Efficient photothermal conversion in a Ti3C2Tx/Bi2Se3 hybrid for infrared stealth
Abstract
The advancement of infrared detection technologies necessitates the development of novel stealth materials that can actively manipulate thermal signatures. Here, we report a Ti3C2Tx/Bi2Se3 hybrid designed with high-efficiency photothermal conversion for an active infrared thermal stealth strategy. Bi2Se3 was prepared using bismuth on Se nanodisks grown via Cu2+-induced strategy, and Ti3C2Tx MXene and Bi2Se3 were successfully composited through a facile low-temperature ultrasonic process. Owing to the efficient light absorption and charge transfer enabled by the strong interaction between Ti3C2Tx MXene and Bi2Se3, the Ti3C2Tx/Bi2Se3 hybrid material exhibited enhanced photothermal conversion, achieving a remarkable photothermal conversion efficiency of approximately 52.03%, with a standard deviation of 1.67%. In the simulated infrared detection, the Ti3C2Tx/Bi2Se3-based dummy target gradually concealed the real target in the environmental background within 10 min through photothermal conversion. This work demonstrates a promising active stealth strategy and underscores the potential of MXene-based photothermal hybrids in next-generation infrared camouflage technologies.
Keywords
INTRODUCTION
Since every object above absolute zero inevitably radiates infrared energy, this physical law establishes infrared detection as one of the cornerstones of contemporary target-acquisition technology. In recent years, various targets have been confronted with an ever-increasing threat of infrared surveillance with the relentless advance of detection technology. Infrared stealth technology is an essential strategy for shielding objects from infrared detection, which has found widespread application across both civilian and military arenas[1]. According to the Stefan-Boltzmann law, the radiated thermal energy per unit area of an object is proportional to the product of its emissivity and the fourth power of its thermodynamic temperature. Infrared stealth can therefore be achieved by modulating either the emissivity or the temperature. Among these, emissivity engineering, offering flexible and pronounced modulation, is regarded as a highly promising route for infrared stealth technology[2,3]. Guided by this concept, a variety of stealth materials, such as ZnS/Ge2Sb2Te5/HfO2/Ge/HfO2/Ni[4], LMXA-MA[5], and FeCo-HITP/HHTP frameworks[6], have been successively developed to satisfy target protection in different environmental requirements. Nevertheless, the accelerating advance of information technology and the growing complexity of detection scenes make these conventional materials difficult to cope with diverse challenges. Therefore, the development of new systems of infrared stealth materials remains an urgent imperative.
Photothermal materials, which harvest incident photons and convert them into heat with high efficiency, have already found vibrant applications in solar-driven water purification[7], photothermal therapy[8], photothermal sterilization[9], deicing[10], and laser-induced ignition[11], among others. Moreover, photothermally driven infrared emitters have recently attracted intensive attention[12]. Inspired by this study, the present work explores the use of photothermal materials to realize active thermal stealth within infrared scenes. Emerging two-dimensional transition metal carbides/nitrides-MXenes, typified by Ti3C2Tx, exhibit intense localized surface plasmon resonance and photothermal conversion efficiencies approaching unity[13], offering an ideal platform for translating this concept into practice. Notably, numerous studies have demonstrated that MXene-based hybrids frequently outperform pristine MXene in photothermal performance, such as MXene/TiN[14], MXene/holey graphene[15], and MXene/Pt[16]. Bi2Se3, a canonical topological insulator with a narrow bulk band gap of ~0.3 eV, possesses an insulating interior coexisting with metallic surface states. This unique electronic structure endows it with exceptional promise in catalysis, optics, sensing, and energy storage[17], rendering it an outstanding candidate for coupling with MXene to construct high-performance hybrid photothermal systems. Moreover, the Ti3C2Tx/Bi2Se3 hybrid has rarely been explored, underscoring the novelty and scientific value of our work.
In this work, we report the design and construction of a Ti3C2Tx/Bi2Se3 hybrid for infrared thermal stealth applications via photothermal conversion. Benefiting from the strong localized surface plasmon resonance of Ti3C2Tx and the robust interfacial interaction between Ti3C2Tx and Bi2Se3, the Ti3C2Tx /Bi2Se3 hybrid achieved a high photothermal conversion efficiency under 808 nm laser irradiation. During the infrared detection simulation scenario test, the Ti3C2Tx/Bi2Se3-based dummy target could efficiently absorb the energy of the excitation source and convert it into heat and then conceal the real target in its thermal environmental background, showing an effective high-temperature protection strategy. This work demonstrates the great potential of the photothermal materials in the field of new infrared stealth technology.
EXPERIMENTAL
Reagents
Titanium aluminum carbide (Ti3AlC2, 400 mesh) was purchased from 11 Technology Co. Ltd. Sodium hydroxide (NaOH), ascorbic acid (AA), selenium dioxide (SeO2), bismuth acetate, cupric acetate and lithium fluoride (LiF) were purchased from Macklin Biochemical Co., Ltd. Hexadecyltrimethylammonium bromide (CTAB) and Hydrochloric acid (HCl) were purchased from Sinopharm Chemical Reagent Co., Ltd. All chemicals were used as received without further purification. Deionized water with a resistivity of about 18.25 MΩ·cm was used as the solvent in all experiments.
Synthesis of Bi2Se3 nanoflowers
To synthesize Bi2Se3 nanoflowers, Se nanodisks were employed as structural templates. For Se nanodisks,
Synthesis of Ti3C2Tx and Ti3C2Tx/Bi2Se3
Ti3C2Tx MXene was synthesized by a typical etching method. Briefly, 1 g of LiF was incrementally added to a beaker containing 20 mL of HCl. Then, 1 g of Ti3AlC2 was introduced in increments every 5 min under magnetic stirring in an ice-water bath. The mixture was stirred at room temperature for 1 h and transferred to a 35 °C oil bath for 24 h to remove the aluminum layer. Etched MXene was centrifuged at
Photothermal conversion test
To evaluate the photothermal conversion of MXene/Bi2Se3 hybrids, the mixed suspension (1.4 mL,
Characterizations
The morphology and microstructure were characterized using Scanning Electron Microscopy (ZEISS GeminiSEM 300, Germany) and Transmission Electron Microscopy (FEI Talos F200X G2, America). The materials were further analyzed on the X-ray photoelectron spectroscopy (Thermo Scientific K-Alpha, America). The surface elemental properties of the samples were analyzed using X-ray diffraction (Rigaku SmartLab SE, Japan) with Cu Kα radiation. The energy band structure were tested on Ultraviolet Photoelectron Spectroscopy (Thermo Fisher Scientific ESCALAB Xi, America) and Ultraviolet/visible/near-infrared diffuse reflectance (Shimadzu UV-3600i Plus, Japan). The photoelectrochemical measurements were conducted by an electrochemical workstation (CHI 760E, China) with a three-electrode cell. Absorption spectra and temperature curves were recorded on the spectrophotometer (MAPADA P4, China) and thermocouple (KAIPUSEN YET-610, China). Infrared images were captured on a thermal imaging camera (FLIR A615, America). And the laser power was calibrated with a power meter (THORLABS PM100D, America). The error bars in this work represent the standard deviation calculated using the row statistics analysis function built into Origin 2021 software.
RESULTS AND DISCUSSION
Preparation and characterization
The Bi2Se3 nanoflowers were synthesized via a facile hydrothermal method employing preformed Se nanodisks as sacrificial templates, with a detailed process illustrated in Figure 1A. First, the sacrificial templates of Se were synthesized via Cu2+-directed growth under alkaline conditions. The SEM image in Supplementary Figure 1 demonstrates its two-dimensional disk-like nanostructure. Subsequent injection of a Bi precursor into the solution initiated epitaxial development of ultrathin Bi2Se3 nanosheets uniformly across the Se-disk surfaces. Figure 1B and Supplementary Figure 2 show the SEM images of the finally formed Bi2Se3, which possesses a distinct flower-like structure assembled from numerous beautiful ultrathin Bi2Se3 nanosheets. Supplementary Figure 3 presents the electron image and energy-dispersive X-ray spectroscopy (EDS) data acquired on the same area imaged in Figure 1B. The EDS mapping exhibits strong Bi and Se signals, confirming the successful synthesis of Bi2Se3 nanoflowers. Additionally, the absence of a discernible Cu signal in the elemental mapping indicates the inducing role of Cu2+ during synthesis and confirms the reliability of our Cu2+-guided growth strategy of Se nanodisks. The trace oxygen in the mapping may be attributed to slight surface oxidation of the sample during the preparation of SEM specimens. As shown in Supplementary Figure 4, monodisperse Ti3C2Tx MXene nanosheets were prepared by HF etching in solution. The as-prepared Bi2Se3 nanoflowers were subsequently dispersed in the Ti3C2Tx MXene solution, and the two components were successfully combined via low-temperature ultrasonication. As depicted in
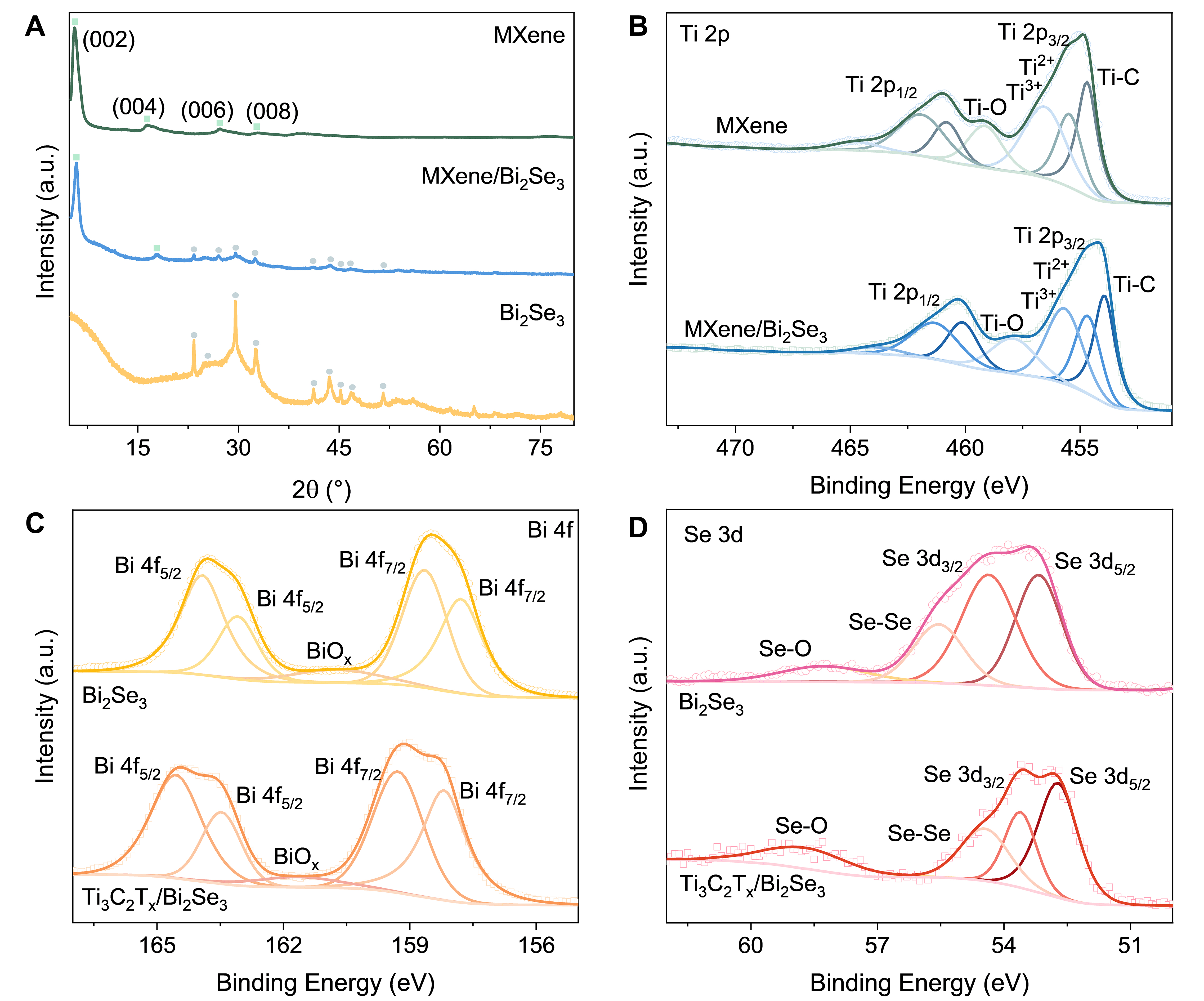
Figure 1. (A) Schematic illustration of the synthetic process of MXene/Bi2Se3. (B) SEM images of Bi2Se3 and (C) MXene/Bi2Se3. (D) TEM image, (E and F) HRTEM Images (The insert scalebar is 1 nm), (G) HAADF-STEM image (The insert scalebar is 20 nm), and (H-L) EDS mappings of MXene/Bi2Se3.
The XRD measurement was employed to characterize the crystalline structure of the Ti3C2Tx/Bi2Se3 hybrid. As shown in Figure 2A, diffraction peaks at 5.62°, 16.3°, 27.3°, and 32.92° are indexed to the (002), (004), (006), and (008) planes of Ti3C2Tx, respectively[20,21]. For the Ti3C2Tx/Bi2Se3 composite, the peaks located at 25.0°, 29.4°, 43.7°, 47.6°, 53.5°, 61.0°, 66.6° and 71.5° can be assigned to (101), (015), (110), (0015), (205), (0210), (1115) and (125) planes of hexagonal Bi2Se3 (PDF#33-0214), respectively, which are consistent with previously reported[22,23]. Notably, phases originating from both MXene and Bi2Se3 are clearly observed in the hybrid, indicating the successful formation of the Ti3C2Tx/Bi2Se3. In addition, XPS analysis was used to study the composition and chemical states of the Ti3C2Tx/Bi2Se3. In Supplementary Figure 7, the XPS survey manifests the presence of Ti, C, F, Bi, and Se elements in the hybrid. Figure 2B displays the high-resolution XPS spectra of Ti 2p in Ti3C2Tx and Ti3C2Tx/Bi2Se3. For Ti3C2Tx, peaks at 454.7 and 460.8 eV are assigned to Ti-C bonds. And peaks around 455.5 and 462.0 eV correspond to Ti2+ 2p3/2 and Ti2+ 2p1/2, while those around 456.6 and 464.5 eV are associated with Ti3+ 2p3/2 and Ti3+ 2p1/2[24-27]. For Ti3C2Tx/Bi2Se3, the Ti-C component shifts to 453.9 and 460.1 eV, the Ti2+ 2p3/2 and Ti2+ 2p1/2 peaks move to 454.7 and 461.4 eV, respectively, and the Ti3+ 2p3/2 and Ti3+ 2p1/2 signals appear at 455.7 and 463.9 eV. As shown in Figure 2C, the Bi 4f high-resolution XPS spectra illustrate that the two peaks at higher binding energy correspond to Bi 4f5/2, while the three at lower energy are assigned to Bi 4f7/2[28,29]. In Bi2Se3, the Bi 4f5/2 peaks are located at 163.1 and 164.0 eV, and the peaks of Bi 4f7/2 stand at 157.8 and 158.6 eV, respectively. In the Ti3C2Tx/Bi2Se3 hybrid, these peaks shift to 164.6, 163.5, 159.3, and 158.1 eV, respectively. These results indicate electron gain by Ti3C2Tx and loss from Bi2Se3, confirming the establishment of a built-in electric field directed from Bi2Se3 to Ti3C2Tx[30-32]. Figure 2D presents the high-resolution Se 3d XPS spectra of Bi2Se3 and Ti3C2Tx/Bi2Se3. The peaks located at 53.2 and 54.4 eV are assigned to Se 3d3/2 and Se 3d5/2 of Se2-[33,34], respectively. And
Enhanced photothermal conversion performance and mechanism study
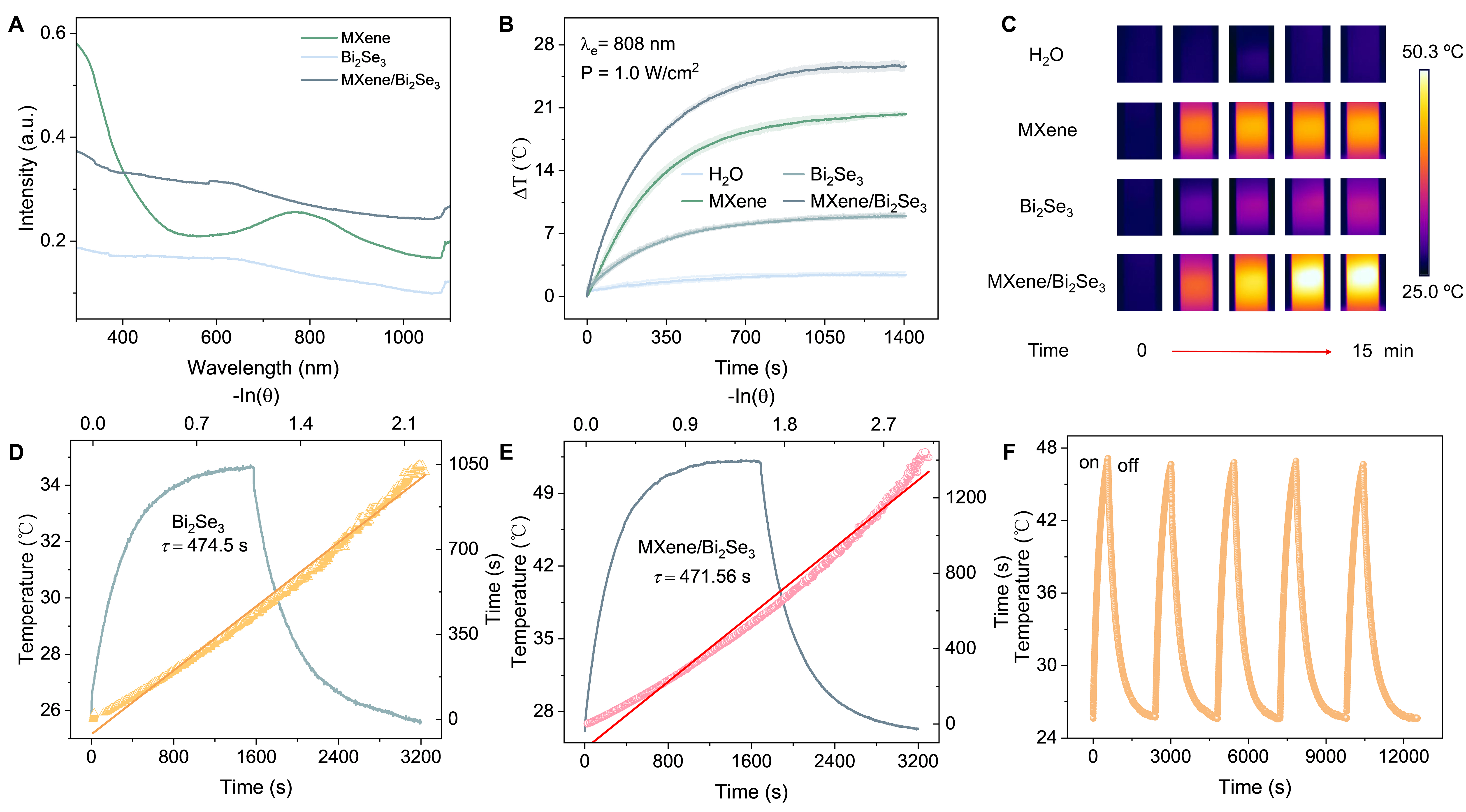
Recently, Ti3C2Tx MXene has gained widespread recognition as an efficient photothermal material. To harness this optic merit and explore the infrared stealth potential of the hybrid, the photothermal performance of Ti3C2Tx/Bi2Se3 was systematically evaluated. Broadband light absorption is a prerequisite for efficient photothermal conversion. Therefore, the UV-VIS-NIR absorbance of Ti3C2Tx, Bi2Se3, and the hybrid is first compared. The result is presented in Figure 3A. Ti3C2Tx MXene exhibits a pronounced plasmonic resonance centered at about 768 nm, attributed to its transversal surface plasmon modes. Meanwhile, it has great broadband light absorption across the UV-to-NIR region. For Bi2Se3, it displays a continuous, wide-range extinction profile from the UV to the NIR owing to its intrinsically narrow bandgap, exhibiting a broad and smooth curve without distinct peaks. Additionally, for Ti3C2Tx/Bi2Se3, the spectrum follows the broad contour of Bi2Se3 while exceeding the absorbance of either standalone component, indicating additive broadband absorption from the intimate union of Ti3C2Tx and Bi2Se3.
Figure 3. (A) Extinction spectra of MXene, Bi2Se3, and MXene/Bi2Se3. (B) Photothermal curves of the solution (25 μg mL-1) under 808 nm (1.0 W cm-2) laser irradiation. (The solid line represents data from a single test, while the shaded area indicate the error bands (SD) derived from three replicate tests. (3) of the corresponding sample by row statistics method). (C) Thermal images of DI water, Bi2Se3, and MXene/Bi2Se3. The heating and cooling curve of Bi2Se3 (D) and MXene/Bi2Se3 (E). (F) Cyclic test of photothermal curves of MXene/Bi2Se3 (1:5).
Based on the absorption characteristics, we further evaluate the photothermal conversion performance of the as-prepared samples. Under irradiation with an 808 nm near-infrared laser, dispersions of Ti3C2Tx MXene, Bi2Se3, and Ti3C2Tx/Bi2Se3 are tested under identical conditions (25 µg mL-1), with deionized water serving as the blank control. The temperature variation of each solution is recorded in real time using a thermocouple coupled with an infrared thermal camera. Figure 3B shows the photothermal curves with error bands of the samples. Benefiting from strong optical absorption and efficient plasmonic heating, the Ti3C2Tx sample increases by about 15.18 °C after 7 min of continuous irradiation. Under identical conditions, deionized water exhibits a negligible increase of about 1.77 °C, whereas the Bi2Se3 suspension warms by about 6.8 °C, demonstrating its relatively weak photothermal property. For Ti3C2Tx/Bi2Se3, we further tune the mass ratio of the two constituents (1:1, 1:5, 1:7) and evaluate the photothermal performance of the different hybrids. As a result, the temperature increments reach about 16.34, 20.02, and 20.49 °C at Ti3C2Tx/Bi2Se3 with a mass ratio of 1:1, 1:5 and 1:7 [Supplementary Figure 9], respectively. Supplementary Figure 10 presents the extinction spectra of the samples, which increase monotonically with Bi2Se3 content. This indicates that the complementary absorption of the two constituents, together with their interfacial coupling, may enhance the overall absorption capability of Ti3C2Tx/Bi2Se3. This may also be one of the reasons for the observed temperature rise. Nevertheless, although the 1:7 ratio exhibits stronger absorption than 1:5, the temperature rises by merely 0.47 °C. In addition, the two are essentially identical for the 10-min infrared thermal stealth application tests because of the overlap around 10 min on the curves. Taking all these factors into account, the hybrid with a mass ratio of Ti3C2Tx and Bi2Se3 of 1:5 is selected for further experimentation. Its temperature rise is 1.32 times that of Ti3C2Tx and 2.94 times that of Bi2Se3, respectively, indicative of the excellent synthetic effect of photothermal conversion. Hereafter, all samples denoted as Ti3C2Tx/Bi2Se3 refer specifically to the hybrid with a 1:5 mass ratio. Figure 3C presents infrared images of DI water, Ti3C2Tx MXene, Bi2Se3, and Ti3C2Tx/Bi2Se3. The color-coded temperature contrast provides intuitive visual evidence of the temperature differences among the samples. These results demonstrate the enhanced photothermal performance of Ti3C2Tx MXene/Bi2Se3. PCE is a key metric for evaluating photothermal conversion capability. To quantify the enhanced performance of Ti3C2Tx/Bi2Se3, we collect the heating and cooling data of bare Bi2Se3 and the Ti3C2Tx/Bi2Se3 hybrid at room temperature [Supplementary Figure 11] and calculate their respective PCE. Figures 3D and E show that the time constants fitted from the curves are 474.5 s for bare Bi2Se3 and 471.56 s for Ti3C2Tx/Bi2Se3. Based on previous works[35-37], the corresponding PCEs are calculated to be 31.88% for Bi2Se3 and 51.71% for Ti3C2Tx/Bi2Se3, with the computational details and corresponding parameters [Supplementary Table 1] provided in the Supplementary Material. Furthermore,
Photothermal conversion performance under 980 nm irradiation is also evaluated.
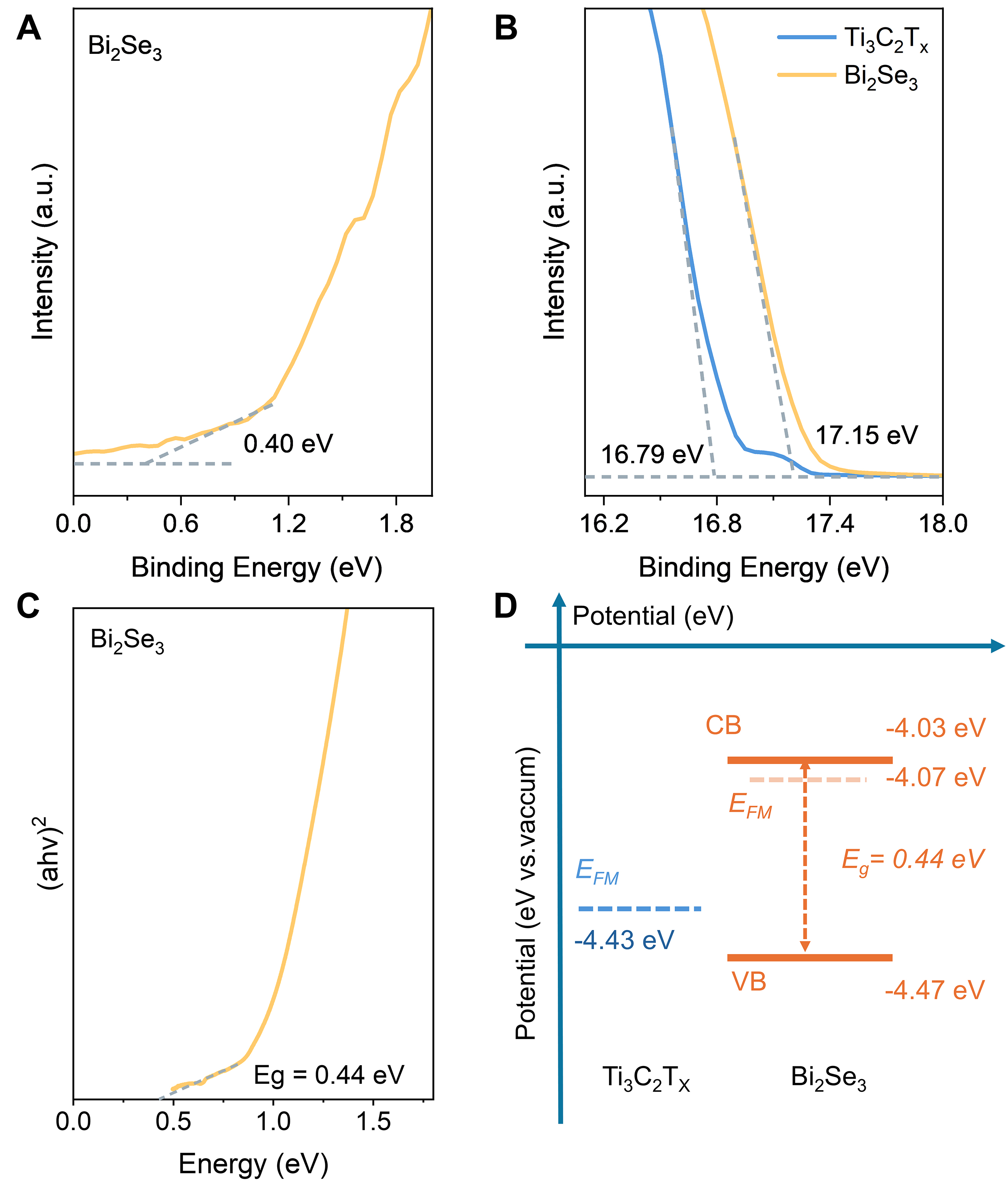
As previously reported, efficient charge separation in hybrids can significantly prolong the lifetime of carriers and thereby enhance photothermal conversion efficiency[42,43]. Guided by this, we suppose that effective charge transfer between Ti3C2Tx and Bi2Se3 constitutes one of the key mechanisms underlying the enhanced photothermal performance of the hybrid. A three-electrode system was employed to experimentally verify the anticipated charge transfer. As shown in Supplementary Figure 18A, Ti3C2Tx/Bi2Se3 exhibits a significantly higher photocurrent density than bare Bi2Se3, evidencing efficient charge transfer and an elevated photogenerated-carrier concentration in the hybrid. In Supplementary Figure 18B, the electrochemical impedance spectroscopy (EIS) of Ti3C2Tx/Bi2Se3 displays the smallest EIS radius, further confirming its superior carrier generation and charge-separation capability. To further illustrate the charge-transfer pathway within the hybrid, the band structures of Ti3C2Tx and Bi2Se3 were investigated by ultraviolet photoelectron spectroscopy (UPS) and diffuse-reflectance spectroscopy (DRS). According to Figure 4A-C, the energy maximum of the valence band with respect to the Fermi level of Bi2Se3 is 0.40 eV. And its band gap, calculated from the Tauc plot, is 0.44 eV. The measured secondary electron cutoff energies are 16.79 eV for Ti3C2Tx and 17.15 eV for Bi2Se3, yielding work functions of 4.43 and 4.07 eV (vs. vacuum), respectively. Thus, the positions of the conduction band and valence band of Bi2Se3 are calculated to be -4.03 and -4.47 eV, with respect to the vacuum level, respectively. Based on the above results, the energy band structures of the Ti3C2Tx and Bi2Se3 are illustrated in Figure 4D. As it shows, the Fermi level of Ti3C2Tx is lower than that of Bi2Se3. Therefore, the electrons will flow from Bi2Se3 into Ti3C2Tx until a common Fermi level is established at equilibrium. Consequently, Bi2Se3 becomes electron-deficient and positively charged, inducing upward band bending, whereas Ti3C2Tx accumulates excess electrons and acquires a negative
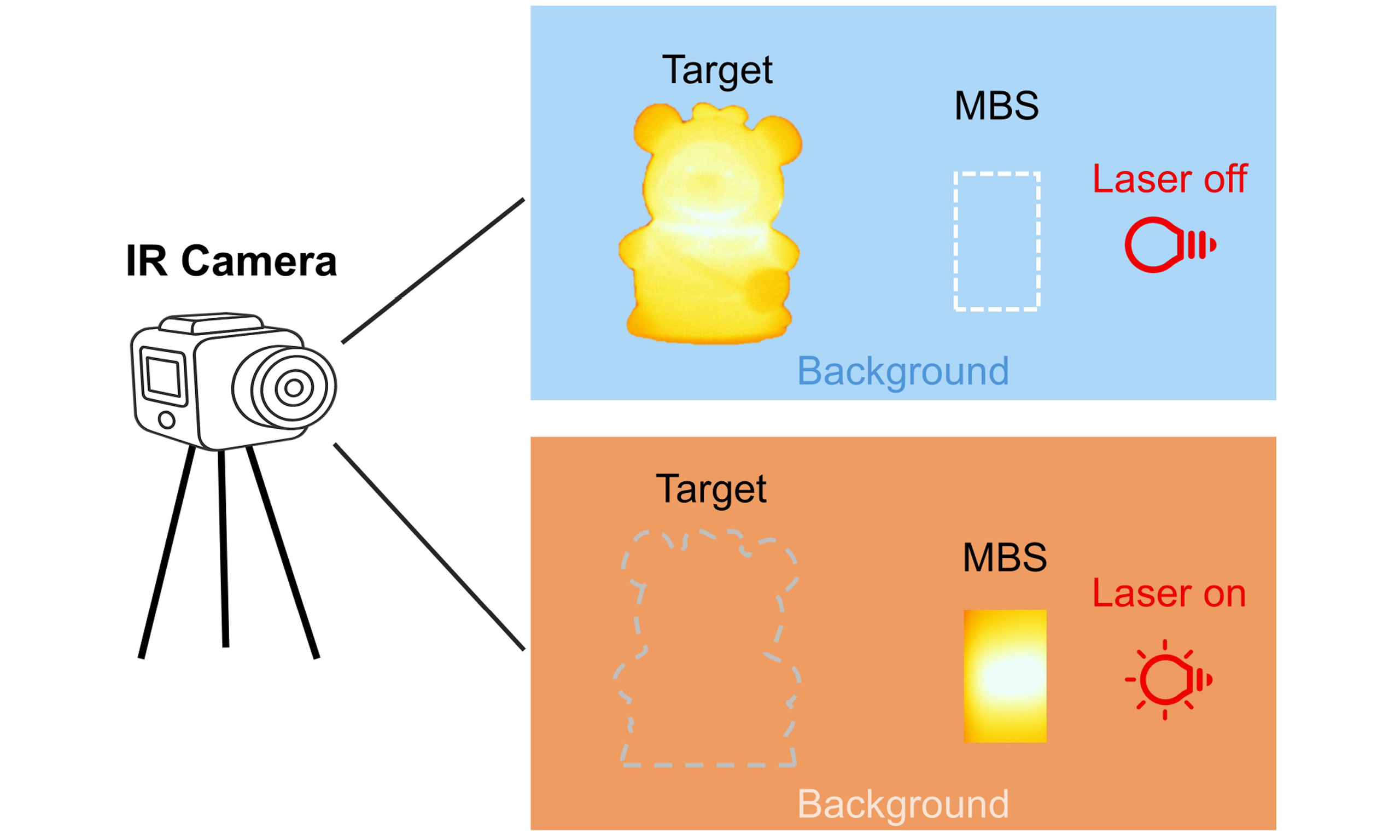
Active infrared thermal stealth property
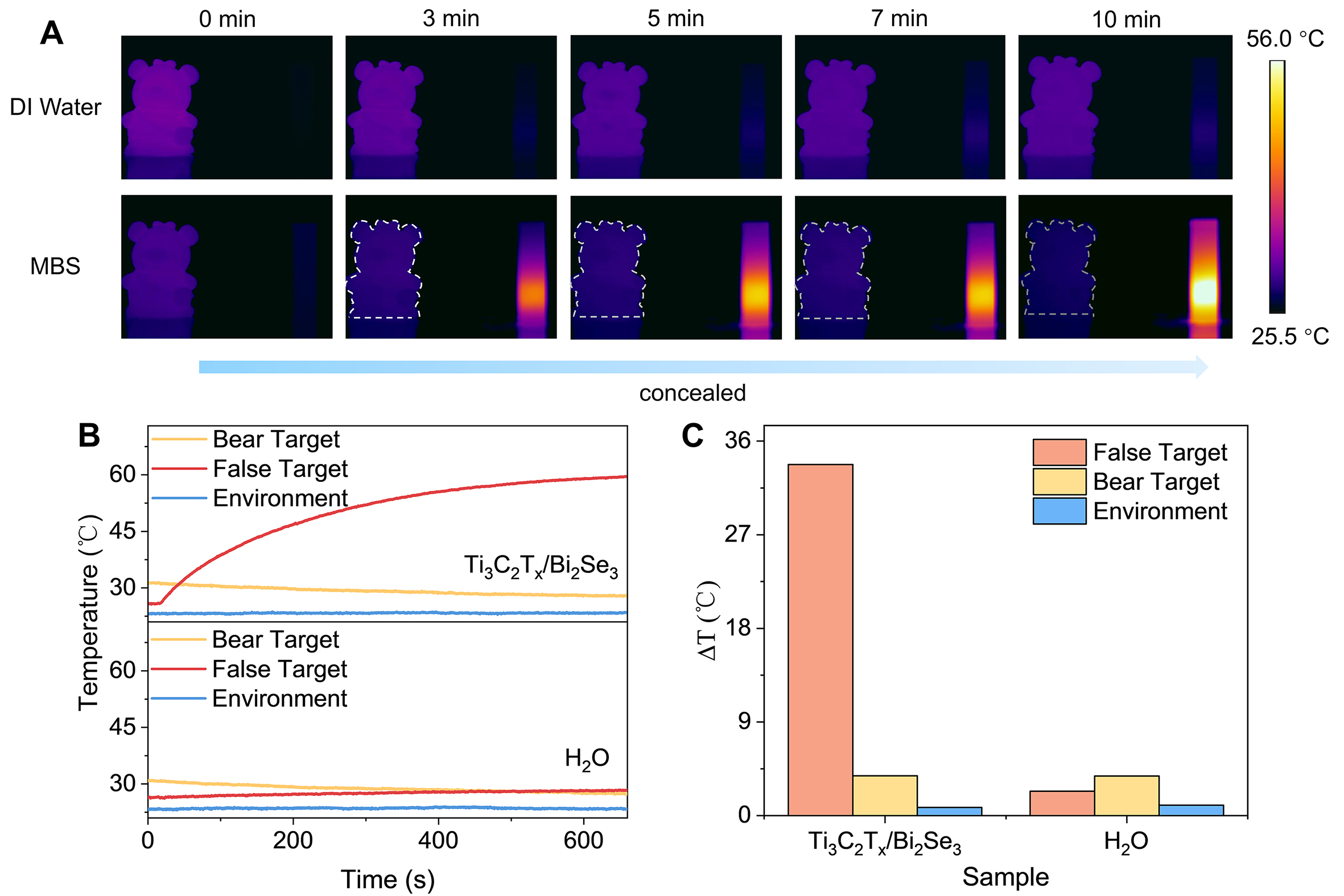
Conventional stealth materials minimize the apparent contrast between an object and its background by suppressing the thermal emissivity of the object. Guided by this principle, we consider whether the same contrast can instead be eliminated by actively raising the emissivity of the surrounding environment, allowing the target to conceal in the thermal background. Motivated by this concept, the exceptional photothermal conversion efficiency and robust stability demonstrated by the Ti3C2Tx/Bi2Se3 hybrid offer an ideal platform for exploring such an inverse infrared stealth strategy. We constructed a simplified infrared-detection scenario [Supplementary Figure 19] to demonstrate the proposed thermal-stealth concept. In the scene, a bear model around the temperature of 30 °C serves as the genuine target on the left, while a decoy object fabricated from the Ti3C2Tx/Bi2Se3 hybrid is positioned on the right under identical surrounding conditions. Drawing on the preceding photothermal results, the Ti3C2Tx/Bi2Se3 solution is selected as the test sample, and the 808 nm laser is employed as the excitation source. Besides, a water control is included to highlight the infrared thermal stealth capability enabled by the photothermal conversion of Ti3C2Tx/Bi2Se3. As illustrated in Figure 5A, in the initial state, the bear target can be clearly identified in both the H2O and Ti3C2Tx/Bi2Se3 systems due to the inherent temperature of the bear target is higher than that of the background. However, the weak photothermal conversion capability of H2O results in negligible temperature changes within the entire testing time. Therefore, the bear target remains distinctly visible in the detection area even after 10 min of irradiation. In contrast, based on the excellent photothermal response of Ti3C2Tx/Bi2Se3, it efficiently absorbs the incident light energy and converts it into thermal during irradiation. As the irradiation time increases, the temperature gradually rises and eventually becomes the dominant heat source in the detection environment. Under this circumstance, the temperature of the bear target is gradually lower than that of the heat source environment formed by Ti3C2Tx/Bi2Se3. As observed in Figure 5A, the bear target gradually becomes concealed in the background with the increase in time. The corresponding video is provided in the Supplementary Movie. Supplementary Figure 20 compares the surface temperatures of the different objects in the scene when H2O or Ti3C2Tx/Bi2Se3 is employed as the decoy at the indicated measurement times. As shown, the maximum temperature difference among background, real target, and decoy is only 9.5 °C, and the real target remains the warmest spot, making it readily detectable by infrared detectors when H2O serves as the decoy. In contrast, the Ti3C2Tx/Bi2Se3 decoy enlarges the temperature span to 35.6 °C and becomes the hottest object in the scene, furnishing a conspicuously elevated thermal background that may mask the genuine target. Figures 5B and C further present the time-dependent temperature curves of the three objects and the corresponding temperature difference recorded during the 10-min test. The temperature-difference comparison in the figure also underscores the strong photothermal-conversion capability of Ti3C2Tx/Bi2Se3 and its potential for achieving infrared thermal stealth through photothermal heating. This experiment strongly validates the great potential of photothermal materials for infrared thermal stealth via photothermal conversion. It can be noted that this work offers only a proof-of-concept demonstration of infrared thermal stealth enabled by photothermal materials, as prepared
CONCLUSIONS
In summary, a Ti3C2Tx/Bi2Se3 hybrid was successfully fabricated and demonstrated to enable infrared thermal stealth through efficient photothermal conversion. The synergy of the plasmon resonance of Ti3C2Tx and the robust interaction with Bi2Se3 nanoflowers brings a strong electric field, efficient charge transfer, and broadband absorption, resulting in superior photothermal performance with a PCE of approximately 52.03%. A simulated IR-detection scenario demonstrated that the hybrid allows targets to effectively fade into its thermal background under laser irradiation, demonstrating its great promise for infrared thermal stealth applications. This work provides a promising strategy for developing advanced photothermal materials in stealth technologies.
DECLARATIONS
Authors’ contributions
Conceptualization: Zou, J. W.; Zhang, Y. S.; Chen, Y.; Hu, Y. H.
Data curation, visualization, resources: Zou, J. W.; Sun, W. B.; Xia, Y. H.
Formal analysis: Zou, J. W.; Zhang, Y. S.; Chen, Y.; Huang, H.;
Funding acquisition, project administration: Zhang, Y. S.; Hu, Y. H.
Supervision: Zhang, Y. S.; Chen, Y.; Huang, H.; Luo, H. Q.; Ye, Q.; Hu, Y. H.
Writing - original draft: Zou, J. W.; Chen, Y.
Writing - review and editing: Zou, J. W.; Chen, Y.; Huang, H.; Luo, H. Q.; Ye, Q.; Zhang, Y. S.; Hu, Y. H.
Availability of data and materials
The data that support the findings of this work can be obtained upon request from the first author.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was financially supported by the National Natural Science Foundation of China (61871389, 62304256), the College of Electronic Engineering (KY22C213, KY24A204), the State Key Laboratory of Pulsed Power Laser Technology (KY21C604), and Jianghuai Advanced Technology Center.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Deng, C.; Dong, H.; Sun, K.; et al. Synchronous visual/infrared stealth using an intrinsically flexible self-healing phase change film. Adv. Funct. Mater. 2023, 33, 2212259.
2. Li, M.; Cheng, Y.; Fang, C.; et al. Superior dynamic infrared stealth of VO2 achieved by multielement doping. Chem. Eng. J. 2025, 524, 169243.
3. Lyu, J.; Liu, Z.; Wu, X.; Li, G.; Fang, D.; Zhang, X. Nanofibrous kevlar aerogel films and their phase-change composites for highly efficient infrared stealth. ACS. Nano. 2019, 13, 2236-45.
4. Qin, B.; Zhu, H.; Zhu, R.; Zhao, M.; Qiu, M.; Li, Q. Space-to-ground infrared camouflage with radiative heat dissipation. Light. Sci. Appl. 2025, 14, 137.
5. Zhu, X.; Liu, J.; Zhang, L.; Zhao, W.; Cao, Y.; Liu, X. Dual-mode MXene-based phase-change composite towards enhanced photothermal utilization and excellent infrared stealth. Small 2024, 20, e2405694.
6. Jin, Y.; Cheng, J.; Jiang, S.; et al. Conductance reinforced relaxation attenuation with strong metal-N coordination in multivariate π-conjugated MOFs for integrated radar-infrared camouflage. Adv. Mater. 2025, 37, e2501330.
7. Meng, X.; Wang, X.; Yin, K.; et al. Integration of photothermal water evaporation with photocatalytic microplastics upcycling via nanofluidic thermal management. Proc. Natl. Acad. Sci. USA. 2024, 121, e2317192121.
8. Feng, H.; Wang, W.; Wang, T.; Pu, Y.; Ma, C.; Chen, S. Interfacial regulation of BiOI@Bi2S3/MXene heterostructures for enhanced photothermal and photodynamic therapy in antibacterial applications. Acta. Biomater. 2023, 171, 506-18.
9. Yang, Q.; Lou, S.; Zhang, Y.; et al. NIR-II responsive multifunctional scaffold enabling "Kill-Modulation-Build" synergistic therapy for infectious bone defects. Adv. Sci. 2025, 12, e08948.
10. Zhou, P.; Wang, Y.; Zhang, X. Bi2Se3 nanosheets-based photothermal composites with hydrophobic surface for synergistic anti-/de-icing. Compos. Sci. Technol. 2023, 233, 109916.
11. Li, Y.; Hou, X.; Yao, L.; Yang, Y.; Shen, R.; Wu, L. Single-layer/few-layer and multilayer Ti3C2Tx MXene: divergent mechanisms in laser ignition performance of CL-20 via interface activity and structural confinement. Small 2025, 21, e06361.
12. Hao, Y.; Yang, S.; Ling, C.; et al. Ultralarge pixel array photothermal film based on 3D self-suspended microbridge structure for infrared scene projection. Small 2023, 19, e2208262.
13. Chang, P.; Mei, H.; Zhao, Y.; et al. Nature-inspired 3D spiral grass structured graphene quantum Dots/MXene nanohybrids with exceptional photothermal-driven pseudo-capacitance improvement. Adv. Sci. 2022, 9, e2204086.
14. Zou, J. W.; Hu, Y. H.; Sun, W. B.; et al. Manipulating optical properties of MXene/TiN with strong nonmetallic plasmonic coupling for multifunctional imaging attenuation. InfoMat 2025, 7, e70024.
15. Cai, Z.; Ma, Y.; Yun, M.; et al. Multifunctional MXene/holey graphene films for electromagnetic interference shielding, Joule heating, and photothermal conversion. Compos. Part. B. Eng. 2023, 251, 110477.
16. He, X.; Lv, Y.; Lin, Y.; et al. Platinum nanoparticles regulated V2C MXene nanoplatforms with NIR-II enhanced nanozyme effect for photothermal and chemodynamic anti-infective therapy. Adv. Mater. 2024, 36, e2400366.
17. Zong, Y.; Chen, H.; Wang, J.; et al. Cation defect-engineered boost fast kinetics of two-dimensional topological Bi2Se3 cathode for high-performance aqueous Zn-ion batteries. Adv. Mater. 2023, 35, e2306269.
18. Xie, M.; Li, C.; Zhang, S.; et al. Topological insulator Bi2Se3-assisted heterostructure for ultrafast charging sodium-ion batteries. Small. 2023, 19, e2301436.
19. Lin, Z.; Zhang, W.; Peng, J.; et al. Synergistic regulation of polyselenide dissolution and Na‐ion diffusion of Se‐vacancy‐rich bismuth selenide toward ultrafast and durable sodium‐ion batteries. Adv. Energy. Mater. 2024, 14, 2402110.
20. Jiang, D.; Cao, X.; Shi, Y.; et al. Flexible Ti3C2Tx MXene regulated photoelectrochemical sensing platform for sensitive monitoring of dopamine. Adv. Funct. Mater. 2024, 34, 2410546.
21. Liu, M.; Wang, J.; Song, P.; Ji, J.; Wang, Q. Metal-organic frameworks-derived In2O3 microtubes/Ti3C2Tx MXene composites for NH3 detection at room temperature. Sens. Actuators. B. Chem. 2022, 361, 131755.
22. Niu, X.; Liu, Y.; Li, X.; Wang, W.; Yuan, Z. NIR light-driven Bi2Se3-based nanoreactor with “three in one” hemin-assisted cascade catalysis for synergetic cancer therapy. Adv. Funct. Mater. 2020, 30, 2006883.
23. Chen, K.; Chong, S.; Yuan, L.; Yang, Y.; Tuan, H. Conversion-alloying dual mechanism anode: nitrogen-doped carbon-coated Bi2Se3 wrapped with graphene for superior potassium-ion storage. Energy. Storage. Mater. 2021, 39, 239-49.
24. Zhu, L.; Wu, W.; Chen, J.; Hu, Z.; Yu, J.; Wang, Y. High-performance electromagnetic wave absorption by two-dimensional mesoporous monolayer Ti3C2Tx MXene. Chem. Eng. J. 2024, 488, 150649.
25. Huang, Z.; Liang, Y.; Wu, Z.; et al. Multifunctional ultrathin Ti3C2Tx MXene@CuCo2O4/PE separator for ultra-high-energy-density and large-capacity lithium-sulfur pouch cells. Adv. Mater. 2025, 37, e2410318.
26. Liu, Y. T.; Zhang, P.; Sun, N.; et al. Self-assembly of transition metal oxide nanostructures on MXene nanosheets for fast and stable lithium storage. Adv. Mater. 2018, 30, e1707334.
27. Peng, W.; Han, J.; Lu, Y. R.; et al. A general strategy for engineering single-metal sites on 3D porous N, P Co-doped Ti3C2Tx MXENE. ACS. Nano. 2022, 16, 4116-25.
28. Lu, Q.; Li, P.; Guo, Z.; et al. Giant tunable spin Hall angle in sputtered Bi2Se3 controlled by an electric field. Nat. Commun. 2022, 13, 1650.
29. Lu, C.; Luo, M.; Dong, W.; et al. Bi2Te3/Bi2Se3/Bi2S3 cascade heterostructure for fast-response and high-photoresponsivity photodetector and high-efficiency water splitting with a small bias voltage. Adv. Sci. 2023, 10, e2205460.
30. Ran, J.; Zhang, H.; Fu, S.; et al. NiPS3 ultrathin nanosheets as versatile platform advancing highly active photocatalytic H2 production. Nat. Commun. 2022, 13, 4600.
31. Deng, X.; Zhang, J.; Qi, K.; Liang, G.; Xu, F.; Yu, J. Ultrafast electron transfer at the In2O3/Nb2O5 S-scheme interface for CO2 photoreduction. Nat. Commun. 2024, 15, 4807.
32. Liu, Y.; Pan, S.; Xia, W.; et al. Strong interaction between plasmon and topological surface state in Bi2Se3/Cu2-xS nanowires for solar-driven photothermal applications. Sci. Adv. 2025, 11, eadt2884.
33. Li, L.; Lu, Y.; Xu, X.; et al. Catalytic-enhanced lactoferrin-functionalized Au-B2Se3 nanodots for Parkinson's disease therapy via reactive oxygen attenuation and mitochondrial protection. Adv. Healthc. Mater. 2021, 10, e2100316.
34. Zang, Y.; Zhang, W.; Wang, P.; et al. Bi2Se3/PAAS hydrogels with photothermal and antioxidant properties for bacterial infection wound therapy by improving vascular function and regulating glycolipid metabolism. Adv. Healthc. Mater. 2025, 14, e2401810.
35. Li, J.; Wang, J.; Zhang, J.; et al. A facile strategy of boosting photothermal conversion efficiency through state transformation for cancer therapy. Adv. Mater. 2021, 33, e2105999.
36. Hou, M.; Jiang, Z.; Sun, W.; Chen, Z.; Chu, F.; Lai, N. C. Efficient photothermal anti-/deicing enabled by 3D Cu2-xS encapsulated phase change materials mixed superhydrophobic coatings. Adv. Mater. 2024, 36, e2310312.
37. Xi, D.; Xiao, M.; Cao, J.; et al. NIR light-driving barrier-free group rotation in nanoparticles with an 88.3% photothermal conversion efficiency for photothermal therapy. Adv. Mater. 2020, 32, e1907855.
38. Pan, S.; Xia, W.; Feng, W.; Ma, L.; Ding, S.; Chen, X. Enhanced photothermal conversion in ReS2/Bi2Se3 nanoflowers for solar-driven thermoelectric power generation and water evaporation. J. Alloys. Compd. 2025, 1020, 179390.
39. Xu, L.; Li, Y.; Li, Z.; et al. Synergizing piezoelectric field and photothermal effect modulated charge migration behavior in Bi4O5Br2/Bi2S3 S-scheme heterojunction for enhanced near-infrared-light-driven photocatalytic activity. Chem. Eng. J. 2025, 519, 165096.
40. Liu, X.; Sun, R.; Zhou, Z.; Tang, Y. In situ growth of nano-hydroxyapatite on multilayered Ti3C2Tx MXene as a drug carrier with superior-performance. J. Mater. Sci. Technol. 2024, 180, 91-101.
41. Jin, C.; Sun, D.; Sun, Z.; et al. Interfacial engineering of Ni-phytate and Ti3C2Tx MXene-sensitized TiO2 toward enhanced sterilization efficacy under 808 nm NIR light irradiation. Appl. Catal. B. Environ. 2023, 330, 122613.
42. Yuan, H.; Qin, H.; Sun, K.; et al. Ultrafast hot electron transfer and trap-state mediated charge separation for boosted photothermal-assisted photocatalytic H2 evolution. Chem. Eng. J. 2024, 494, 153058.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].