Customized polymer electrolytes for high-energy-density lithium batteries
Abstract
Solid-state batteries with lithium-rich manganese layered oxide (LRMO) cathodes, anode-free architectures, and polymer electrolytes offer high energy density and enhanced safety. However, unstable cathode morphology and irreversible redox reactions at the electrolyte-cathode interface lead to severe interfacial degradation and poor cycling stability. Recently, a fluoropolyether-based polymer electrolyte has been developed, which is a copolymer synthesized via in situ polymerization of poly(ethylene glycol) methyl ether acrylate and fluorohydrocarbon monomers. Its anion-rich solvation environment drives the in situ formation of fluorine-rich interphases at both electrodes and significantly improves the redox reversibility of LRMO. This quasi-solid polymer electrolyte, containing 30 wt% trimethyl phosphate, enables the LRMO cathode to achieve energy densities of 604 Wh kg-1 and 1,027 Wh L-1 in pouch batteries. Despite this progress, practical deployment still requires the development of low-fluorine electrolytes, uniform in situ polymerization in large-format batteries, improved mechanical robustness, and long-term stability with lithium metal and high-voltage LRMO cathodes.
Keywords
DISCUSSION
With the widespread use of rechargeable batteries in daily life, research efforts have increasingly focused on solid-state batteries (SSBs) using non-flammable solid electrolytes (SEs) to enhance safety and energy density[1-4]. Conventional polymer electrolytes suffer from low oxidative stability, low Li+ transference numbers, and poor interfacial stability at high voltages, which adversely affect long-term cycling performance. The concept of solvation chemistry, including ion solvation structures and bulk/interphase formation, plays a crucial role in controlling ionic transport and interphase formation and offers new design options beyond conventional ionic conductivity optimization.
Traditional cathode materials, such as LiCoO2 and LiNi1-x-yMnxCoyO2 (NCM), exhibit intrinsic limitations in energy density[5,6]. In contrast, lithium-rich manganese layered oxide (LRMO) cathodes have attracted significant attention due to their unique ability to utilize both anionic (oxygen) and cationic (transition metal, TM) redox reactions. However, their practical implementation in SSBs remains challenging: anionic redox is often difficult to activate; moreover, the redox processes are partially irreversible, leading to low cathode capacity and poor cycling stability[7].
LiTMO2 (TM = Ni, Co, Mn, etc.) and Li2MnO3 are the structural origins of cationic and anionic redox in LRMO. Excess lithium in TM layers forms Li-O-Li and Li-O-□ (□ represents a vacancy) configurations, which activate high-energy oxygen states [8]. In liquid electrolytes (LEs), rapid Li+ transport is enabled by high ionic mobility; however, LRMO|SE interfaces suffer from poor contact and the intrinsically low conductivity of Li2MnO3, leading to slow kinetics. An unstable oxygen redox configuration can induce O=O dimerization, oxygen release, and irreversible interfacial phase formation with SEs, thereby increasing interfacial impedance[9]. Excess oxygen activity also triggers a layered-to-spinel phase transition in LRMO, which is a major cause of degradation in SSBs. Therefore, stabilizing lattice oxygen and enhancing Li+ transport are crucial for efficient anion redox.
The reversibility of anionic redox at high voltages depends on bulk electronic activation and electrolyte-electrode interfacial properties (electronically blocking, ionically conductive, chemically inert). Over the past few years, interfacial challenges in LRMO-based SSBs have been a research focus. For example, Li3BO3 coating suppresses reactive oxygen species and electron transfer, but this approach has not been demonstrated to be effective at high voltages (> 4.5 V)[10]. Li2WO4 incorporation via a one-step synthesis reduces the Li+ migration barrier[11], but its high brittleness causes mechanical cracking. Similarly, Li2B4O7 (LBO) doping suppresses oxygen release[9], but it easily leads to coating cracking.
These methods rely on fixed surface layers, with inherent limitations in dynamic systems, such as soft-pack and anode-free configurations. More recent studies on polymer electrolytes have focused on integrating bulk solvation/coordination microenvironment engineering with interfacial phase engineering. For instance, a puzzle-like molecular assembly strategy for in situ polymerized solid polymer electrolytes (SPEs) enhances salt dissociation and Li+ transport, while forming robust inorganic-rich interphases[12]. The solvation structure of SPEs can be modulated through polymer backbone chemistry and weak coordination environments to improve ionic conductivity, Li+ transference numbers, and electrode compatibility, thereby establishing a new paradigm beyond traditional additive/coating strategies.
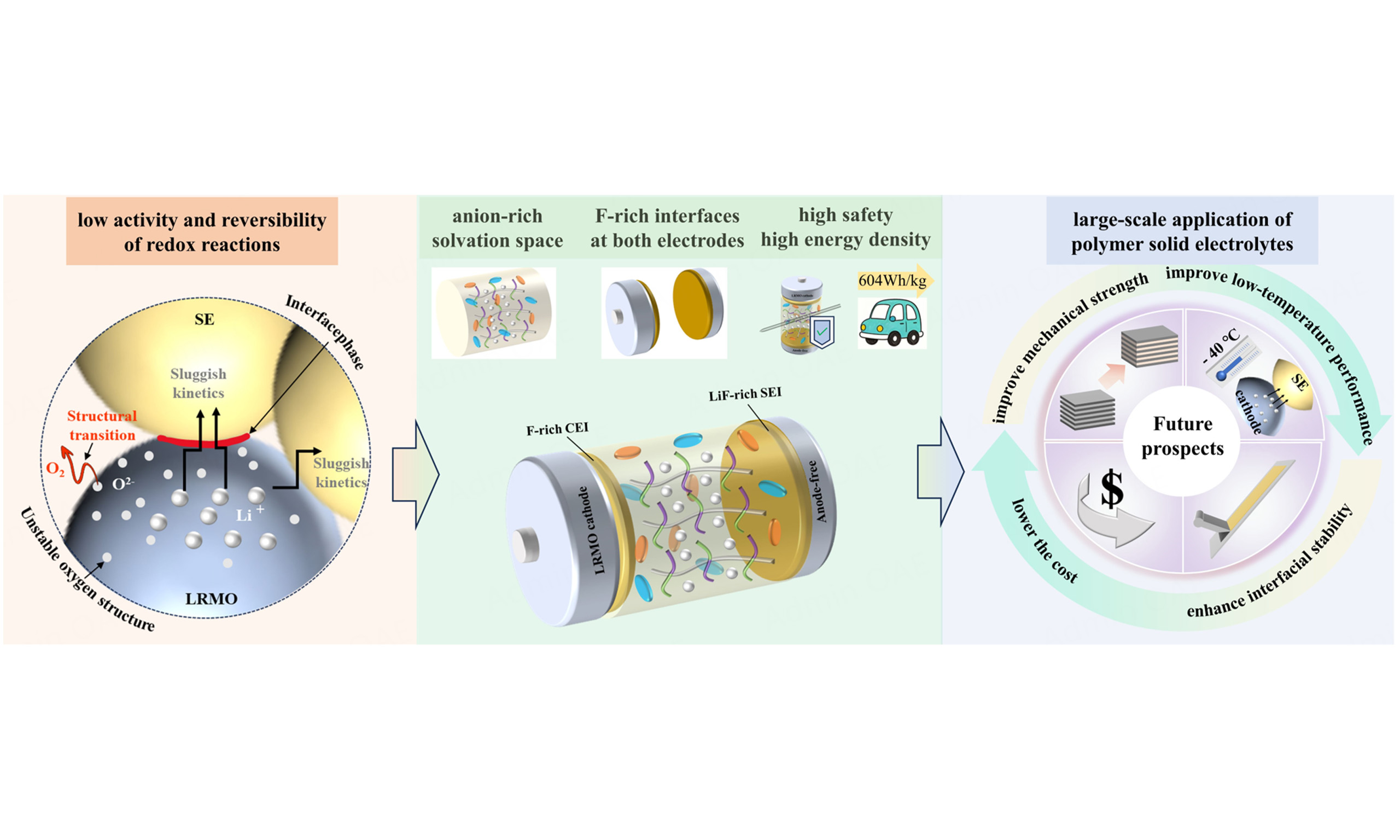
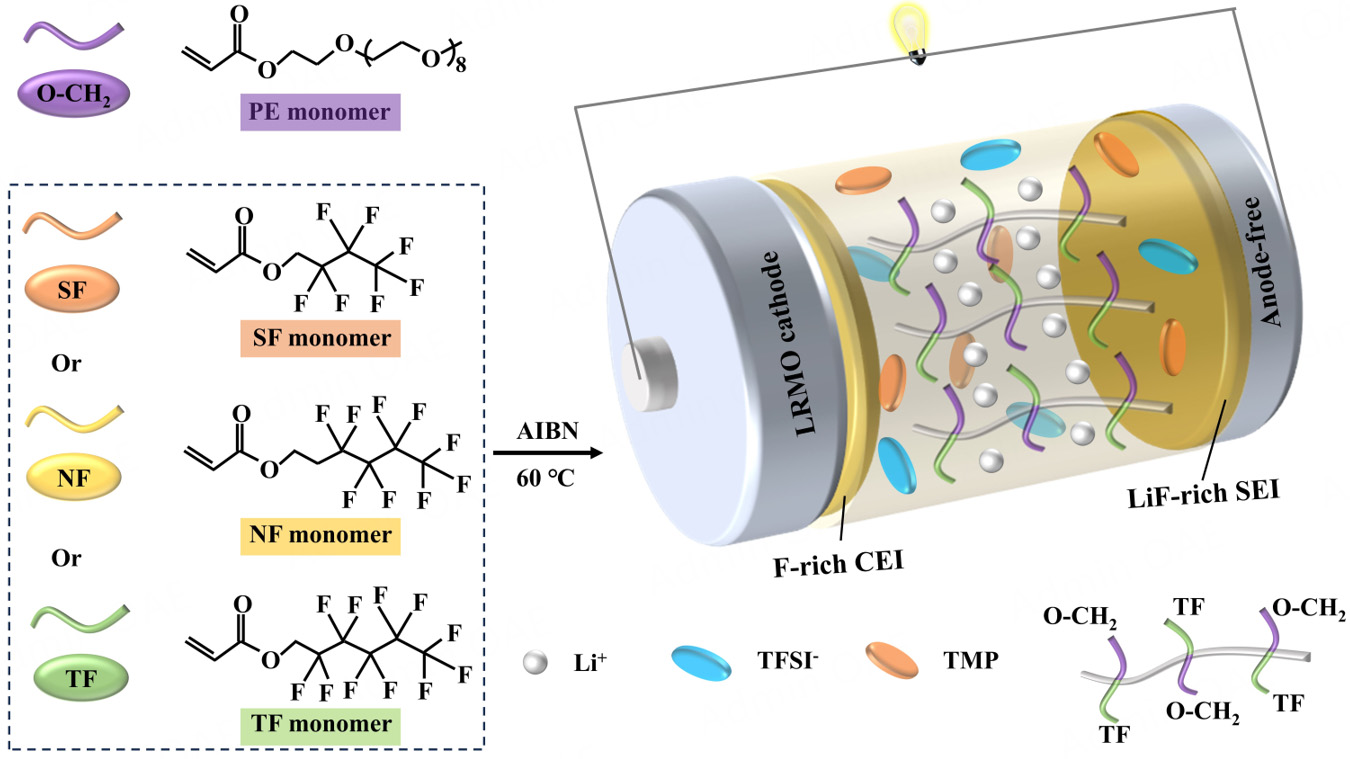
Unlike these strategies, Huang et al. proposed an anion-solvated design for a fluoropolyether-based polymer electrolyte (PTF-PE-SPE)[13], enhancing LRMO-SPE interfacial stability and reaction kinetics via bulk solvation optimization and in situ formation of an inorganic-rich interphase. This system achieves a high areal capacity (> 8 mAh cm-2) and an ultrahigh energy density (604 Wh kg-1 ), overcoming the trade-off between interfacial stability and energy density. Through in situ polymerization of miscible, strongly solvating polyether monomers and weakly solvating fluorinated alkyl monomers, PTF-PE-SPE forms an anion-rich solvation structure [Figure 1]. This structure facilitates Li+ dissociation from polymer chains and promotes anion participation in contact ion pairs/aggregates, leading to the formation of an F-rich cathode electrolyte interphase (CEI; LiF outer layer, Mn-F inner layer) on LRMO, as well as a solid electrolyte interphase (SEI) on the anode.
Figure 1. Schematic illustration of in situ polymerization of fluoropolyether-based polymer electrolytes catalyzed by 2,2′-azobis(2-methylpropionitrile) (AIBN).
Li|PTF-PE-SPE|LRMO batteries retained 72.1% of their capacity after 500 cycles at 0.5 C, with an average Coulombic efficiency of 99.5%. Cu|PTF-PE-SPE|LRMO pouch batteries achieved an initial discharge capacity of 8.96 Ah, along with energy densities of 604 Wh kg-1 and 1,027 Wh L-1 at 0.05 C. Fully charged pouch batteries showed no fire or thermal runaway (at 120 °C for 6 h) during puncture and thermal tests, demonstrating superior safety performance compared with quasi-solid and gel polymer electrolytes.
In comparison with previous fluorine-containing polymer electrolytes, PTF-PE-SPE has three distinct innovations: (1) Solvation structure design: balancing the strong solvating ability of polyether segments with the weak solvating nature of fluorohydrocarbon pendants to construct an anion-rich solvation environment (not merely incorporating fluorine for oxidation stability); (2) Interfacial regulation mechanism: an in situ-formed CEI with a LiF outer layer and Mn-F inner layer (where F- replaces O2- in the LRMO lattice via anion exchange), thereby stabilizing lattice oxygen; (3) Comprehensive performance breakthroughs: this system is the first to simultaneously achieve ultrahigh energy density (604 Wh kg-1), high areal capacity
The anion-rich solvation strategy provides three key contributions. First, it establishes a new electrolyte design paradigm based on solvation structure regulation of interfacial chemistry, going beyond conventional surface modification strategies such as surface coating. Second, the in situ-formed homogeneous F-rich CEI/SEI with Mn-F bonds and LiF overlayers offers a universal approach for stabilizing high-voltage cathodes and Li metal anodes, which can be extrapolated to other systems. Third, the anode-free pouch batteries with an energy density of 604 Wh kg-1 establish a new benchmark for practical SSBs, demonstrating that anode-free architectures are feasible in the high-energy-density regime.
Although this seminal Nature work has achieved significant advances, several non-negligible limitations remain: (1) Cost and scalability: The high loading of perfluorinated monomers (e.g., TF) and the trimethylolpropane (TMP) plasticizer (30 wt%) increases costs and supply chain risks. In situ polymerization requires oxygen-free conditions and precise temperature control, which are difficult to scale up; (2) Mechanical properties: Although PTF-PE-SPE exhibits an elongation at break of 860% in laboratory tests, its mechanical robustness under rigorous cycling and high stack pressures in large-format batteries has not been fully validated; (3) Interfacial stability: The stability of Mn-F bonds and the LiF-rich interphase above 120 °C or at 1.0 C needs further exploration. Interfacial compatibility remains a major bottleneck for SPEs, as side reactions and volume changes can cause severe degradation; (4) Low-temperature ionic transport: PTF-PE-SPE has a glass transition temperature (Tg) of -75 °C, but its ionic conductivity and Li+ transference number at -20 °C have not been reported, limiting its applicability at low temperatures.
To mitigate these limitations and enhance practical applicability, the following efforts are needed: (1) Cost-effectiveness and process optimization for scalable SPE production: Partial substitution of perfluorinated monomers with weakly polar monomers (e.g., partially fluorinated acrylates or fluorine-free polymer ether-based moieties) can preserve anion-rich solvation. A dual-curing system (e.g., ultraviolet-initiated prepolymerization followed by thermal polymerization) improves process control, shortens curing time, and saves energy; (2) Enhancement of mechanical strength in SPEs: Nanoscale reinforcement or semi-interpenetrating polymer networks can improve mechanical strength and prevent interfacial delamination. Silane coupling agents (e.g., KH560) alleviate agglomeration and enhance filler-polymer compatibility; (3) Improvement of interfacial stability: Coating current collectors with ultrathin functional films (e.g., fluorinated carbon coatings or controlled oxide layers) can promote uniform formation of a LiF-rich SEI; (4) Improvement of low-temperature performance: Incorporating copolymer units (e.g., ethylene oxide-propylene oxide or other low-Tg soft blocks) can reduce crystallinity, thereby facilitating rapid Li+ migration at low temperatures.
Future research directions can be categorized into four aspects: (1) Multifunctional electrolyte design: Combining anion-rich solvation with single-ion conduction or self-healing properties to enhance ionic transport efficiency and mechanical stability; (2) Synergy between cathode and electrolyte engineering: Surface engineering of LRMO cathodes with fluorine-containing functional groups may promote stable Mn-F bond formation. Manipulation of polymer solvation shells to regulate competitive coordination and anion behavior is central to stable high-voltage cathode interfaces with high areal capacity and long cycle life; (3) Scalable production technologies: Developing continuous in situ polymerization processes and roll-to-roll coating with controlled thermal treatment for large-format batteries; (4) Low-temperature optimization: Tailoring monomer structures to maintain anion-rich solvation and ionic conductivity at -20 to -40 °C, extending the operating range of high-energy-density SSBs. These strategies build on Huang et al.’s solvation-interphase framework and provide guidance for the scalable development of polymer electrolyte-based SSBs with high energy density, enhanced safety, and broad environmental tolerance[13].
According to the design principle of anion-rich solvation-interphase regulation, the future development of all-solid-state batteries lies in collaborative innovation in multicomponent electrolyte systems and high-capacity electrodes to break the performance bottlenecks of single systems. For electrolytes, in addition to optimizing low-fluorination modification, as well as the mechanical strength and low-temperature performance of polymer electrolytes, the research and development of polymer-inorganic ceramic and sulfide composite electrolytes will integrate the advantages of ionic transport and interfacial compatibility from each system. For electrodes, the stabilization of lattice oxygen and the regulation of anionic redox in LRMO cathodes should be further enhanced; meanwhile, interfacial adaptation strategies for high-capacity anodes (e.g., lithium metal and silicon-based anodes) need to be developed. Furthermore, challenges related to large-scale preparation and full-interface dynamic regulation in all-solid-state batteries must be addressed, and the cross-system solvation-interphase structure-activity relationships should be clarified, ultimately enabling the industrialization of all-solid-state batteries with high energy density, enhanced safety, and wide temperature tolerance for applications such as power batteries and energy storage systems.
It should be noted that the central design approach of anion-dominant solvation engineering and integrated electrode-electrolyte interface control proposed in this work is not limited to PTF-PE-SPE and LRMO cathodes, but rather represents a generalizable and scalable design philosophy for high-energy-density solid-state lithium batteries. This paradigm can be extended to a wide range of cathode materials, including high-nickel layered oxides (NCM/NCA), spinel oxides (LiNi0.5Mn1.5O4), sulfur cathodes, and even organic cathodes, for which rational solvation modulation can effectively suppress cation dissolution, polysulfide shuttling, and active species dissolution, respectively. For polymer electrolyte families, the solvation/coordination optimization strategy can be applied to polycarbonate-, polyester-, and polyimide-based electrolytes, as well as polymer-inorganic composite electrolytes (e.g., polymer-ceramic/polymer-sulfide). By optimizing the solvation microenvironment, interfacial compatibility with high-voltage and high-capacity electrodes can be greatly enhanced, along with ionic transport kinetics. In essence, the interphase-solvation synergy represents a key strategy for addressing universal interfacial instability and performance degradation in polymer electrolyte-based solid-state lithium batteries. The lessons learned and future outlooks can be generalized to other high-performance solid-state battery systems, helping to overcome material-specific drawbacks.
In summary, the recent work[13] employs a PTF-PE-SPE to autonomously form fluorine-rich, conformal interphases at the electrode interfaces, suppressing oxygen-anion side reactions, enhancing interfacial kinetics, and achieving pouch battery-level gains in energy density and safety (604 Wh kg-1, 1,027 Wh L-1). This study outlines a practical route toward LRMO-based SSBs. These key design concepts—anion-dominant solvation engineering and integrated interface regulation-are broadly applicable to a wide range of high-energy cathode materials and polymer electrolyte systems for solid-state lithium batteries. For large-scale deployment, further progress is required in the development of low-cost, fluorine-free polymer electrolytes, uniform in situ polymerization in large-format batteries, improved mechanical robustness of polymer electrolytes, and long-term interfacial stability with lithium metal and high-voltage LRMO cathodes. Coordinated advances in these areas, grounded in solvation/coordination engineering and integrated interface design, are poised to enable anode-free LRMO batteries approaching ~600 Wh kg-1 with enhanced safety. This represents a credible near-term target for polymer-electrolyte SSBs and a significant step toward their commercial viability.
DECLARATIONS
Authors’ contributions
Literature survey, writing - original draft and graphical illustration: Fang, S.
Organization/curation: Fang, S.; Qiao, J.; Liu, L.; Chen, Y.
Analysis: Fang, S.; Qiao, J.; Liu, L.; Chen, Y.; Eliseeva, S.; Holze, R.
Funding acquisition, writing - review & editing: Yuan, X.; Fu, L.; Wu, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Project on Carbon Emission Peak and Neutrality of Jiangsu Province (BE2022031-4), the National Natural Science Foundation of China (22409027, 52131306, 52403001), the National Key R&D Program (2022YFB2404600), and the Big Data Computing Center of Southeast University.
Conflicts of interest
Prof. Wu, Y. is the Editor-in-Chief of Energy Materials, and Prof. Chen, Y. is an Associate Editor of Energy Materials. Prof. Wu, Y. and Prof. Chen, Y. were not involved in any part of the editorial process, including reviewer selection, manuscript handling, or decision making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Wang, T.; Chen, B.; Liu, Y.; et al. Fatigue of Li metal anode in solid-state batteries. Science 2025, 388, 311-6.
2. Du, J.; Sun, Z.; Peng, B.; et al. Another way to realize LiMn2O4 as a solid electrolyte. Adv. Funct. Mater. 2024, 35, 2421179.
3. Wang, Z.; Yang, H.; Li, S.; et al. Small components play a big role - fillers in composite solid-state electrolytes for lithium metal batteries. Energy. Mater. 2024, 4, 400080.
4. Zhang, H.; Ni, Z.; Wang, Z.; et al. Manipulating competitive Li+ coordination of F-rich polymer and high flash point glyme electrolyte enabling high rate and intrinsically safe quasi-solid-state Li metal batteries. Angew. Chem. Int. Ed. 2025, 64, e202508281.
5. Peng, B.; Zhang, W.; Du, J.; et al. Decoupling electronic and ionic conductivities of electrochemically active LiCoO2 as solid-state electrolyte for Li metal batteries. Adv. Funct. Mater. 2025, 36, e14420.
6. Roh, J.; Do, N.; Lee, H.; et al. Towards practical all-solid-state batteries: structural engineering innovations for sulfide-based solid electrolytes. Energy. Mater. 2025, 5, 500061.
7. Kong, W. J.; Zhao, C. Z.; Sun, S.; et al. From liquid to solid-state batteries: Li-rich Mn-based layered oxides as emerging cathodes with high energy density. Adv. Mater. 2024, 36, e2310738.
8. Yang, M.; Chen, T.; Wang, G.; et al. A surface-to-interface boronation engineering strategy stabilizing the O/Mn redox chemistry of lithium-rich manganese based oxides towards high energy-density cathodes. Energy. Environ. Sci. 2025, 18, 6168-79.
9. Sun, S.; Zhao, C. Z.; Liu, G. Y.; et al. Boosting anionic redox reactions of Li-rich cathodes through lattice oxygen and Li-ion kinetics modulation in working all-solid-state batteries. Adv. Mater. 2025, 37, e2414195.
10. Zhang, Z.; Liu, J.; Chen, N.; et al. Construction of high chemical and electrochemical compatible interface of Li-rich Mn-based cathode for all-solid-state lithium batteries. J. Energy. Chem. 2025, 106, 803-11.
11. Kong, W. J.; Zhao, C. Z.; Shen, L.; et al. Bulk/Interfacial structure design of Li-rich Mn-based cathodes for all-solid-state lithium batteries. J. Am. Chem. Soc. 2024, 146, 28190-200.
12. Chen, J.; He, C.; Peng, X.; et al. Puzzle-like molecular assembly of non-flammable solid-state polymer electrolytes for safe and high-voltage lithium metal batteries. Nat. Commun. 2025, 16, 8494.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].