SD: standard deviation; DMPO: 5,5-dimethyl-1-pyrroline N-oxide; TBA: tert-butyl alcohol; EDTA: ethylenediaminetetraacetic acid disodium salt.
Hence, these results collectively define a mechanistic framework in which •OH governs the primary oxidative pathway, synergistically aided by SO4•- and h+. The exceptional activity of the SBN-HBM/PMS system arises from mechanically induced radical processes, underscoring the intrinsic efficacy of the defect-polarization-driven piezocatalytic PMS activation route.
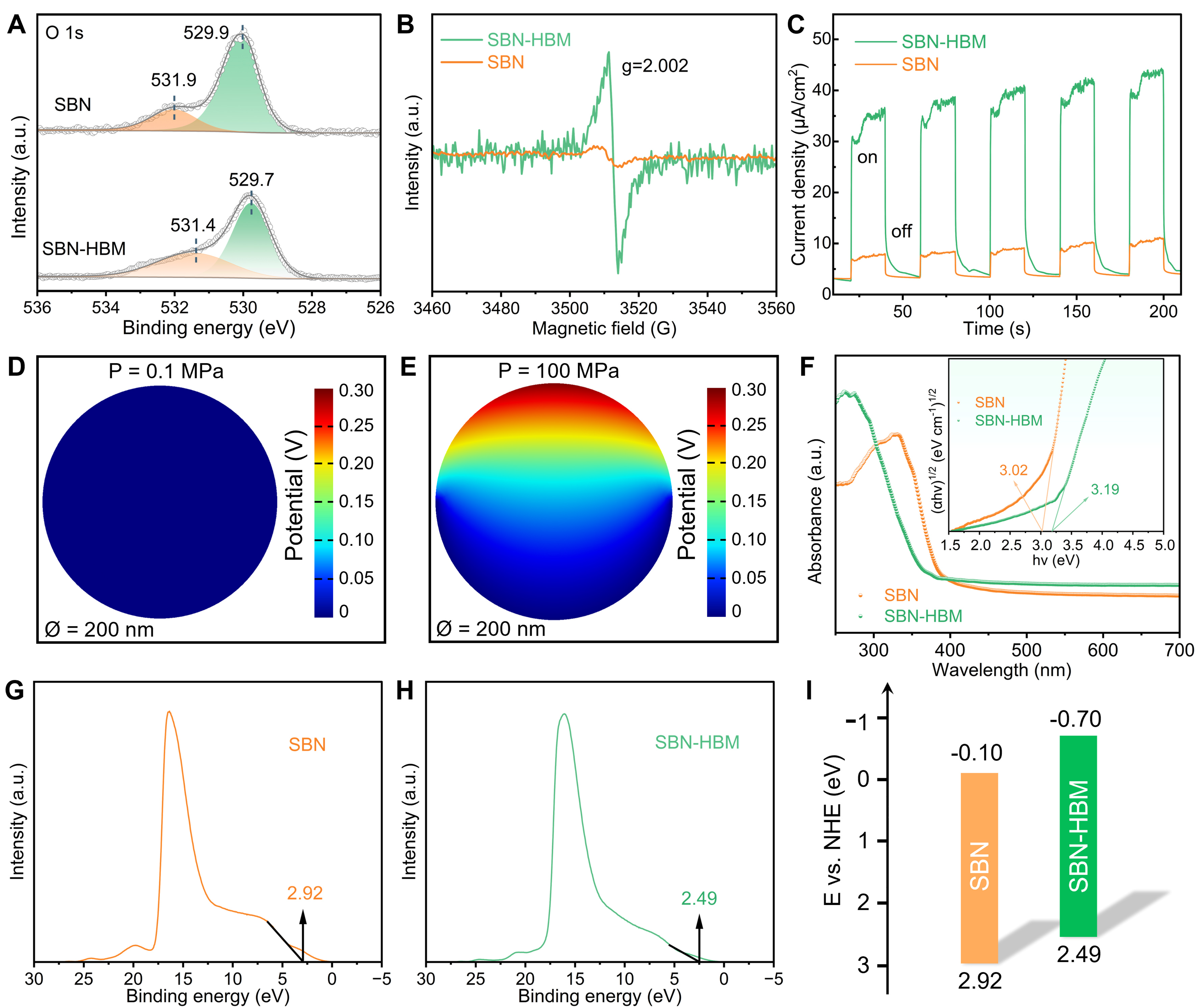
While the radical analysis clarifies the dominant oxidative pathways, the superior catalytic activity of SBN-HBM suggests the involvement of intrinsic structural factors that govern charge excitation and transfer. In particular, the interplay between lattice defects and piezoelectric polarization plays a decisive role, as oxygen vacancies modulate band energetics and simultaneously amplify piezoelectric polarization, thereby facilitating more efficient charge mobility[39]. To verify this correlation, the oxygen vacancy states were systematically examined through XPS and EPR analyses, providing direct insight into the defect landscape underpinning the catalytic performance. Figure 5A displays the high-resolution XPS spectra of the O 1s region for both SBN and SBN-HBM samples. In each case, the O 1s peak can be deconvoluted into two components: a low-binding-energy peak near 529.9 eV (529.7 eV for SBN-HBM) corresponding to lattice oxygen, and a higher-binding-energy peak near 531.9 eV (531.4 eV for SBN-HBM) associated with oxygen vacancy-related species[17]. While the peak assignments remain consistent between samples, the OV component in SBN-HBM exhibits a discernible shift toward a lower binding energy position accompanied by a notable increase in relative peak area, reflecting a higher density of oxygen vacancies introduced by high-energy ball milling[40]. EPR measurements further confirm this conclusion [Figure 5B]. Both samples display a characteristic resonance at the Landé g factor of 2.002, indicative of unpaired electrons trapped at oxygen vacancies[41]. The significantly enhanced EPR signal intensity in SBN-HBM provides direct evidence for the increased oxygen vacancy concentration, in strong agreement with the XPS analysis. Furthermore, transient piezoelectric current measurements were conducted to evaluate the charge response under mechanical excitation. As shown in Figure 5C, both samples exhibit periodic current fluctuations under US on-off cycles. Notably, SBN-HBM delivers a substantially higher piezo-current density than pristine SBN, reflecting its more efficient generation, separation, and migration of charge carriers[42].
Figure 5. (A) XPS O 1s spectra, (B) EPR spectra, (C) the piezo-current responses for SBN and SBN-HBM. FEM simulation of the piezoelectric potential distribution in SBN-HBM under a cavitation pressure of (D) 0.1 and (E) 100 MPa; (F) UV-vis absorption spectra (inset: Tauc plot), and UPS spectra of (G) SBN and (H) SBN-HBM; (I) Band schematic diagram for SBN and SBN-HBM. XPS: X-ray photoelectron spectroscopy; EPR: electron paramagnetic resonance; SBN: Sr0.5Ba0.5Nb2O6; HBM: high-energy ball milling; FEM: finite-element method; UV-vis: ultraviolet-visible.
To gain deeper insight into the underlying piezoelectric mechanism, finite-element method (FEM) simulations were performed to evaluate the potential distribution in SBN-HBM under varying mechanical pressures. Considering that ambient pressure is typically around 105 Pa, while acoustic cavitation during ultrasonication can induce localized pressures approaching ~108 Pa, representative simulations were conducted under 0.1 and 100 MPa to reflect these contrasting conditions. A particle diameter (Ø) of 200 nm was used to match the characteristic dimension of SBN-HBM. As illustrated in Figure 5D, the applied low pressure (0.1 MPa) yielded negligible piezoelectric potential, indicating insufficient mechanical deformation to activate a strong piezoelectric response. In sharp contrast, a substantial potential output of 0.3 V was generated under 100 MPa [Figure 5E], confirming that high mechanical stress is essential to effectively polarize the material and induce an internal electric field. These simulation outcomes are in excellent agreement with the experimental piezoelectric current results, reinforcing the critical role of mechanical excitation intensity in modulating charge separation efficiency.
Subsequently, a systematic investigation into the band structures of SBN and SBN-HBM was conducted to understand the electronic basis of the enhanced piezoelectric response. As shown in Figure 5F, UV-vis diffuse reflectance spectroscopy (DRS) combined with Tauc plot fitting revealed optical bandgap energies (Eg) of 3.02 eV for pristine SBN and 3.19 eV for SBN-HBM. The increased Eg of SBN-HBM, arising from its refined nanoscale dimensions, reflects the modified electronic landscape under strong quantum confinement[15,43]. More importantly, the enhanced piezoelectric response and defect-driven band alignment synergistically promote charge separation and transfer, underpinning the superior piezocatalytic activity. Ultraviolet photoelectron spectroscopy (UPS) measurements [Figure 5G and H] further determined the valence band maximum (EV) to be 2.92 eV and 2.49 eV versus the normal hydrogen electrode (NHE) for SBN and SBN-HBM, respectively. Subsequently, based on the fundamental band position relationship (Eg = EV EC), the conduction band minimum (EC) was calculated to be -0.10 eV for SBN and -0.70 eV for SBN-HBM[44]. This band alignment is further illustrated by a schematic diagram in Figure 5I, which delineates the relative shifts in valence and conduction band positions induced by oxygen vacancies and nanoscale effects. Such a band structure reconfiguration, coupled with the defect-mediated charge polarization, provides a mechanistic basis for the observed improvement in piezocatalytic PMS activation.
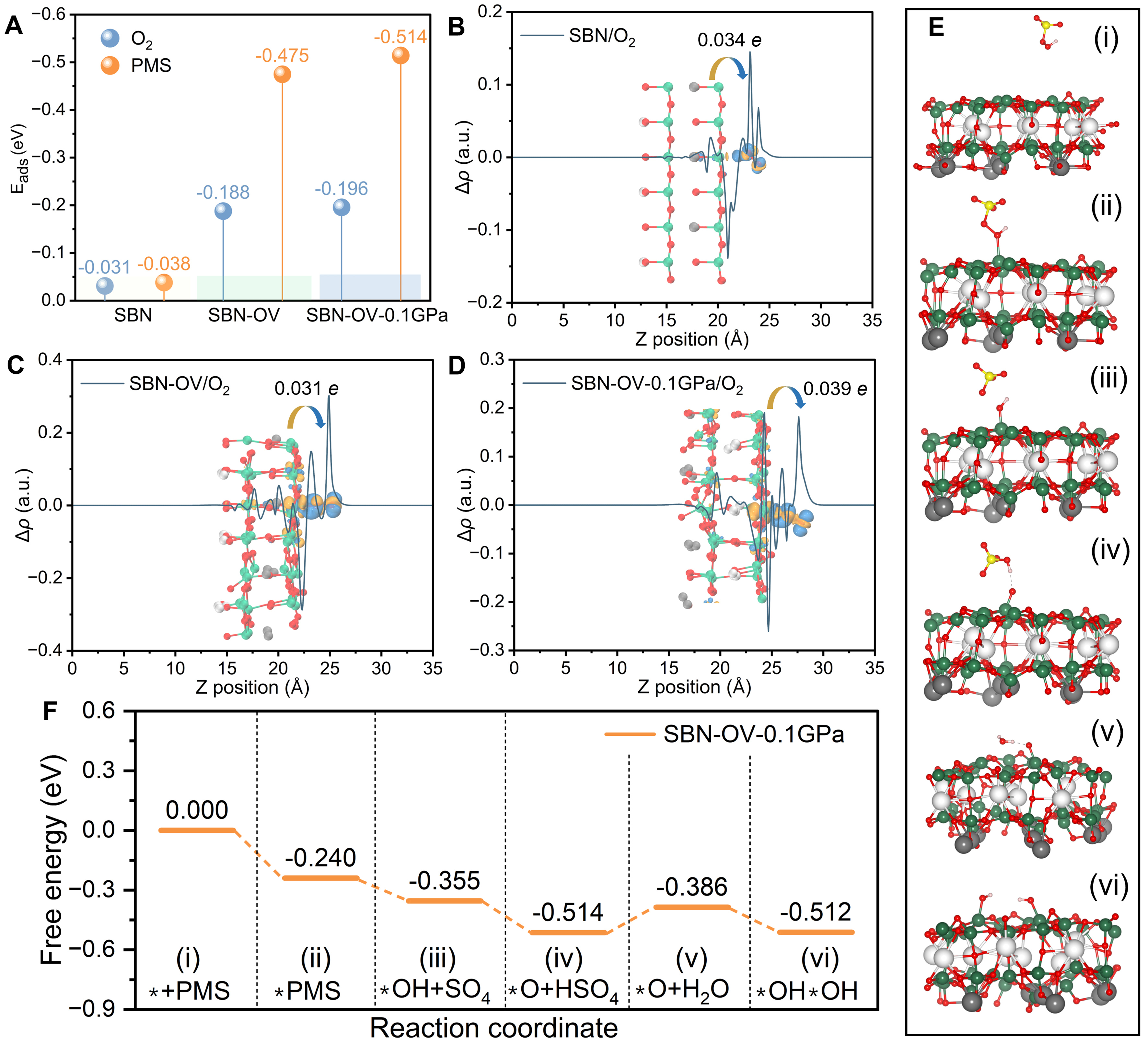
To gain atomic-level insight into the interfacial processes, first-principles calculations were conducted on the system. The computational methodology is detailed in the Supplementary Materials. Key parameters - including adsorption energies [Eads, Figure 6A], charge density differences for O2 adsorption [Figure 6B-D], and reaction intermediates of defective SBN (SBN-OV) piezo-activated PMS system [Figure 6E] along with their energy profiles [Figure 6F] - were systematically examined to elucidate the interfacial reaction mechanism. As shown in Figure 6A, compared with pristine SBN (defect-free), the SBN-OV presents a clear enhancement in Eads of O2 (from -0.031 to -0.188 eV) and PMS (from -0.038 to -0.475 eV). Meanwhile, the Eads will be further boosted for SBN-OV (-0.196 eV for O2 and -0.514 eV for PMS) when an external force is applied, indicating the effective piezoelectric effect for regulating interfacial processes. Furthermore, the charge density differences for O2 adsorption on SBN, SBN-OV, and SBN-OV-0.1GPa were calculated to gain insight into the interfacial electronic interactions. The corresponding charge displacement curves, revealing the net electron transfer volumes, are presented in Supplementary Figure 4. As shown in Figure 6B, pristine SBN exhibits a noticeable electron depletion, whereas the adsorbed O2 gains electrons, resulting in an interfacial charge transfer of 0.034 e. In the SBN-OV system, a comparable charge transfer of 0.031 e to O2 is observed [Figure 6C], along with additional internal electron redistribution induced by oxygen vacancies. In contrast, SBN-OV-0.1GPa shows a more enhanced charge transfer of 0.039 e from SBN-OV to O2 [Figure 6D], which can be attributed to the enhanced polarization associated with the strong piezoelectric effect. The formation pathway of reactive species on the SBN-OV catalyst surface under mechanical stress (0.1 GPa) was systematically investigated to understand the piezo-activated PMS process. Free radical generation was initiated by PMS adsorption, followed by a sequence of surface reactions, where * denotes the adsorption site on the catalyst. The optimized free-energy profile for the piezo-activated system follows the sequence * + PMS (i) → *PMS (ii) → *OH + SO4 (iii) → *O + HSO4 (iv) → *O + H2O (v) → *OH*OH (vi) [Figure 6E and F], with the calculated free energies for each step being 0.000 → -0.240 → -0.355 → -0.514 → -0.386 → -0.512 eV. PMS adsorption (i → ii) is exergonic (ΔG = -0.240 eV), reflecting strong binding facilitated by the polarized surface under mechanical stress. The subsequent O-O bond cleavage leading to *OH + SO4 (ii → iii) is further downhill (ΔG = -0.115 eV), indicating spontaneous radical formation. Formation of *O + HSO4 (iii → iv) is the most exergonic step (ΔG = -0.159 eV), highlighting the favorable generation of high-energy surface-bound oxygen species enhanced by piezoelectric polarization. The reaction of *O with water from the solution (iv → v) shows a slight endergonic trend (ΔG = +0.128 eV), corresponding to a transient transition state (TS) associated with proton transfer from water. This TS is readily overcome under experimental conditions to form the final *OH*OH intermediate (v → vi, ΔG = -0.126 eV), stabilizing the system. Overall, the pathway is thermodynamically feasible, and the piezoelectric effect under mechanical stress promotes electron transfer and surface reactivity, enabling efficient PMS activation and continuous generation of reactive oxygen species on SBN-OV.
Figure 6. (A) Calculated adsorption energies of O2 and PMS molecules on SBN (without external stress), SBN-OV (without external stress), and SBN-OV-0.1 GPa (with an external stress of 0.1 GPa) systems; (B-D) Charge density difference diagrams and corresponding local integration curves for O2 adsorption on different SBN systems; (E) Structures of reaction intermediates involved in free radical generation in the SBN-OV piezo-activated PMS system under an external stress of 0.1 GPa, and (F) the associated energy profiles. PMS: Peroxymonosulfate; SBN: Sr0.5Ba0.5Nb2O6; SBN-OV: reaction intermediates of defective SBN.
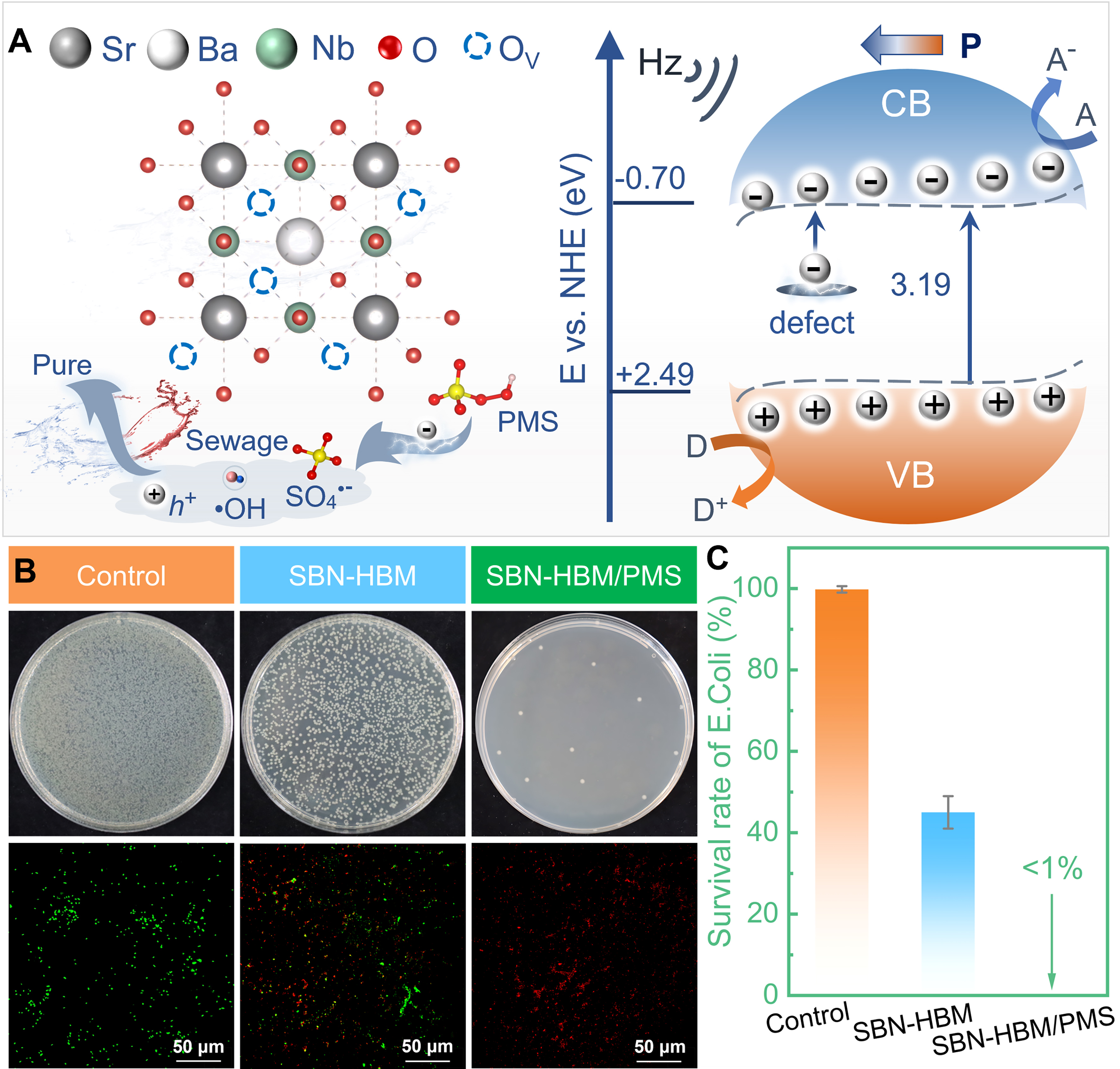
Based on integrated experimental evidence and theoretical analysis, a mechanistic model for piezoelectric PMS activation by SBN-HBM is illustrated in Figure 7A. High-energy ball milling induces abundant surface oxygen vacancies, which create defect states near the conduction band, tailoring the electronic structure and facilitating charge carrier transport[45,46]. Upon mechanical excitation, piezoelectric polarization in the SBN-HBM lattice generates a built-in electric field, promoting efficient separation and directional migration of electrons and holes [Equation 1]. Electrons are excited into the conduction band and partially transferred through defect-assisted pathways[47]. Concurrently, h+ contributes to pollutant degradation through dual routes: direct oxidation or by reacting with water to form •OH [Equations 2 and 3][48]. Meanwhile, PMS molecules adsorbed on the catalyst surface undergo homolytic cleavage upon interaction with piezo-induced electrons, yielding both •OH and SO4•- [Equations 4 and 5][49]. These reactive oxygen species, together with piezo-generated h+, synergistically enhance the oxidative degradation of organic contaminants [Equations 6 and 7].
Figure 7. (A) Schematic illustration of the piezo-driven PMS activation mechanism mediated by SBN-HBM; (B) Photographs of E. coli colonies from different treatment groups and corresponding live/dead staining images; (C) Bacterial survival rates of different groups. Results are reported as the average ± SD (n = 3). PMS: Peroxymonosulfate; SBN: Sr0.5Ba0.5Nb2O6; E. coli: Escherichia coli; SD: standard deviation; NHE: normal hydrogen electrode; CB: conduction band; VB: valence band; HBM: high-energy ball milling.
Overall, this integrated mechanism underscores the cooperative interplay between defect engineering and piezoelectric charge modulation, offering mechanistic insights into the design of advanced piezocatalysts for energy-efficient environmental remediation.
In addition to the efficient degradation of organic pollutants, the antibacterial performance of the system was also investigated using E. coli as a model bacterium. Three groups were tested: Control (US only), SBN-HBM, and SBN-HBM/PMS. As shown in Figure 7B, the control group exhibited dense bacterial colonies and predominantly green fluorescence under fluorescence microscopy, indicating high cell viability. In contrast, the SBN-HBM/US treatment led to a substantial reduction in bacterial colonies and a mixture of green and red fluorescence signals, suggesting partial inactivation. Notably, the SBN-HBM/PMS/US group showed almost complete disappearance of colonies and dominant red fluorescence, corresponding to a bacterial inactivation rate exceeding 99%. This was further supported by survival rate histograms shown in Figure 7C, which quantitatively demonstrate the high antibacterial efficacy of the SBN-HBM/PMS/US system.
CONCLUSIONS
This study demonstrates a simple mechanical nanostructuring strategy to convert bulk SBN piezoceramics into high-performance piezocatalysts for efficient PMS activation. The HBM treatment induces nanosizing and oxygen vacancies, which synergistically enhance piezoelectric response and charge carrier separation, leading to effective PMS activation for accelerated degradation of organic pollutants and superior antibacterial activity. Mechanistic analysis indicates that defect-polarization coupling plays a key role in modulating the electronic structure and facilitating redox reactions. Overall, this work provides a scalable and environmentally friendly approach to improve the intrinsic catalytic activity of bulk ceramics, offering potential applications in decentralized water treatment and disinfection technologies.
DECLARATIONS
Acknowledgements
The authors extend their gratitude to Scientific Compass (www.shiyanjia.com) for the DRS and TEM measurements.
Authors’ contributions
The conception and design of the work: Dai, J.; Long, Y.; Wang, D.; Chang, Y.
The acquisition and analysis of data: Dai, J.; Fan, Z.; Cheng, M.; Wang, B.; Huang, F.; Yue, W.; Deng, Y.
The theoretical calculations: Zhu, Y.; Fan, Z.
The interpretation of data: Dai, J.; Evcin, A.; Mohamad, A. A.
The writing and revising: Dai, J.; Long, Y.; Wang, D.; Chang, Y.
The supervision: Long, Y.; Wang, D.; Chang, Y.
Availability of data and materials
The Supplementary Materials associated with this article is available from the online version. Further data used to support the findings of this study are available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was financially supported by the National Natural Science Foundation of China (Nos. 52472122, 52372106 and 52072092), the National Key R&D Program of China (No. 2024YFB3211800), the University Key Research Project in Guangdong Province (No. 2022ZDZX4103).
Conflicts of interest
Wang, D. is an Associate Editor of the journal Microstructures, but was not involved in any steps of editorial processing, notably including reviewer selection, manuscript handling, and decision making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Sobek, A.; Abel, S.; Sanei, H.; et al. Organic matter degradation causes enrichment of organic pollutants in hadal sediments. Nat. Commun. 2023, 14, 2012.
2. Liu, J.; Qi, W.; Xu, M.; Thomas, T.; Liu, S.; Yang, M. Piezocatalytic techniques in environmental remediation. Angew. Chem. Int. Ed. 2023, 62, e202213927.
3. Hong, K.; Xu, H.; Konishi, H.; Li, X. Piezoelectrochemical effect: a new mechanism for azo dye decolorization in aqueous solution through vibrating piezoelectric microfibers. J. Phys. Chem. C. 2012, 116, 13045-51.
4. Ali, A.; Chen, L.; Nasir, M. S.; Wu, C.; Guo, B.; Yang, Y. Piezocatalytic removal of water bacteria and organic compounds: a review. Environ. Chem. Lett. 2022, 21, 1075-92.
5. Chen, X.; Wang, J.; Wang, Z.; et al. Low-frequency mechanical energy in the environment for energy production and piezocatalytic degradation of organic pollutants in water: a review. J. Water Process Eng. 2023, 56, 104312.
6. Bai, Y.; Zhao, J.; Lv, Z.; Lu, K. Enhanced piezo-phototronic effect of ZnO nanorod arrays for harvesting low mechanical energy. Ceram. Int. 2019, 45, 15065-72.
7. Wang, B.; Zhang, Q.; He, J.; Huang, F.; Li, C.; Wang, M. Co-catalyst-free large ZnO single crystal for high-efficiency piezocatalytic hydrogen evolution from pure water. J. Energy Chem. 2022, 65, 304-11.
8. Liu, H.; Wang, J.; Odunmbaku, O.; et al. Highly recyclable and efficient piezocatalyst of Nafion decorated MoS2 with hierarchical structure for organic pollutants degradation. Sep. Purif. Technol. 2023, 320, 124142.
9. Mondal, S.; Dilly Rajan, K.; Patra, L.; Rathinam, M.; Ganesh, V. Sulfur vacancy-induced enhancement of piezocatalytic H2 production in MoS2. Small 2025, 21, e2411828.
10. Truong Hoang, Q.; Huynh, K. A.; Nguyen Cao, T. G.; et al. Piezocatalytic 2D WS2 nanosheets for ultrasound-triggered and mitochondria-targeted piezodynamic cancer therapy synergized with energy metabolism-targeted chemotherapy. Adv. Mater. 2023, 35, e2300437.
11. Porwal, C.; Sharma, M.; Gaur, A.; et al. Effect of surface/bulk polarization on piezocatalysis using BaTiO3. J. Mater. Sci.: Mater. Electron. 2024, 35, 573.
12. Xue, K.; Jiang, Y.; Mofarah, S. S.; et al. Composition-driven morphological evolution of BaTiO3 nanowires for efficient piezocatalytic hydrogen production. Chemosphere 2023, 338, 139337.
13. Wang, Y.; Yang, M.; Cui, K.; et al. Band engineering of BiFeO3 nanosheet for boosting hydrogen evolution by synergetic piezo-photocatalysis. ACS Sustainable Chem. Eng. 2024, 12, 2300-12.
14. Amdouni, W.; Fricaudet, M.; Otoničar, M.; et al. BiFeO3 nanoparticles: the “holy-grail” of piezo-photocatalysts? Adv. Mater. 2023, 35, e2301841.
15. Liao, J.; Lv, X.; Sun, X. X.; et al. Boosting piezo-catalytic activity of KNN-based materials with phase boundary and defect engineering. Adv. Funct. Mater. 2023, 33, 2303637.
16. Dai, J.; Shao, N.; Zhang, S.; et al. Enhanced piezocatalytic activity of Sr0.5Ba0.5Nb2O6 nanostructures by engineering surface oxygen vacancies and self-generated heterojunctions. ACS Appl. Mater. Interfaces 2021, 13, 7259-67.
17. Chen, W.; Chen, Q.; Song, F.; et al. Engineering heterostructured piezoelectric nanorods with rich oxygen vacancy-mediated piezoelectricity for ultrasound-triggered piezocatalytic cancer therapy. Adv. Funct. Mater. 2024, 34, 2405929.
18. Meng, N.; Liu, W.; Jiang, R.; et al. Fundamentals, advances and perspectives of piezocatalysis: a marriage of solid-state physics and catalytic chemistry. Prog. Mater. Sci. 2023, 138, 101161.
19. Zhang, Q.; Jia, Y.; Wu, W.; et al. Review on strategies toward efficient piezocatalysis of BaTiO3 nanomaterials for wastewater treatment through harvesting vibration energy. Nano Energy 2023, 113, 108507.
20. Mani, A. D.; Li, J.; Wang, Z.; et al. Coupling of piezocatalysis and photocatalysis for efficient degradation of methylene blue by Bi0.9Gd0.07La0.03FeO3 nanotubes. J. Adv. Ceram. 2022, 11, 1069-81.
21. Tu, S.; Guo, Y.; Zhang, Y.; et al. Piezocatalysis and piezo-photocatalysis: catalysts classification and modification strategy, reaction mechanism, and practical application. Adv. Funct. Mater. 2020, 30, 2005158.
22. Li, H. R.; Chen, Y. M.; Jin, C. C.; et al. High-performance piezocatalytic hydrogen evolution over bismuth oxyhalides with halogen-dependent piezoelectricity and surface activity. Rare Met. 2025, 44, 5475-85.
23. Su, C. J.; Li, C. L.; Wang, W. H. Efficient piezocatalytic activation of peroxydisulfate over Bi2Fe4O9: thickness-dependent synergy effect between peroxydisulfate activation and piezocatalysis. Rare Met. 2023, 42, 4005-14.
24. Yang, J.; Zhang, M.; Chen, M.; Zhou, Y.; Zhu, M. Oxygen vacancies in piezoelectric ZnO twin-mesocrystal to improve peroxymonosulfate utilization efficiency via piezo-activation for antibiotic ornidazole removal. Adv. Mater. 2023, 35, e2209885.
25. Yu, C.; He, J.; Lan, S.; Guo, W.; Zhu, M. Enhanced utilization efficiency of peroxymonosulfate via water vortex-driven piezo-activation for removing organic contaminants from water. Environ. Sci. Ecotechnol. 2022, 10, 100165.
26. Li, Z.; Lan, S.; Zhu, M. Piezoelectricity activates persulfate for water treatment: a perspective. Environ. Sci. Ecotechnol. 2024, 18, 100329.
27. Li, C.; Liu, W.; Chen, X.; Li, L.; Lan, S.; Zhu, M. Adsorption and activation of peroxymonosulfate on BiOCl for carbamazepine degradation: the role of piezoelectric effect. Chin. Chem. Lett. 2024, 35, 109652.
28. Zhao, X.; Zhang, Z. Heterogeneous peroxymonosulfate-based advanced oxidation mechanisms: new wine in old bottles? Environ. Sci. Technol. 2025, 59, 5913-24.
29. Dai, J.; Zhu, Y.; Dai, D.; et al. Defect-polarization synergy unlocks sustained nonradical piezocatalysis via iron redox cycling. Adv. Funct. Mater. 2025, 36, e18904.
30. Liu, W.; Fu, P.; Zhang, Y.; Xu, H.; Wang, H.; Xing, M. Efficient hydrogen production from wastewater remediation by piezoelectricity coupling advanced oxidation processes. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2218813120.
31. Deng, X.; Chen, P.; Cui, R.; et al. Dynamic reconfiguration and local polarization of NiFe-layered double hydroxide-Bi2MoO6-x heterojunction for enhancing piezo-photocatalytic nitrogen oxidation to nitric acid. Adv. Sci. 2024, 11, e2401667.
32. Wang, C.; Hu, C.; Chen, F.; Ma, T.; Zhang, Y.; Huang, H. Design strategies and effect comparisons toward efficient piezocatalytic system. Nano Energy 2023, 107, 108093.
33. Lan, S.; Chen, Y.; Zeng, L.; Ji, H.; Liu, W.; Zhu, M. Piezo-activation of peroxymonosulfate for benzothiazole removal in water. J. Hazard. Mater. 2020, 393, 122448.
34. Long, Y.; Huang, Y.; Wu, H.; Shi, X.; Xiao, L. Peroxymonosulfate activation for pollutants degradation by Fe-N-codoped carbonaceous catalyst: Structure-dependent performance and mechanism insight. Chem. Eng. J. 2019, 369, 542-52.
35. Long, Y.; Dai, J.; Zhao, S.; Su, Y.; Wang, Z.; Zhang, Z. Atomically dispersed cobalt sites on graphene as efficient periodate activators for selective organic pollutant degradation. Environ. Sci. Technol. 2021, 55, 5357-70.
36. Long, Y.; Huang, S.; Sun, J.; Peng, D.; Zhang, Z. Markedly boosted peroxymonosulfate- and periodate-based Fenton-like activities of iron clusters on sulfur/nitrogen codoped carbon: key roles of a sulfur dopant and compared activation mechanisms. Sci. Total. Environ. 2023, 855, 158752.
37. Long, Y.; Xiao, G.; Peng, D.; Li, H.; Dai, J. Constructing atomic Co sites into defective carbon for enhanced degradation of sulfamethoxazole via catalytic generation of Co(IV)=O and radicals. Mater. Lett. 2025, 382, 137923.
38. Wu, Q. Y.; Yang, Z. W.; Wang, Z. W.; Wang, W. L. Oxygen doping of cobalt-single-atom coordination enhances peroxymonosulfate activation and high-valent cobalt-oxo species formation. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2219923120.
39. Dong, H.; Zhou, Y.; Wang, L.; Chen, L.; Zhu, M. Oxygen vacancies in piezocatalysis: a critical review. Chem. Eng. J. 2024, 487, 150480.
40. Cai, H.; Chen, F.; Hu, C.; et al. Oxygen vacancies mediated ultrathin Bi4O5Br2 nanosheets for efficient piezocatalytic peroxide hydrogen generation in pure water. Chin. J. Catal. 2024, 57, 123-32.
41. Liu, D.; Zhang, J.; Jin, C.; et al. Insight into oxygen-vacancy regulation on piezocatalytic activity of (Bi1/2Na1/2)TiO3 crystallites: experiments and first-principles calculations. Nano Energy 2022, 95, 106975.
42. Li, S.; Zhao, Z.; Li, J.; et al. Mechanically induced highly efficient hydrogen evolution from water over piezoelectric SnSe nanosheets. Small 2022, 18, e2202507.
43. Bandi, S.; Vidyasagar, D.; Adil, S.; Singh, M. K.; Basu, J.; Srivastav, A. K. Crystallite size induced bandgap tuning in WO3 derived from nanocrystalline tungsten. Scr. Mater. 2020, 176, 47-52.
44. Miao, Y.; Tian, W.; Han, J.; et al. Oxygen vacancy-induced hydroxyl dipole reorientation in hydroxyapatite for enhanced piezocatalytic activity. Nano Energy 2022, 100, 107473.
45. Wang, Y. C.; Wu, J. M. Effect of controlled oxygen vacancy on H2-production through the piezocatalysis and piezophototronics of ferroelectric R3C ZnSnO3 nanowires. Adv. Funct. Mater. 2019, 30, 1907619.
46. Sha, Z.; Hu, C.; Tu, S.; Li, T.; Chen, F.; Huang, H. Piezo-activating oxygen vacancy regulates quantum well effect and p-band center for exceptional photocatalysis. Nano Energy 2025, 139, 110919.
47. Ning, J.; Kang, Z.; Qin, N.; Chen, M.; Wu, J.; Bao, D. Boosting piezocatalytic performance of perovskite BiFeO3 with controlled oxygen vacancies. J. Alloys Compd. 2024, 978, 173370.
48. Mushtaq, F.; Chen, X.; Hoop, M.; et al. Piezoelectrically enhanced photocatalysis with BiFeO3 nanostructures for efficient water remediation. iScience 2018, 4, 236-46.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].