Interface-assembled batch synthesis of homogeneous 2D-AgPd nanosheets toward electrocatalytic CO2 to CO
Abstract
The electrochemical CO2 reduction reaction (CO2RR) represents a promising strategy for converting greenhouse gases into value-added fuels and chemicals. While silver nanomaterials are recognized for their high CO selectivity, their limited structural tunability and underutilization of active sites restrict catalytic efficiency. Here, we report an interface-confined etching strategy to fabricate two-dimensional AgPd nanosheets with tunable bimetallic composition for enhanced CO2RR performance. By precisely controlling the etching time and alloying degree, we synthesize Ag53Pd47 nanosheets that exhibit outstanding activity and selectivity, achieving a CO Faradaic efficiency of 92.00% and excellent stability at -1.10 V vs. Reversible Hydrogen Electrode (RHE). The synergistic interaction between Ag and Pd atoms modulates *COOH intermediate adsorption and facilitates CO desorption, thus enhancing catalytic kinetics while suppressing competing hydrogen evolution. This study highlights the critical role of composition engineering and interfacial design in constructing high-performance electrocatalysts, providing a generalizable approach for advancing CO2RR technologies.
Keywords
INTRODUCTION
Utilizing renewable energy to electrochemically convert carbon dioxide into useful compounds in aqueous solutions is expected to reduce the level of carbon dioxide and protect renewable energy[1-6]. The catalytic CO2 electro-reduction reaction (CO2RR) usually generates a variety of products ranging from hydrocarbons to oxygen-containing compounds and from C1 to C3 due to the effects of multiple protons and electrons[7-17]. Among the various products resulting from CO2RR (such as HCOOH, CO, CH4, C2H4, C2H5OH and CH3OH), the liquid-phase products of CO2RR typically have lower Faradaic efficiencies (FEs) and require energy-intensive downstream separation[18-25]. CO is a highly valuable alternative due to its large market potential and multifunctional feasibility. Moreover, it can be easily separated and purified using existing chemical technologies and serves as a useful fuel and raw material for chemical synthesis[26-29]. Although CO2RR to CO has an absolute advantage at room temperature and normal pressure, the competitive reduction of protons in KHCO3 aqueous solution requires a high overpotential for the electrochemical reduction of CO2 to CO, while the hydrogen evolution reaction (HER) is kinetically more favorable. Therefore, developing new types of metals with high FE for carbon dioxide emission reduction is extremely important. The key challenge in addressing this issue is to design and develop more efficient electrocatalysts that are more conducive to CO2RR rather than HER catalysis.
Silver- and palladium-based noble metal catalysts are both potential candidates for CO2RR to produce CO[30-35]. Early studies have shown that these two metals exhibit catalytic performance that can be regulated by altering the particle size and morphology[36-39]. However, the main challenges for the CO2 conversion to CO in silver and palladium are different. On the silver surface, the activation of the *COOH intermediate by CO2 is affected by a large energy barrier, while the subsequent desorption of *CO is relatively easy. Palladium has a stronger *COOH adsorption capacity and thus has an advantage in activating CO2, while the desorption of *CO is more difficult[40,41]. Meanwhile, the alloys of Pd-Au and Pd-Ag can be adjusted over a wide range to make the electron/surface modulation easier. The redistribution of electrons in the Pd-Au alloy weakens the Pd-C bond, balances the *COOH and *CO bonds, and achieves high efficiency and high-quality activity for CO[42]. Therefore, by taking advantage of the respective advantages of palladium and silver, it is expected that a bimetallic electrocatalyst for CO2RR can be fabricated[43]. Unfortunately, most of them usually require a large overpotential to achieve a satisfactory current density. Moreover, although these nanocatalysts can provide a large number of electroactive centers, they are all solid and dense nanostructures, which hinder the electron transport, mass diffusion, and accessibility of the electroactive sites. The nano-porous thin sheet metal structures, due to their high surface area and large pore volume, are conducive to mass transportation and exposed active sites, and are widely regarded as being very ideal[44].
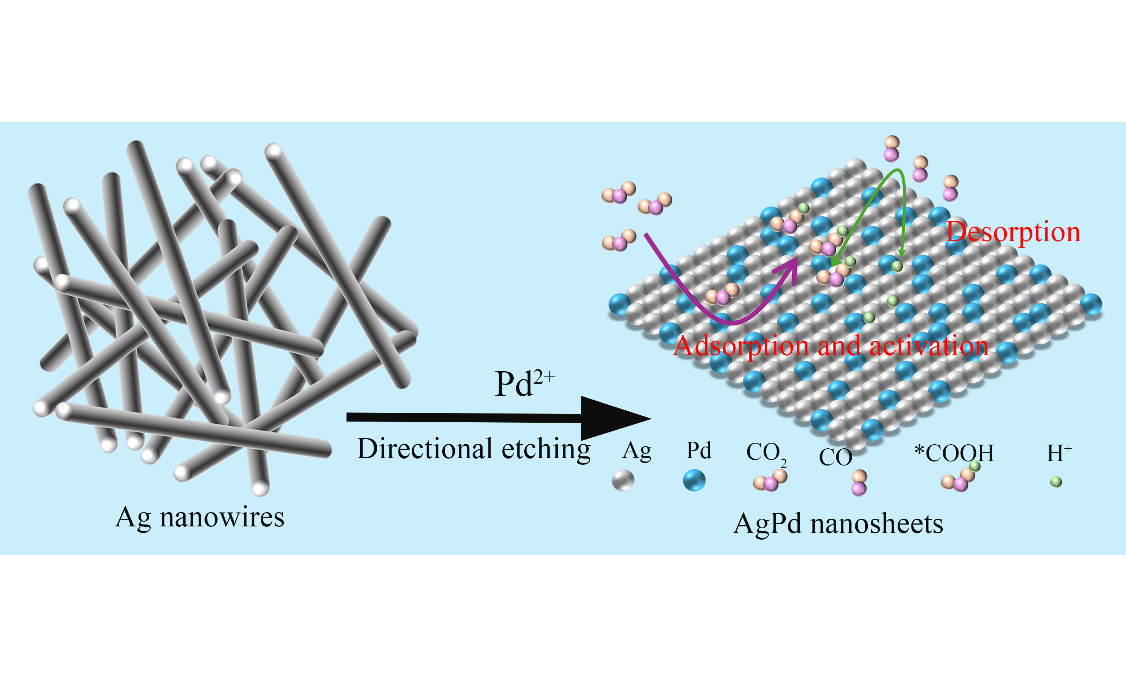
In this work, we employed the interface assembly etching technique and the reaction between Pd(NO3)2 in aqueous solution and the assembled silver nanowire (AgNW) array on the interface to batch-convert the AgNWs into AgPd nanostructures. These nanostructures exhibited similar morphology and uniform structure at room temperature. By controlling the reaction time, we successfully synthesized AgPd nanosheets (AgPdNSs) of different alloying degrees. The AgPdNSs exhibit strong catalytic performance for the CO2 electroreduction reaction, with a FE of CO2 electroreduction to CO of 92.00%. The study shows that compared with pure AgNWs, the AgPd nanomaterials of different alloying degrees have higher catalytic activity, attributed to the synergistic effect of their composition and morphology.
EXPERIMENTAL
Synthesis of silver nanowires
AgNWs are synthesized with a polyol method as reported previously[45]. Typically, 10 mL of ethylene glycol (EG) is refluxed in a three-necked round-bottom flask at 160 °C for 1 h with stirring. Into this, 5 mL of an EG solution containing 0.2 mol L-1 AgNO3 and 5 mL of EG solution containing 0.3 mol L-1 PVP are injected at a rate of ~ 0.2 mL min-1. After 45 min, the solution color turns from pale yellow to opaque gray, indicative of the formation of AgNWs. AgNWs are collected by centrifugation and washed successively with acetone and anhydrous ethanol. The purified Ag nanowires are redispersed in 8 mL of ethanol for nanowire self-assembly.
Liquid-gas interface assembly of silver nanowires
A monolayer array of AgNWs is assembled using a previously reported method with minor modifications[29,46]. Briefly, the AgNWs are assembled into a monolayer at the liquid-gas interface. The specific steps are as follows: 600 μL of the AgNW ethanol solution is added to 3 mL of toluene and mixed evenly. The mixture is then quickly poured into a 10 mL beaker containing 7 mL of water. After a short period (10 min), a pale white film with a metallic luster appears at the water/toluene interface. Most of the toluene is removed with a syringe, and the remaining toluene is allowed to evaporate naturally at room temperature. Once the toluene has completely evaporated, a monolayer array of AgNWs with metallic luster is observed at the air/water interface.
Preparation of two-dimensional silver-palladium nanosheets materials
Two-dimensional (2D) silver-palladium sheet structures can be prepared through interfacial etching at room temperature. Briefly, 15 μL of Pd(NO3)2 (40 mmol L-1) aqueous solution is injected into the aqueous phase using a syringe and gently stirred for 10 seconds with a magnetic stirrer (50.00 rpm/min) at room temperature (without disturbing the AgNW monolayer array at the interface). After the reaction system is left undisturbed for a period of time (2 min), 2D nanosheets are obtained by controlling the etching time (10 h and 24 h) as Pd(NO3)2 reacts with the AgNWs. The interfacial products could be easily transferred onto solid substrates (silicon wafers or glass slides). Before conducting electrochemical experiments, the collected products are washed by centrifugation with ethanol and water to remove unreacted Pd(NO3)2, and then dispersed in 4 mL of Mill-Q water for electrocatalytic reduction of CO2.
RESULTS AND DISCUSSION
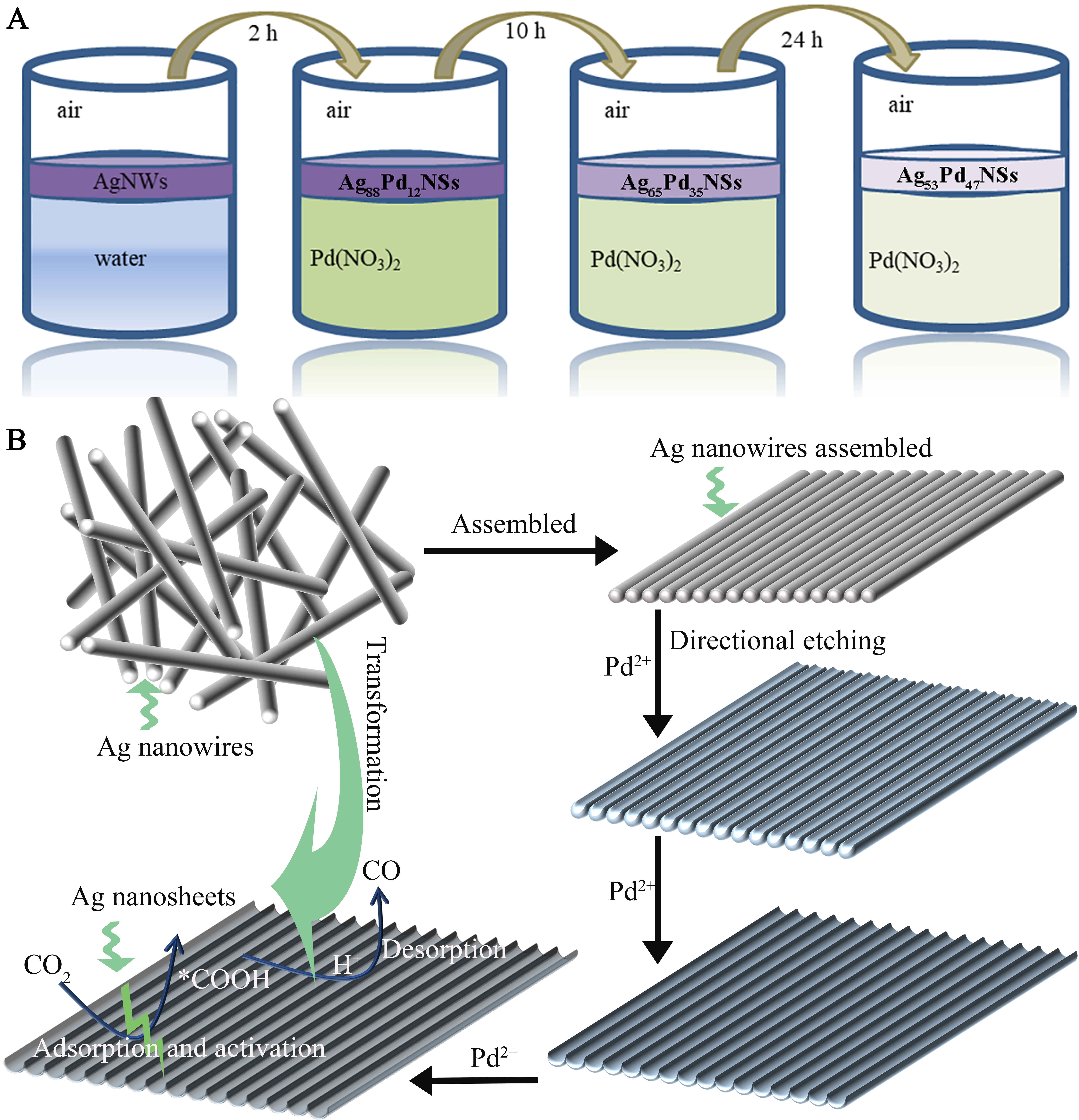
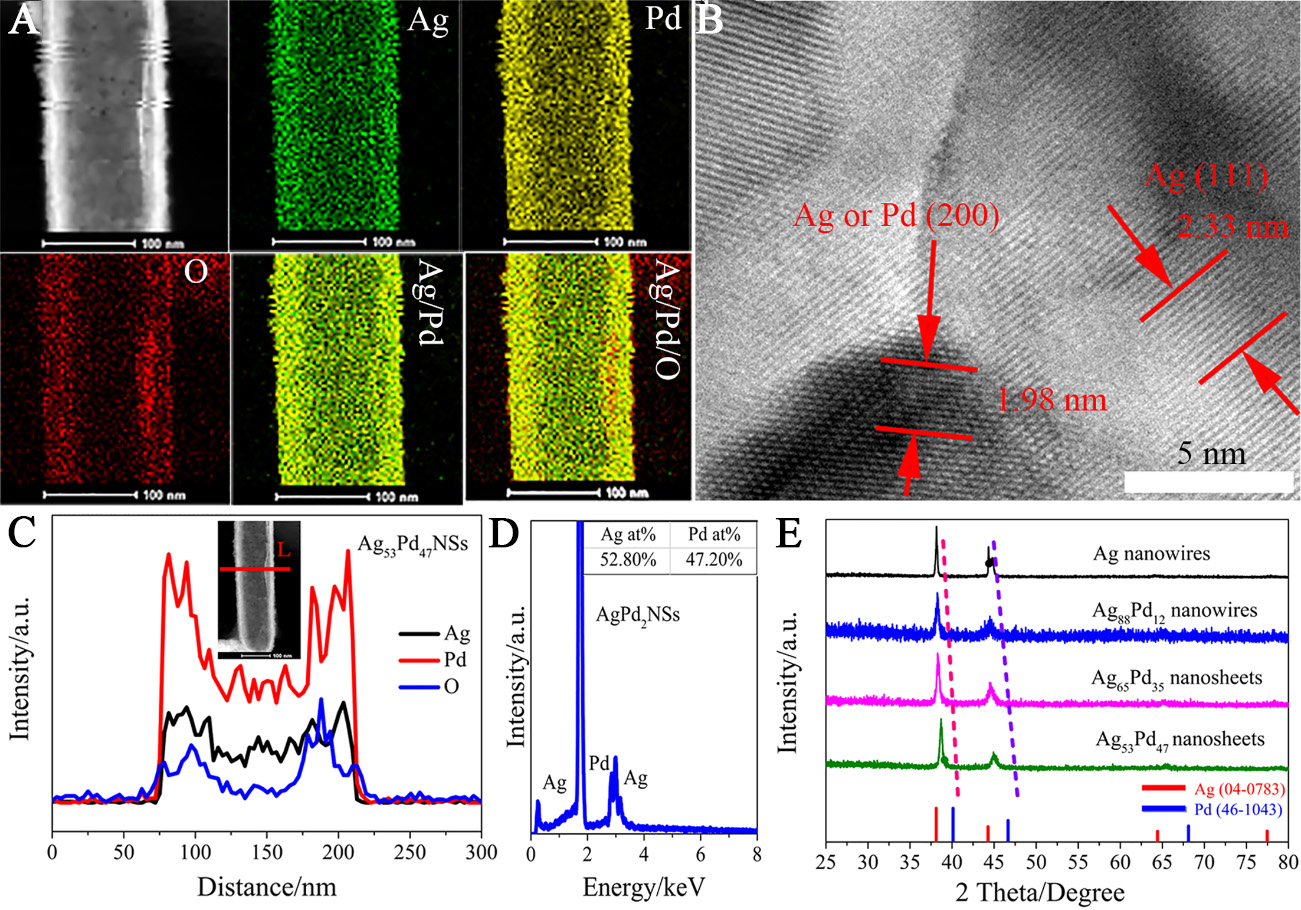
Characterization of the morphological structure of silver-palladium nanomaterials and exploration of their formation mechanism: The reaction between Pd(NO3)2 in aqueous solution and the AgNWs at the interface was carried out through interface assembly, with the reaction time carefully controlled. As a result, AgPd nanomaterials with varying degrees of alloying were synthesized. The reaction scheme is shown in Figure 1A and B. Initially, the AgNWs were assembled into a neat array at the liquid-gas interface, as shown in the corresponding SEM image [Figure 2A]. After 24 h of reaction between Pd(NO3)2 and the interface AgNWs, AgPdNSs were formed. The surface of the AgPd nanomaterials became rough, but the AgPdNSs maintained a neat array and exhibited highly uniform morphology, as seen in the SEM images of the Ag53Pd47NSs [Figure 2B and C]. The corresponding TEM images of the AgNWs and AgPd nanowire arrays are shown in Figure 2D-F, respectively. To further investigate the structural composition of the AgPdNSs, elemental analysis was performed on the AgPdNSs after 24 h. It can be observed that silver and palladium are uniformly distributed on the entire AgPdNS, and some oxygen is attached to the surface [Figure 3A]. In addition, high-magnification transmission electron microscopy (HRTEM) analysis [Figure 3B] clearly reveals the lattice spaces of Ag and Pd, specifically Ag(111), Ag(200) and Pd(200), respectively. Simultaneously, we performed side-line scanning energy-dispersive X-ray spectroscopy (EDS) on the AgPdNSs after 24 h, as shown in Figure 3C. The elemental linear scan clearly shows that Ag, Pd and O are uniformly distributed, with the Pd metal element being more concentrated at the edges of the AgPdNSs compared to the Ag metal element. Elemental content analysis of the AgPdNSs [Figure 3D] revealed an atomic ratio of Ag:Pd of 52.80:47.20, which is close to 1:1, and the material is denoted as Ag53Pd47NSs.
Figure 1. (A) Schematic diagram of the synthesis process for different alloyed AgPd nanostructures and (B) the specific steps for preparing AgPd nanosheets through interface assembly and directional etching of AgNWs. AgNW: Silver nanowire.
Figure 2. (A-C) SEM images of the silver nanowire and Ag53Pd47NSs monolayer array at the air/water interface; (D-F) TEM images of the AgNW and Ag53Pd47NSs. SEM: Scanning electron microscope; AgNW: silver nanowire; NS: nanosheet; TEM: transmission electron microscope.
Figure 3. (A) EDS elemental distribution map of Ag53Pd47NSs; (B) High-magnification transmission electron microscopy (HRTEM) image of the middle part of Ag53Pd47NSs; (C) EDS elemental linear scan profiles of Ag53Pd47NSs along the red line, as marked in the corresponding STEM inset; (D) EDS energy spectrum of the Ag53Pd47NSs; (E) XRD of pure silver and AgPd nanomaterials of different alloying degrees and palladium metal, and the diffraction angles move towards higher directions. EDS: Energy-dispersive X-ray spectroscopy; NS: nanosheet; STEM: scanning transmission electron microscope; XRD: X-ray diffraction.
To further investigate the formation mechanism of AgPdNSs, we collected samples for analysis at 2 h and 10 h, respectively. SEM and transmission electron microscopy (TEM) images of the AgPd nanomaterials revealed that, initially, small slot-like holes appeared on the nanowires at 2 h, indicating a reaction between Pd(NO3)2 in the aqueous solution and the AgNWs at the liquid-gas interface [Supplementary Figure 1]. As the reaction progressed, at 10 h, Ag was partially replaced by Pd2+ ions, resulting in the formation of AgPdNSs with a certain thickness. To further examine the structural composition and morphology of the AgPdNSs after 10 h, we conducted EDS surface scanning elemental analysis [Supplementary Figure 2A], which confirmed the formation of an AgPd alloy, with oxygen also detected. EDS line scan analysis [Supplementary Figure 2B] showed an atomic ratio of Ag:Pd of 65.00:35.00 [Supplementary Figure 2C], and the material was denoted as Ag65Pd35NSs. Silver, palladium, and oxygen were all present in the Ag65Pd35NSs material. Additionally, EDS content analysis of the AgPdNWs at 5 h (denoted as Ag88Pd12NWs, Supplementary Figure 2D) and the Ag65Pd35NSs revealed that, as the reaction time increased, more silver was replaced by palladium. This caused the AgNWs to become less dense, effectively increasing the exposure of active sites, which is beneficial for improving the catalytic activity of the AgNWs.
The X-ray diffraction (XRD) patterns of pure Ag nanowires and Ag/Pd nanomaterials with atomic ratios of 88.00/12.00 (Ag88Pd12NWs), 65.00/35.00 (Ag65Pd35NSs), and 52.80/47.20 (Ag52.8Pd47.2NSs) are shown in Figure 3E. The standard PDF cards are marked at the bottom of the XRD patterns. The results indicate that metallic Ag and Pd exhibit distinct face-centered cubic diffraction peaks (Ag: PDF No. 04-0783; Pd: PDF No. 46-1043), with 2θ values of 38.03°/39.93°, 43.85°/46.15°, and 64.11°/67.25° corresponding to the (111), (200), and (220) planes of the face-centered cubic (fcc) Ag/Pd phase, respectively. The XRD spectrum of the AgPd alloy nanomaterial confirms its fcc crystal structure. As expected, as the Ag/Pd ratio decreases, the diffraction peaks of the AgPd bimetallic nanomaterials shift between pure silver and palladium, with the diffraction angles moving towards higher values.
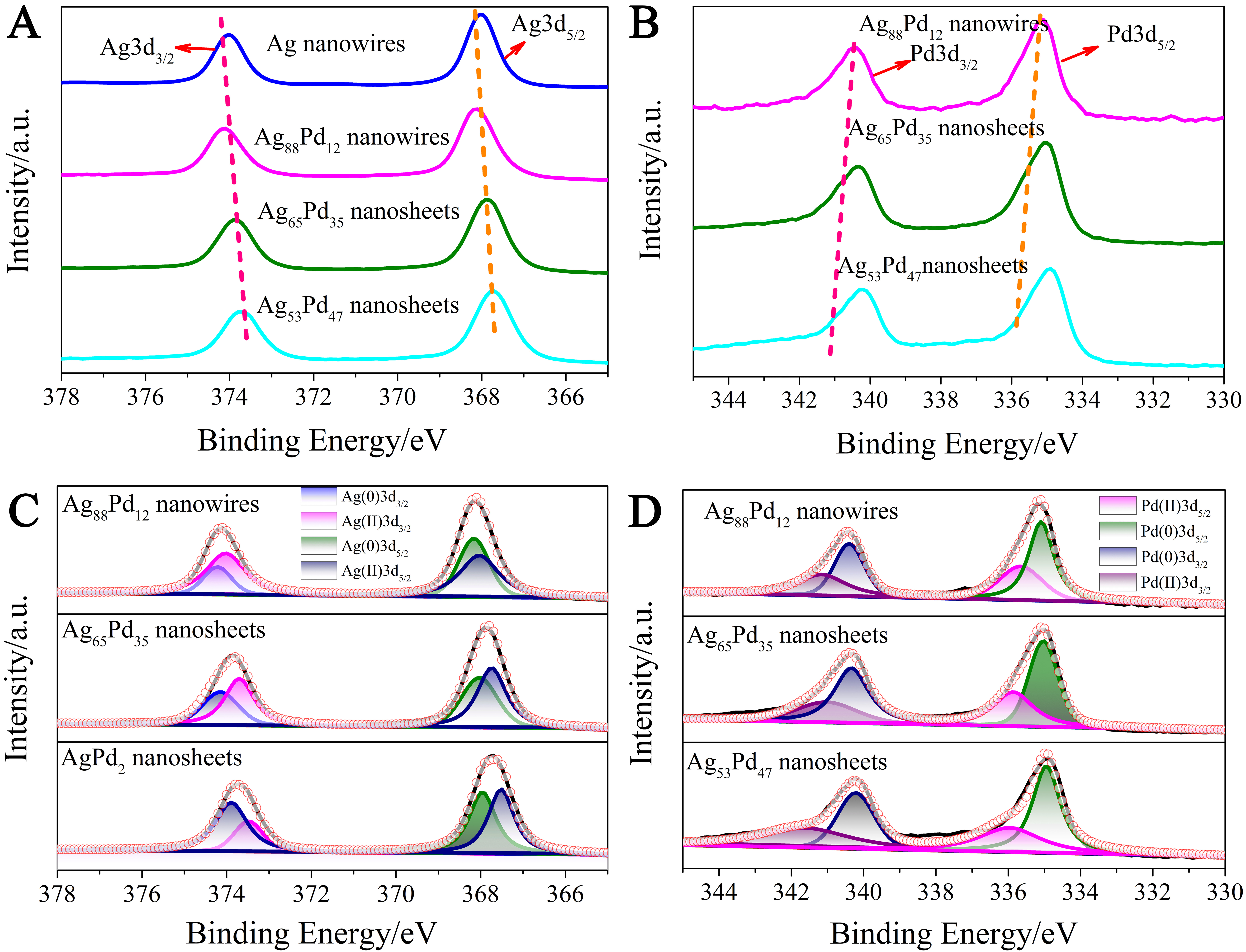
We investigated the chemical states of the nanomaterials Ag88Pd12NWs, Ag65Pd35NSs, and Ag53Pd47NSs, and compared them with pure AgNWs using X-ray photoelectron spectroscopy (XPS). As shown in Figure 4A and B, the binding energies of zero-valent palladium in the AgPd alloy samples are significantly shifted to higher values compared with those of pure palladium at 335.40/340.60 eV. The observed shifts in binding energies for the AgPd alloys in the XPS spectra can be primarily attributed to electronic modifications resulting from alloy formation between Ag and Pd. When Ag and Pd form a homogeneous alloy, charge redistribution occurs due to their different electronegativities and electronic band structures. This leads to a shift in the core-level binding energies: specifically, the Pd 3d peaks shift to higher binding energies, while the Ag 3d peaks shift to lower binding energies relative to their pure metallic states. These shifts indicate a transfer of electron density from Pd to Ag, which modulates the d-band center and optimizes the adsorption energies of key intermediates (e.g., *COOH and *CO) during CO2 electroreduction. This increase was observed with a decrease in the ratio of Ag/Pd in the alloy. Compared with Ag(0) at 374.04 (3d3/2) and 368.04 eV (3d5/2), the AgPd nanomaterials of different alloy degrees shifted towards lower values as the ratio of Ag/Pd in the alloy decreased, indicating that the silver-palladium alloy indeed affected their respective energy bands. This further confirmed the existence of Ag and Pd with an alloy form. We also analyzed the peaks in Figure 4A and B, which show that the alloys contain oxides [Figure 4C and D], influencing the shift in the metal band energies of the AgPd alloy.
Figure 4. XPS spectra of pure AgNWs and AgPd nanomaterials with different alloying degrees for Ag3d3/2 and Ag3d5/2 (A) and Pd3d3/2 and Pd3d5/2 (B); XPS spectra after peak separation of Ag3d (C) and Pd3d (D) in AgPd nanomaterials; The red circle curve represents the fitted curve, which is in agreement with the original curve. AgNW: Silver nanowire; XPS: X-ray photoelectron spectroscopy.
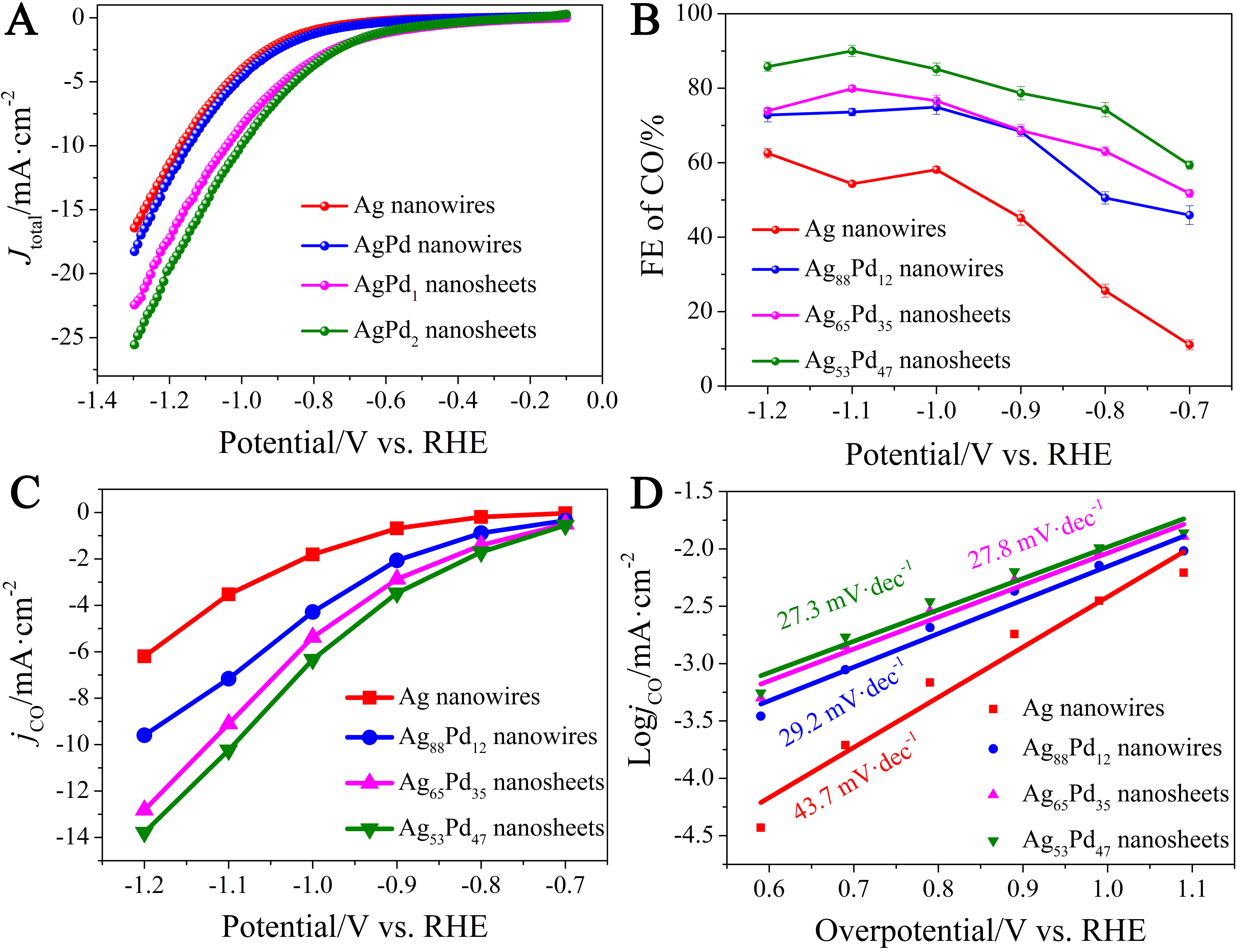
In this work, CO2 electroreduction at different applied voltages was carried out in a CO2-saturated 0.1 mol L-1 KHCO3 solution. Conductive carbon paper is used as the working electrode. Under the saturated state, all the catalysts can reach their respective equilibrium positions and exhibit better catalytic performance. Here, the current density is normalized to the geometric area of the conductive carbon paper (0.09 cm2). Linear sweep voltammetry (LSV) tests were conducted on four materials, and the results show that as the palladium content increases, Ag53Pd47NSs exhibits the highest current density at the same voltage [Figure 5A]. At the initial potential, different degrees of AgPd nanomaterials all show a large catalytic current density (J): J(Ag53Pd47NSs) > J(Ag65Pd35NSs) > J(Ag88Pd12NWs) > J(AgNWs) [Figure 5A]. However, CO2RR is often accompanied by the occurrence of HER. To solve this problem, we used AgNWs, Ag88Pd12NWs, Ag65Pd35NSs and Ag53Pd47NSs as catalysts, and by optimizing the nanomaterials of different alloy degrees, we effectively inhibited the occurrence of HER.
Figure 5. Electrochemical catalytic performance of AgNWs, Ag88Pd12NWs, Ag65Pd35NSs and Ag53Pd47NSs for CO2RR in CO2-saturated 0.10 mol L-1 KHCO3: (A) Cathode LSV curve, (B) CO FEs at different applied potentials (V vs. RHE); (C) CO current density; (D) Tafel curve. The error bars in (B) represent the standard deviation of at least three independent measurements. LSV: linear sweep voltammetry; RHE: reversible hydrogen electrode.
Gas analysis identified two gaseous products, CO and H2, while 1H nuclear magnetic resonance (1H NMR) did not detect any liquid products [Supplementary Figure 3]. Figure 5B shows the CO FE of AgPd nanomaterials with varying alloying degrees and pure Ag nanowires at different applied potentials. The four catalysts exhibit varying catalytic performances in the electrocatalytic CO2 reduction process. Ag53Pd47NSs is the best catalyst among them (AgNWs, Ag88Pd12NWs, Ag65Pd35NSs, and Ag53Pd47NSs), with a CO FE of 92.00%. This indicates that alloying of Ag and Pd can effectively improve the catalytic performance for the selective generation of CO in the electroreduction of CO2. For pure Ag nanowires, the weak adsorption of *COOH is insufficient to activate CO2 molecules. The AgPdNSs exhibit strong catalytic performance for the CO2 electroreduction reaction, achieving a FE of 92.00% for CO production. The results conclude that compared with pure AgNWs, the AgPd nanomaterials of different alloying degrees have higher catalytic activity, attributed to the synergistic effect of their composition and morphology. However, the addition of Pd reduces the availability of the active sites for the electrochemical reduction of CO2 due to the strong adsorption of CO. In contrast, for AgPd alloy materials, the synergistic effect in the alloy reduces the adsorption of CO on the active sites or enhances the adsorption of *COOH on the active sites, thereby narrowing the energy gap between *COOH and *CO and jointly promoting the conversion of CO2 to CO during the electrochemical reduction process. Figure 1 depicts the key steps involved in the CO2-to-CO conversion on the AgPdNSs surface, including (i) Adsorption and activation of CO2 on the alloy surface; (ii) Formation of the ∗COOH intermediate via proton-coupled electron transfer; (iii) Conversion of ∗COOH to ∗CO and desorption of CO as the final product.
As the content of Pd increases, we can observe that the FE of CO can effectively inhibit HER and promote the generation of CO at different voltages. This conclusion has also been confirmed by researchers[34]. We also calculated the partial current density of CO for AgPd nanomaterials with varying alloying degrees at different voltages [Figure 5C] and examined the effect of the CO partial current density (jCO) on the CO yield in the CO2RR electrocatalyst. Ag53Pd47NSs maintained a relatively high partial current density of CO at different voltages. Compared to AgNWs, the Ag53Pd47NSs obtained through our interface assembly etching technique exhibit higher catalytic performance, although they cannot fully suppress HER. The Tafel slopes of AgNWs, Ag88Pd12NWs, Ag65Pd35NSs and Ag53Pd47NSs were approximately 43.70, 29.20, 27.80 and 27.30 mV dec−1, respectively [Figure 5D]. Among them, Ag53Pd47NSs demonstrated the smallest Tafel slope, indicating that it supports the rapid kinetic process of CO2RR. Furthermore, in a 14-hour long-term test at -1.10 V vs. reversible hydrogen electrode (RHE), Ag53Pd47NSs exhibited strong electrocatalytic stability [Supplementary Figure 4].
CONCLUSIONS
In this study, we developed an interface-confined etching strategy to synthesize 2D AgPdNSs with tunable alloying degrees for efficient electrocatalytic CO2 reduction. The optimized Ag53Pd47 nanosheets achieve a CO FE of 92.00% at -1.10 V vs. RHE and exhibit excellent long-term stability, owing to the synergistic modulation of *COOH and *CO adsorption on the alloy surface. This method not only broadens the synthetic toolbox for 2D nanomaterials but also provides a platform for exploring structure-property relationships in electrocatalysis. Moreover, our findings offer valuable insights into the electronic structure modulation of alloy surfaces, which can guide the rational design of next-generation electrocatalysts for efficient CO2 conversion.
DECLARATIONS
Acknowledgements
We acknowledge the resources provided by the High-Performance Computing Center of Central South University.
Author contributions
Conceptualization: Sang, J. L.
Methodology: Sang, J. L.
Data curation: Sang, J. L.
Validation: Sang, J. L.; Liu, Q.
Writing - original draft preparation: Sang, J. L.
Writing - reviewing and editing: Sang, J. L.; Zhang, Y.; Liu, M.
Investigation: Liu, Q.
Funding acquisition: Zhang, Y.; Liu, M.
Resources: Zhang, Y.; Liu, M.
Availability of data and materials
All data required to support the conclusions of this paper are included in the manuscript and the Supplementary Materials. Additional data related to this paper can be obtained from the corresponding authors upon request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (NSFC) (21872048 and 22376222), the Science and Technology Innovation Program of Hunan Province (2023RC1012), the Central South University Research Program of Advanced Interdisciplinary Studies (2023QYJC012), and the Key Research and Development Program of Xinjiang Autonomous Region (2022B02031-1).
Conflicts of interest
Liu, M. serves as an Editorial Board Member of Greenverse Science; however, Liu, M. had no involvement in the editorial process of this manuscript, including reviewer selection, manuscript handling, or decision-making. The other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Song, R. B.; Zhu, W.; Fu, J.; et al. Electrode materials engineering in electrocatalytic CO2 reduction: energy input and conversion efficiency. Adv. Mater. 2020, 32, e1903796.
2. Overa, S.; Ko, B. H.; Zhao, Y.; Jiao, F. Electrochemical approaches for CO2 conversion to chemicals: a journey toward practical applications. Acc. Chem. Res. 2022, 55, 638-48.
3. De Luna, P.; Hahn, C.; Higgins, D.; Jaffer, S. A.; Jaramillo, T. F.; Sargent, E. H. What would it take for renewably powered electrosynthesis to displace petrochemical processes? Science 2019, 364, eaav3506.
4. Wang, W. H.; Himeda, Y.; Muckerman, J. T.; Manbeck, G. F.; Fujita, E. CO2 hydrogenation to formate and methanol as an alternative to photo- and electrochemical CO2 reduction. Chem. Rev. 2015, 115, 12936-73.
5. Resasco, J.; Bell, A. T. Electrocatalytic CO2 reduction to fuels: progress and opportunities. Trends. Chem. 2020, 2, 825-36.
6. Scott, V.; Gilfillan, S.; Markusson, N.; Chalmers, H.; Haszeldine, R. S. Last chance for carbon capture and storage. Nature. Clim. Change. 2012, 3, 105-11.
7. Park, J. W.; Choi, W.; Noh, J.; et al. Bimetallic gold-silver nanostructures drive low overpotentials for electrochemical carbon dioxide reduction. ACS. Appl. Mater. Interfaces. 2022, 14, 6604-14.
8. Fu, H. Q.; Liu, J.; Bedford, N. M.; et al. Synergistic Cr2O3@Ag heterostructure enhanced electrocatalytic CO2 reduction to CO. Adv. Mater. 2022, 34, e2202854.
9. Long, C.; Wan, K.; Qiu, X.; et al. Single site catalyst with enzyme-mimic micro-environment for electroreduction of CO2. Nano. Res. 2021, 15, 1817-23.
10. Wu, J.; Sharifi, T.; Gao, Y.; Zhang, T.; Ajayan, P. M. Emerging carbon-based heterogeneous catalysts for electrochemical reduction of carbon dioxide into value-added chemicals. Adv. Mater. 2019, 31, e1804257.
11. Li, H.; Yue, X.; Qiu, Y.; et al. Selective electroreduction of CO2 to formate over the co-electrodeposited Cu/Sn bimetallic catalyst. Mater. Today. Energy. 2021, 21, 100797.
12. Gao, J.; Bahmanpour, A.; Kröcher, O.; Zakeeruddin, S. M.; Ren, D.; Grätzel, M. Electrochemical synthesis of propylene from carbon dioxide on copper nanocrystals. Nature. Chem. 2023, 15, 705-13.
13. Ji, Y.; Lv, X.; Wei, R.; et al. Unconventional electrocatalytic CO conversion to C2 products on single-atomic Pd-Agn sites. Angew. Chem. Int. Ed. Engl. 2024, 63, e202411194.
14. Shin, H.; Hansen, K. U.; Jiao, F. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain. 2021, 4, 911-9.
15. Lin, R.; Ma, X.; Cheong, W.; et al. PdAg bimetallic electrocatalyst for highly selective reduction of CO2 with low COOH* formation energy and facile CO desorption. Nano. Res. 2019, 12, 2866-71.
16. Sang, J.; Liu, Q.; Zhang, Y.; Liu, M. Liquid–gas interface confined AgPd nanosheets construct corn-shaped AgPd@Cu nanowires for electrochemical CO2 reduction to C2H4. Nano. Research. 2026, 19, 94908282.
17. Sang, J. L.; Zhang, S. D.; Liu, Q.; et al. Liquid-liquid interface -driven reconstruction of CuAg nanocomposites for selective CO2 to C2H4 electroreduction. Small 2025, 21, e08803.
18. Vasileff, A.; Xu, C.; Jiao, Y.; Zheng, Y.; Qiao, S. Surface and interface engineering in copper-based bimetallic materials for selective CO2 electroreduction. Chem 2018, 4, 1809-31.
19. Wang, L.; Nitopi, S.; Wong, A. B.; et al. Electrochemically converting carbon monoxide to liquid fuels by directing selectivity with electrode surface area. Nat. Catal. 2019, 2, 702-8.
20. Jouny, M.; Hutchings, G. S.; Jiao, F. Carbon monoxide electroreduction as an emerging platform for carbon utilization. Nat. Catal. 2019, 2, 1062-70.
21. Ji, Y.; Guan, A.; Zheng, G. Copper-based catalysts for electrochemical carbon monoxide reduction. Cell. Rep. Phys. Sci. 2022, 3, 101072.
22. Zhu, J.; Chen, J. L.; Qi, X. Z.; et al. Electron-Rich Subnanometer Cu clusters facilitate CO-CO coupling in CO2 electroreduction. J. Am. Chem. Soc. 2026, 148, 4008-19.
23. Wang, S.; Zhao, J.; Akdim, O.; et al. Oxygen-bridged dual catalytic sites enable asymmetric C-C coupling for efficient CO2 electroreduction to ethanol. Angew. Chem. Int. Ed. Engl. 2026, 65, e24425.
24. Feng, A.; Fang, N.; Yuan, X. T.; et al. Yb-doped Cu-based catalyst boosting electrochemical CO2-to-C2+ reduction across pH range at ampere-level current density. Angew. Chem. Int. Ed. Engl. 2026, 65, e10755.
25. Zhang, G.; Zheng, X.; Zhao, Z.; et al. Oxygen vacancy bridged Cu-Ce pairs boost CO2 electroreduction to C2H4 via accelerating *H supply and asymmetric C-C coupling. ACS. Energy. Lett. 2025, 11, 625-34.
26. He, Q.; Liu, D.; Lee, J. H.; et al. Electrochemical conversion of CO2 to syngas with controllable CO/H2 ratios over Co and Ni single-atom catalysts. Angew. Chem. Int. Ed. Engl. 2020, 59, 3033-7.
27. Duyar, M. S.; Tsai, C.; Snider, J. L.; et al. A highly active molybdenum phosphide catalyst for methanol synthesis from CO and CO2. Angew. Chem. Int. Ed. Engl. 2018, 57, 15045-50.
28. Bushuyev, O. S.; De Luna, P.; Dinh, C. T.; et al. What should we make with CO2 and how can we make it? Joule 2018, 2, 825-32.
29. Sang, J.; Yu, L.; Song, X.; Geng, W.; Zhang, Y.; Li, Y. Nanoarchitectonics of 2D-thin and porous Ag-Au nanostructures with controllable alloying degrees toward electrocatalytic CO2 reduction. J. Alloys. Compd. 2023, 944, 169155.
30. Cui, M.; Johnson, G.; Zhang, Z.; et al. AgPd nanoparticles for electrocatalytic CO2 reduction: bimetallic composition-dependent ligand and ensemble effects. Nanoscale 2020, 12, 14068-75.
31. Yang, X.; Lee, J. H.; Kattel, S.; Xu, B.; Chen, J. G. Tuning reaction pathways of electrochemical conversion of CO2 by growing Pd shells on Ag nanocubes. Nano. Lett. 2022, 22, 4576-82.
32. Yang, X.; Wu, S.; Zhang, Q.; et al. Surface structure engineering of PdAg alloys with boosted CO2 electrochemical reduction performance. Nanomaterials. (Basel). 2022, 12, 3860.
33. Mahyoub, S. A.; Qaraah, F. A.; Yan, S.; Hezam, A.; Zhong, J.; Cheng, Z. Rational design of low loading Pd-alloyed Ag nanocorals for high current density CO2-to-CO electroreduction at elevated pressure. Mater. Today. Energy. 2022, 24, 100923.
34. Zeng, J.; Zhang, W.; Yang, Y.; Li, D.; Yu, X.; Gao, Q. Pd-Ag alloy electrocatalysts for CO2 reduction: composition tuning to break the scaling relationship. ACS. Appl. Mater. Interfaces. 2019, 11, 33074-81.
35. Zeng, Q.; Tian, S.; Liu, H.; et al. Fine AgPd nanoalloys achieving size and ensemble synergy for high-efficiency CO2 to CO electroreduction. Adv. Funct. Mater. 2023, 33, 2307444.
36. Gao, D.; Zhou, H.; Wang, J.; et al. Size-dependent electrocatalytic reduction of CO2 over Pd nanoparticles. J. Am. Chem. Soc. 2015, 137, 4288-91.
37. Kim, C.; Jeon, H. S.; Eom, T.; et al. Achieving selective and efficient electrocatalytic activity for CO2 reduction using immobilized silver nanoparticles. J. Am. Chem. Soc. 2015, 137, 13844-50.
38. Mahyoub, S. A.; Qaraah, F. A.; Chen, C.; Zhang, F.; Yan, S.; Cheng, Z. An overview on the recent developments of Ag-based electrodes in the electrochemical reduction of CO2 to CO. Sustain. Energy. Fuels. 2020, 4, 50-67.
39. Zhao, Y.; Tan, X.; Yang, W.; et al. Surface reconstruction of ultrathin palladium nanosheets during electrocatalytic CO2 reduction. Angew. Chem. Int. Ed. Engl. 2020, 59, 21493-8.
40. Hansen, H. A.; Varley, J. B.; Peterson, A. A.; Nørskov, J. K. Understanding trends in the electrocatalytic activity of metals and enzymes for CO2 reduction to CO. J. Phys. Chem. Lett. 2013, 4, 388-92.
41. Sheng, W.; Kattel, S.; Yao, S.; et al. Electrochemical reduction of CO2 to synthesis gas with controlled CO/H2 ratios. Energy. Environ. Sci. 2017, 10, 1180-5.
42. Liu, H.; Hernandez, E. S. Structure and stability of nanoscale bimetallic clusters. J. Nanosci. Nanotechnol. 2014, 14, 1533-48.
43. Long, C.; Wang, K.; Shi, Y.; et al. Tuning the electronic structure of PtRu bimetallic nanoparticles for promoting the hydrogen oxidation reaction in alkaline media. Inorg. Chem. Front. 2019, 6, 2900-5.
44. Abdinejad, M.; Ferrag, C.; Hossain, M. N.; Noroozifar, M.; Kerman, K.; Kraatz, H. B. Capture and electroreduction of CO2 using highly efficient bimetallic Pd-Ag aerogels paired with carbon nanotubes. J. Mater. Chem. A. 2021, 9, 12870-7.
45. Sun, Y.; Xia, Y. Large-scale synthesis of uniform silver nanowires through a soft, self-seeding, polyol process. Adv. Mater. 2002, 14, 833.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].