A scoping review of artificial intelligence in living donor liver transplantation - current status and untapped potential
Abstract
While the surgical technicalities of living donor liver transplantation (LDLT) have matured since its development several decades ago, clinical challenges remain in pre-transplantation and post-transplantation management. The ability of artificial intelligence (AI) to perform sophisticated analyses of complex non-linear relationships holds potential to aid clinical decision-making. This is particularly relevant in LDLT, where grafts are a precious resource within a dynamic setting of donor, recipient, and procedural factors that must be considered. Clinical issues of graft and patient survival, patient selection and stratification, survival predictors for expanded transplantation criteria, and post-transplantation outcomes remain relevant challenges that benefit from analysis with sophisticated AI models. This scoping review summarised 16 AI studies in pre- and post-transplantation assessment and transplant oncology, providing an overview of the current landscape and future directions for development.
Keywords
INTRODUCTION
Artificial intelligence (AI) has risen to prominent application in a number of disciplines, but widespread adoption in clinical medicine has been more cautious. The wealth of big data in healthcare and transplantation registries holds potential for capturing complex clinical relationships to improve patient outcomes, but must be balanced with ethical and liability considerations. This is particularly relevant in living donor liver transplantation (LDLT), where grafts are a precious resource with a dynamic setting of donor, recipient, and procedural factors to be taken into consideration.
AI encompasses a large umbrella of subfields, including machine learning (ML) and deep learning (DL), that make use of sets of automated computer algorithms[1]. In the studies analysed, Random Forest algorithms were a commonly used ML model, particularly in the analysis of tabular clinical data, such as graft allocation and assessment. DL models such as Convolutional Neural Networks (CNN) were used in the analysis of spatial pattern recognition through multi-layer neural networks, and have been applied in areas such as graft imaging assessment. The ability of these algorithms to analyse and interpret complex variables is particularly relevant in transplantation where numerous factors impact graft and survival outcomes.
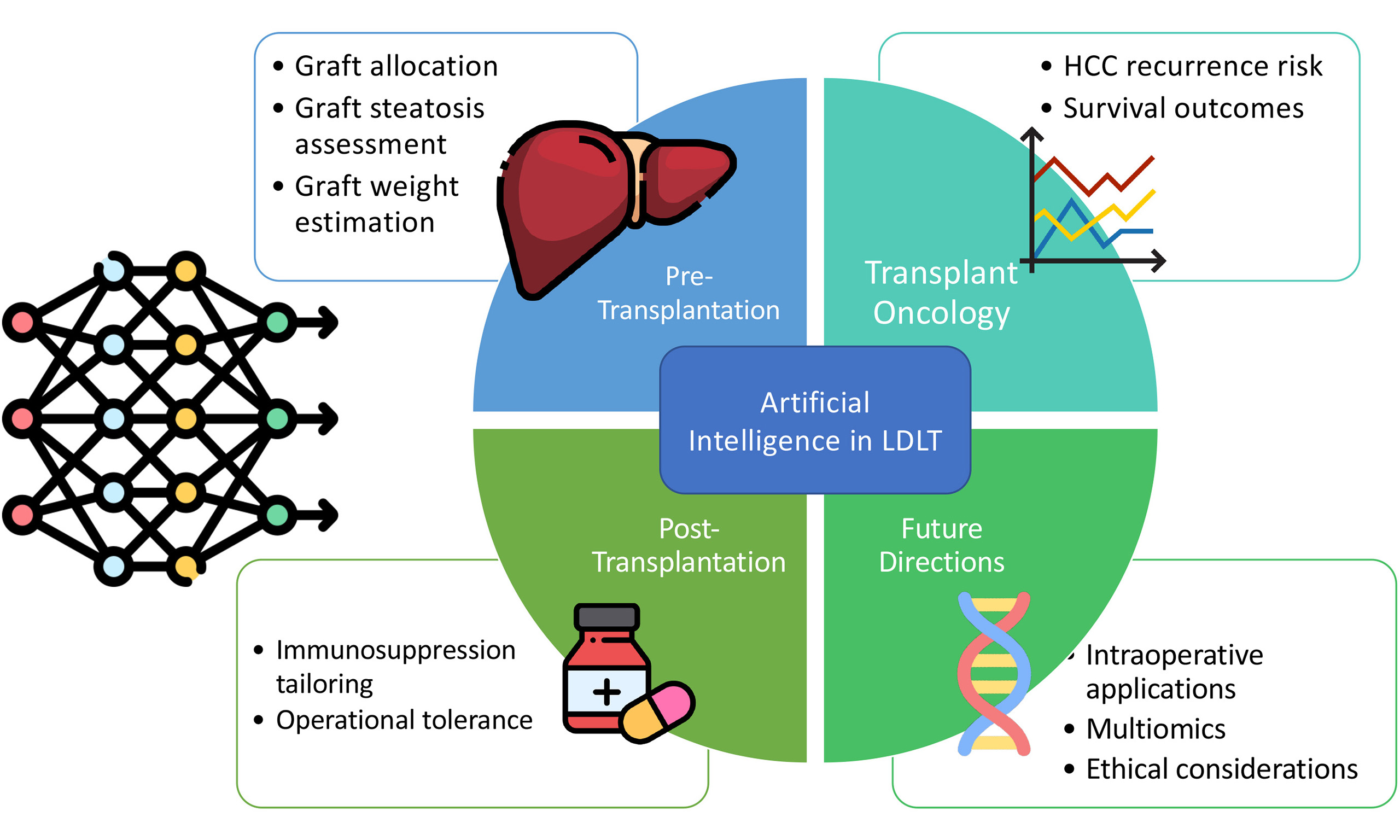
AI in the context of LDLT has principally been explored in the key areas of pre-transplantation assessment, transplant oncology, and post-transplantation management. This scoping review summarised studies that have analysed the use of various AI models in these areas thus far, and discussed potential areas for further development.
METHODOLOGY
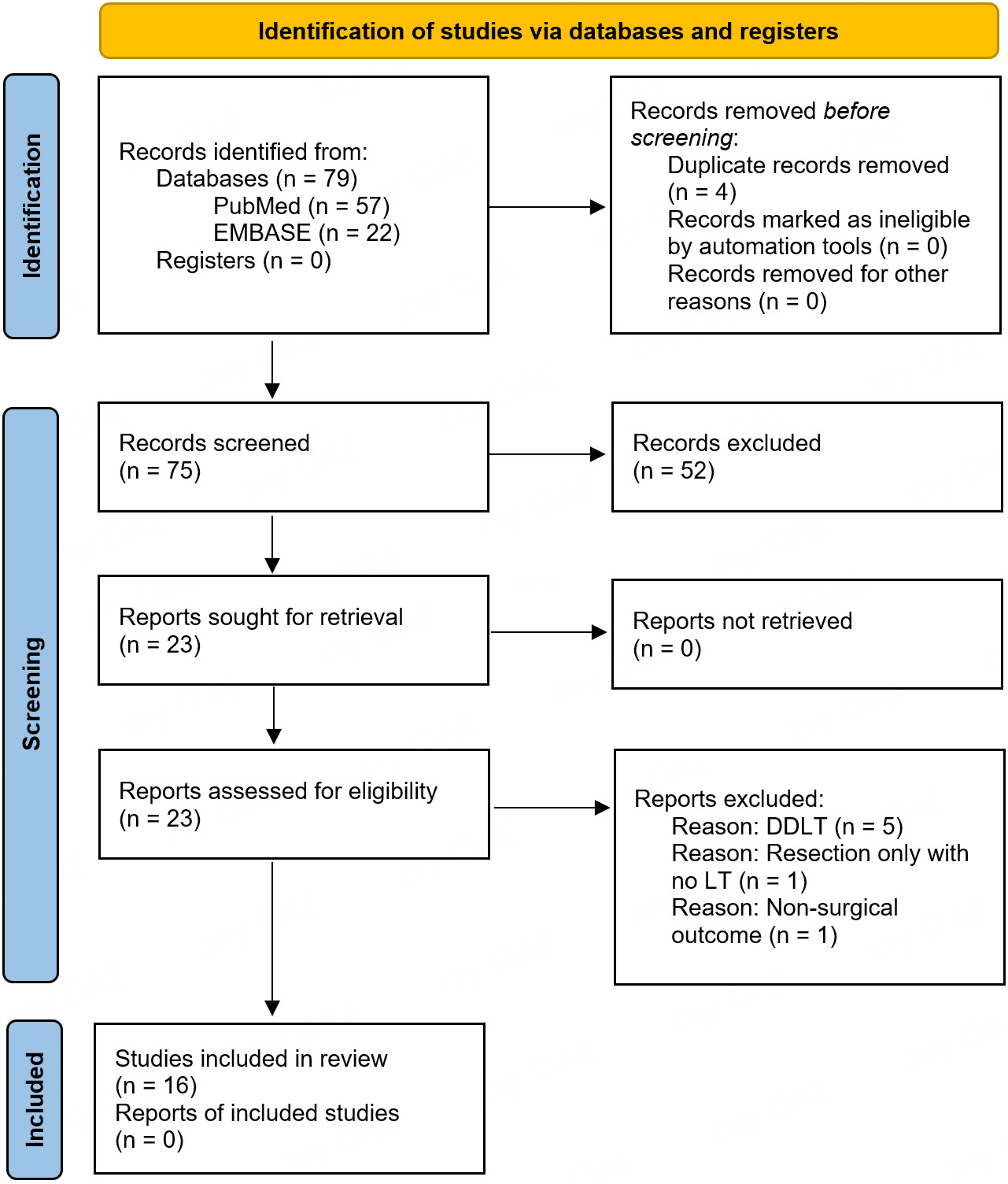
This scoping review synthesised contemporary evidence on the applications of AI in LDLT. The review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) checklist and guidance[2]. The PRISMA flowchart [Figure 1] and PRISMA-ScR checklist [Supplementary Table 1] are provided. The study protocol was not registered due to the International Prospective Register of Systematic Reviews (PROSPERO) ineligibility.
Figure 1. PRISMA flow diagram of article selection process. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; EMBASE: Excerpta Medica Database; DDLT: deceased donor liver transplantation; LT: liver transplantation.
A systematic search was performed in July 2025 on PubMed and EMBASE for English-language studies evaluating the use of AI in relation to LDLT published between January 1, 2015, and June 30, 2025. The search strategy combined relevant keywords and Medical Subject Headings (MeSH) related to the keywords “artificial intelligence”, “machine learning”, “deep learning”, and “living donor liver transplantation” [Supplementary Table 2]. Additional studies were also searched for in the references of relevant articles.
Search results were screened in two stages: title and abstract review, followed by full-text assessment to determine eligibility. Studies were included if they reported outcomes of AI methods applied in the setting of LDLT. Studies were excluded if they involved renal, lung, or cardiac transplants, if they only discussed deceased donor liver transplantations (DDLTs), or if they were not conducted on human subjects. Studies were grouped under the key areas of pre-transplantation assessment (including graft allocation and graft assessment), transplant oncology [including prognostication of hepatocellular carcinoma (HCC) recurrence], and post-transplantation management (including immunosuppression regimens).
Data extraction was performed by two independent reviewers using a standardised data collection method. Extracted variables included study characteristics (authors, year of publication, clinical application), AI model specifics (dataset and size, AI method, input, output), performance metrics (including concordance indices, F1 scores, area under the receiver operating characteristic curve), and relevant variables and outcomes. Any disagreements during data extraction were resolved by consensus.
RESULTS
Studies on pre-transplantation
Living donor graft allocation
While the majority of living donor liver transplantations are ‘directed’ (pre-existing donor-recipient relationship with an intended recipient of the donation), there has been a growing number of non-directed living liver donors[3]. Such precious altruistic donations call for the best utility-based allocation; however, there are yet to be any standardised evidence-based approaches.
An American study developed three separate ML random forest (RF) survival models to predict 10-year graft survival for right-lobe, left-lobe, and left-lateral graft transplantations, covering a total of 6,328 recipients[4], as summarised in Table 1. Decision trees were used to determine which variables influenced graft survival; an importance measure of the variables was then subsequently calculated by determining how the accuracy was affected with exclusion of the variable in the model. The graft-to-recipient weight ratio (GRWR) as a variable was notably not included in this study, and donor-to-recipient body surface area (BSA) was used instead as a surrogate data available from the Organ Procurement and Transplantation Network (OPTN) dataset. Variables identified as relevant in graft survival prediction across all three models included donor-to-recipient BSA, malignant pathology, location at time of transplant [intensive care unit (ICU) or non-ICU], and having moderate ascites. The concordance index (C-index) was 0.70, 0.63, and 0.61 for left-lateral, left, and right-lobe grafts, respectively.
Pre-transplantation - studies addressing graft allocation
| Year | Reference | Clinical use | Dataset & size | AI method | Input | Outcome | Validation | Performance metric | Predictive variables |
| 2023 | Bambha et al.[5] | Graft allocation | OPTN 6,328 grafts: 4,621 right 644 left 1,063 left lateral 70% training 30% test | Random forest survival | Donor and recipient data from both directed and non-directed LDLT | 10-year graft survival | Internal | C-index Left-lateral graft 0.70 Left lobe graft 0.63 Right graft 0.61 | Donor-to-recipient BSA Malignant pathology Location at time of transplant (ICU or non-ICU) Moderate ascites |
While this model is a step toward guiding graft allocation, this is a complex matching process that also varies in the “real-world” context according to an array of practical variables, such as individual transplant centre programmes, LDLT volume and frequency, and the characteristics of local waiting lists. Further ML analyses with a wider dataset may shed light on optimal allocation recommendations.
Graft steatosis assessment
Hepatic macrovesicular steatosis is a significant factor for declining grafts for transplantation[5], as grafts with more than 30% steatosis have been shown to have a higher risk of graft failure and early allograft dysfunction[6,7]. Careful donor selection is important for recipient outcomes, but overly stringent criteria may unnecessarily restrict an already limited pool of donors. A number of studies using mainly RF survival or convolutional neural network (CNN) methods have addressed this clinical issue, as summarised in Table 2.
Pre-Transplantation - studies addressing steatotic graft assessment
| Year | Reference | Clinical use | Dataset & size | AI method | Input | Outcome | Validation | Performance metric | Predictive variables/ relevant outcomes |
| 2021 | Lim et al.[9] | Macrovesicular steatosis with non-invasive variables | Single-centre 1,652 donors 70.5% training 29.5% test | Regularised discriminant analysis Flexible discriminant analysis Mixture discriminant analysis Random forest Support vector machine Deep neural network | Donor demographics and CT Hounsfield units of the liver | Macrovesicular steatosis | Internal | Training set: Random forest 100.0% accuracy [AUROC 1.00 (95%CI 1.00-1.00)] (P < 0.01) Test set: FDA 80.9% accuracy [AUROC 0.87 (95%CI 0.83-0.90)] (P < 0.01) | Liver Hounsfield unit ALT level BMI HDL-cholesterol Total cholesterol Age Glucose level |
| 2020 | Sun et al.[12] | Graft steatosis | Single-centre 96 liver sections 30 training set 66 test set | Convolutional neural network | H&E whole slide image | Steatosis | Internal | Training set ICC 0.88 Test set ICC 0.85 | N/A |
| 2023 | Gambella et al.[14] | Graft steatosis | Single-centre 292 biopsies 504 training 56 testing | Convolutional neural network | H&E whole slide image | Steatosis | Internal | Banff approach ICC 0.93 (P < 0.001) | Steatosis scores higher from pathologist review |
| 2024 | Jiao et al.[15] | Graft steatosis | 113 slides | Segment anything model | Liver biopsy slide | Steatosis | Internal | Accuracy 91.9% | N/A |
| 2021 | Narayan et al.[13] | Graft steatosis and risk of early graft failure | 90 biopsies | Computer vision and convolutional neural networks | H&E slide | Steatosis Early allograft dysfunction | Internal | CVAI steatosis adjusted OR 1.34 (95%CI 1.03-1.75, P = 0.03) | CVAI steatosis was independently associated with early allograft dysfunction; Steatosis scores higher from pathologist review |
Lim et al. developed ML models using only non-invasive variables from 1,652 donors for risk prediction of hepatic steatosis. The model aimed to discriminate between donors with ≤ 5% macrovesicular steatosis who would not require further invasive liver biopsy, from those with > 5% who would otherwise require further investigation[8]. Binary outcome variables were analysed using regularised discriminant analysis (RDA), flexible discriminant analysis (FDA), and mixture discriminant analysis (MDA). Three separate ML algorithms were also used: RF, support vector machine (SVM), and deep neural network (DNN).
The RF model showed the highest accuracy of 100.0% {area under receiver operating characteristic curve (AUROC) 1.00 [95% confidence interval (CI) 1.00-1.00]} (P < 0.01) in the training set, while FDA achieved the highest accuracy in the test set at 80.9% [AUROC 0.87 (95%CI 0.83-0.90)] (P < 0.01). The results may be limited by a lack of external validation, particularly as the results were obtained retrospectively from a single centre. However, the models were developed from a relatively larger sample size, incorporating non-invasive variables including Hounsfield unit on computed tomography (CT) scans that may spare potential donors from avoidable invasive biopsies, and allow a greater number of potential candidates to be considered.
The Banff consensus recommendations were developed to improve standardisation of steatosis assessment on donor livers[9]; however, there remains significant inter-observer variation in clinical application[10]. Existing histological techniques may also affect interpretation of steatosis; artefactual white spaces on frozen section preparations mimic steatosis and may lead to overestimation of steatosis[11]. Given these challenges, four studies developed AI algorithms for steatosis prediction from histological interpretation[11-14]. Gambella performed analysis with the largest sample size of 292 biopsies and produced the first published deep-learning algorithms (DLAs) for steatosis assessment based on Banff recommendations. The CNN model showed promising concordance with pathologist assessment with an intraclass correlation coefficient of 0.93 (P < 0.001) for the final steatosis score that combined both high-power and low-power assessment.
It was also noteworthy that pathologist assessment tended to overestimate steatosis across the different studies, including both CNN models by Gambella and Sun[11], and the computer vision artificial intelligence (CVAI) model by Narayan et al.[12]. Furthermore, two multivariable logistic regression models found that CVAI steatosis scores were significantly associated with early allograft dysfunction with adjusted odds ratio (OR) 1.34 (95%CI 1.03-1.79, P = 0.03), whereas the average of pathologist steatosis scores were not (adjusted OR 1.02, 95%CI 0.99-1.05, P = 0.13) (but may be affected by underpower of the study)[12]. Nevertheless, given that steatosis interpretation is mainly affected by observer and artefactual errors, the AI model's accuracy may be attributable to the segmentation framework, which enabled individual liver droplets to be identified with greater precision. This is a promising finding, suggesting that the application of AI may lead to greater accuracy in histological analysis and allow a greater number of eligible donors to be accepted.
Living donor graft weight estimation
A number of studies investigated applications of AI for pre-operative graft volumetry and weight estimation as summarised in Table 3. This carries particular clinical significance for adult LDLT in which accurate prediction is important for both donor and recipient - small-for-size syndrome is associated with graft malfunction[15], while insufficient donor remnant may threaten donor safety.
Pre-transplantation - studies addressing graft assessment and pre-operative planning
| Year | Reference | Clinical use | Dataset & size | AI method | Input | Outcome | Validation | Performance metric | Relevant variables/outcomes |
| 2024 | Oh et al.[27] | Graft volume and segment vasculature | Single-centre 114 patients; 103 training set 11 validation set | 3D residual U-Net model | Segmented CT images | Graft volume Segment vasculature | Internal | R-squared value 0.76 (P = 0.018) | Included segmentation of portal and hepatic veins |
| 2023 | Giglio et al.[18] | Graft weight estimation | Single-centre 872 living donors; 80% training set 20% test set | Supervised ML models; SVMPOLY, SVMLINEAR, quantile regression with LASSO penalty, MONMLP, robust linear model, QRF, Bayesian regularised neural networks, extreme gradient boosting, bidirectional associative memory, generalised additive models using splines | Donor demographics Graft type CT-estimated graft volume CT-estimated total liver volume | Graft weight | Internal | R-squared value 0.89 | BMI |
| 2022 | Park et al.[23] | Graft weight estimation | Single-centre 581 donors; 207 development group 374 validation group | DLA-generated automated segmentation of whole liver | CT-estimated right lobe graft volume | Graft weight | Internal | Concordance correlation coefficient 0.83 (95%CI 0.80-0.86, P < 0.001) Small change in volume (Bland-Altman 95% LOA 0.05% ± 3.0% measured liver volume) | Sex and BMI; GW tends to underestimated in men (mean bias -2.6%) and higher BMI (mean bias -4.2%) |
| 2023 | Yang et al.[25] | Graft weight estimation | Single-centre 248 patients; 20 donors, 228 healthy persons for check-up | UNETR model (transformer encoder and CNN-based decoder) | CT-estimated right lobe graft volume | Right lobe graft weight | Internal | Segmentation DSC 95.9% ± 1.0 (93.5-97.4) Graft weight estimation PAE 8.6 ± 4.7 (0.1-19.9) | Fully automatic division of liver into left and right lobes |
| 2020 | Kavur et al.[28] | Graft volume | 20 donors | DEU_DeepMedic, DEU-Nifty-Net, ITU_U-Net, METU_U-Net, X_U-Net, Y_U-Net | CT liver segmentation | Graft volume | Internal | Volume overlap Relative volume difference Average symmetric surface distance Root mean square symmetric distance Maximum symmetric surface distance | Deep learning methods more reliable than semi-automatic methods |
The ideal GRWR of at least 0.8 or graft volume standard liver volume ratio of at least 0.4 is recommended[16]. However, the calculation is dependent on accurate liver segmentation and volumetric graft weight estimation. AI and DL or ML have the potential to improve the accuracy of graft volumetry and graft volume-to-weight estimation, and further improve transplant outcomes.
Giglio performed the largest study using data from 872 living donors of right-lobe, left-lobe, or left-lateral sector grafts[17]. Supervised ML models were trained to predict graft weight and compared with currently available methods, such as multiplying graft volume by a conversion factor of 1.19 mL/g[18] or using a correlation coefficient of 0.85[19] for right lobes. ML showed better graft volume estimation than existing methods; the mean absolute error (MAE) was 50 ± 61 g, which was lower than that of the Lemke model (63 ± 53 g; P < 0.001)[20] and the Yoneyama model (62 ± 63g; P < 0.001)[19]. Graft weight prediction by ML also showed higher precision for both right and left lobes. For right-lobe grafts, 76% of predictions had absolute errors ≤ 10%, which was superior to the Lemke model (69%; P < 0.001) and the Yoneyama model (70%; P = 0.001). However, predictions for left-lateral grafts were not significantly improved with ML.
The superiority of ML prediction could be explained by additional information gained from donor characteristic data. Existing methods assume linear relationships between graft volume and weight; however, ML may better capture the non-linear impact of donor body mass index (BMI) and age in relation to hepatic steatosis[21] and actual donor liver density.
Park performed analysis with a focus on automated liver segmentation for right lobe grafts[22]. Portal venous phase CT data was analysed with 30 donors in the development group with an experienced radiologist for training purposes; the data were then subsequently analysed using GoCDSS software (SmartCareworks Inc., Seoul, Korea) which applied a DLA[23] for automated liver segmentation. DLA-generated segmentation showed error requiring correction in 28.6% of donors, but was associated with only a small change in volume [Bland-Altman 95% limits of agreement (LOA), 0.05% ± 3.0% of measured liver volume]. Multivariable linear regression analysis was performed to evaluate any confounding effect of hepatic steatosis on graft weight, and a conversion formula for graft weight from CT-measured graft volume was developed using univariable linear regression analysis. Hepatic steatosis was found not to have a significant effect on graft weight in this cohort (coefficient -0.34; P = 0.67) and thus a formula was developed without excluding donors with steatosis. However, few donors in the data had steatosis, which may affect the generalisability of the findings to moderate or severely steatotic donors. The validation group was subsequently used to analyse agreement between estimated and measured graft weights, and the concordance correlation coefficient was 0.83 (95%CI 0.80-0.86, P < 0.001). This study demonstrated benefits in the use of DL to produce highly accurate segmentation without requiring any additional correction in 70% of donors with an average processing time of 1.8 min.
While Park’s algorithm still required manual division of the liver into left and right lobes along Cantlie’s line, Yang et al. developed a fully automatic model with 248 patients[24]. A UNETR (U-Net Transformer) model was adopted which used a CNN to learn from contextual input (transformer encoder) and predict liver segmentation output (decoder)[25]. The model achieved a slightly higher accuracy in right lobe graft estimation with a correlation coefficient to actual graft weight of 0.86 [percentage absolute error (PAE) 8.6% ± 4.7%].
Another DL-based 3D residual U-Net model developed by Oh et al. with 114 donors demonstrated better volume estimation than manual assessment by surgeons[26]. The DL model showed higher correlation to actual graft weight with an R-squared value of 0.76 (P = 0.02) compared with 0.68 (P = 0.04) for the surgeon. While conventional estimation techniques tend to overestimate graft volume due to the assumption of the same density of liver as water, this model interestingly tended to underestimate graft volume by an average of 1.8%. However, the relatively high level of accuracy may still prove clinically beneficial, particularly for avoiding small-for-size graft syndrome.
Kavur et al. further evaluated 12 DL models: 6 semi-automatic and 6 fully-automatic algorithms, but on a comparatively smaller group of 20 donors (8 training and 12 test datasets)[27]. The automatic models outperformed semi-automatic methods; ensemble analysis was further developed for fusion of individual results - majority voting (MV) and simultaneous truth and performance level estimation (STAPLE) algorithms were combined as inputs in a fusion approach to yield even higher levels of accuracy. The best model was the fully-automatic DL model Istanbul Technical University (ITU)_U-Net that scored 79.6.
Given the highly imaging-based and mathematical nature of graft weight estimation, this is an area that is likely to benefit from AI input; however, expansion with larger datasets across different transplant centres and donor ethnicities and characteristics would likely enhance the generalisability and accuracy of these algorithms.
Studies on transplant oncology
Prognostication - post-transplantation hepatocellular carcinoma recurrence risk
Fewer studies have been performed on long-term survival outcomes after LDLT. These analyses have particular relevance in the setting of LDLT; expert centres may perform LDLT for patients beyond traditional transplant criteria, which are more strictly adhered to in deceased donor transplantation waiting lists. While expanded criteria allow more patients to benefit from LT, AI models may provide clinical guidance on recurrence risks, to better inform patients, donors, and clinicians on post-transplant outcomes. Studies addressing these outcomes are summarised in Table 4.
Studies addressing post-transplantation outcomes
| Year | Reference | Clinical use | Dataset & Size | AI method | Input | Outcome | Validation | Performance metric | Relevant variables/outcomes |
| 2023 | Giglio et al.[42] | Prediction of early graft failure | Multi-centre 88 transplant centres across 18 European countries 80% training set 20% test set | Lasso regression Artificial neural network | Donor and recipient characteristics | Early graft failure | External | AUC 0.69 (95%CI: 0.63-0.75) | 3-month graft failure: Donor age Donor and recipient BMI Left-sided grafts |
| 2024 | Altaf et al.[29] | Prediction of HCC recurrence | Single-centre 192 HCC patients | Multilayer perceptron model | Recipient characteristics Liver disease and cirrhosis parameters Tumour parameters | Risk of HCC recurrence | Internal | AUC validation cohort: 0.77 Accuracy 84.2% Precision 60% F1 score 66.7% | Pre-transplant factors Tumour grade |
| 2025 | Raji et al.[40] | Recipient outcomes | UNOS dataset 135,709 deceased donors 6,180 living donors | Deep learning4j Multilayer perceptron classifier | Donor characteristics | Short-term and long-term survival | Internal | Living donor: Sensitivity 99.9% Specificity 99.9% Accuracy 99.91% MAE 0.0009 RMSE 0.021 RAE 0.26% RRSE 4.99% | Donor age, blood type, gender, BMI Recipient BMI, encephalopathy, cold ischaemic time, albumin, ascites, bilirubin, INR, MELD/PELD score, creatinine, sodium, gender, age, blood group, number of previous transplants |
| 2025 | Gangadhar et al.[41] | Survival difference between LDLT and DDLT | Single-centre 72,581 recipients; 68,108 DDLT 4,473 LDLT Training set 70% Test set 30% | Random forest classifier | Recipient characteristics | 10-year survival difference between LDLT and DDLT | Internal | C-index (LDLT) 0.67 | Survival gain from LDLT greatest for primary sclerosing cholangitis and HCV |
Altaf et al. described a deep-learning model to determine the risk of HCC recurrence after LDLT[28]. A multilayer perceptron model was used to analyse 192 patients who were categorised into low-risk and high-risk post-transplant recurrence groups. Traditionally, tumour dimensions and alpha-fetoprotein (AFP) levels have been used to assess recurrence risk - this model allowed a more dynamic risk assessment by assigning quantitative weight based on Shapley scores. Two AI models were developed; one with pre-transplant factors, and the second incorporating both pre-transplant factors and tumour grade. The AI model using pre-transplant factors had a training and validation area under the curve (AUC) of 0.86 and 0.71, respectively, and accuracy, precision, and F1 scores of 78.9%, 50%, and 55.6%, respectively. The AI model using both pre-transplant factors and tumour grade had training and validation AUCs of 0.87 and 0.77, respectively, and accuracy, precision, and F1 scores of 84.2%, 60%, and 66.7%, respectively, showing better prediction results compared to using pre-transplant factors alone.
Notably, the deep-learning model also showed that response to downstaging (or conversion therapy) was the third most important prognostic factor for HCC recurrence after tumour diameter and AFP levels. This implies that patients beyond traditional criteria may benefit from neoadjuvant therapy to achieve better survival outcomes[29-35].
Survival outcomes
There remains a lack of a precise model to predict post-transplantation survival. The Model for End-stage Liver Disease (MELD) score has traditionally been used as a prognostic indicator to assess severity of chronic liver disease, but it has limitations in precise estimation of survival and does not capture non-hepatic factors that may affect outcomes[36-38].
In light of these challenges, Raji et al. described a prognostic deep-learning model to analyse long-term outcomes comparing living donors with deceased donors[39]. Data was collected from the United Network for Organ Sharing (UNOS) database including 6,180 LDLTs and 135,709 deceased donor transplantations. Data was used to train the DL model Deeplearning4jMultilayer Perceptron (Dl4jMLP) classifier for survival prediction using 23 top-ranked attributes for survival prediction, including donor age, gender, and BMI, recipient MELD/Paediatric End-stage Liver Disease (MELD/PELD) score, ascites, and cold ischaemic time. For the living donor dataset, the model obtained sensitivity, specificity, and accuracy scores of 99.9%, 99.9%, and 99.91%, respectively. The MAE was 0.0009, root mean squared error (RMSE) 0.021, relative absolute error (RAE) 0.26%, and root relative square error (RRSE) 4.99%. The model output was close to actual survival data, which has proved useful in comparison to using the MELD score alone.
A comparison study between LDLT and DDLT was also performed by Gangadhar et al., but with the use of the ML algorithm termed decision path similar matching (DPSM)[40]. Their data was collected from 72,581 recipients from the Scientific Registry of Transplant Recipients database, including 68,108 DDLT and 4,473 LDLT. DPSM was used for matching to minimise differences between the two groups, and demonstrated a mean 10-year survival gain of 10.3% over DDLT, with the greatest benefit for primary sclerosing cholangitis (12.4% ± 5.3%) and hepatitis C (12.1% ± 4.7%) compared to other aetiologies.
Giglio et al. performed a study using data from the European Liver Transplant Registry (ELTR) to analyse early graft failure within 3 months following adult LDLT; both least absolute shrinkage and selection operator (LASSO) and artificial neural network (ANN) algorithms were investigated[41]. The ANN showed better performance; after training from the data of 2,073 transplantations, the final ANN showed an AUC of 0.70 at cross-validation, and 0.69 (95%CI: 0.63-0.75) on the test set. The model was also able to stratify recipients into groups with different risks of early graft failure; predicted risk scores were 6.5%, 29%, and 37% for failure risks < 5%, ≥ 30%, and 40%, respectively. However, this model did not capture graft failure risks related to variables pertaining to graft anatomy, including the additional risks associated with multiple biliary or arterial anastomoses and venous reconstructions. Despite these limitations, the study incorporated MELD and Child-Pugh scores, and may be a step toward providing both the clinical team and potential donors with data that may guide their decisions.
Studies on post-transplantation management
Prediction of tacrolimus concentration
Calcineurin inhibitors, particularly tacrolimus, are an important mainstay for immunosuppression; however, their narrow therapeutic index requires careful titration to avoid both toxicity with overexposure and risk of graft rejection in underdosing[42]. Furthermore, pharmacological studies have produced recommendations for adult patients, but less evidence has been available for paediatric recipients[43]. Tacrolimus samples have also shown that target range concentration is difficult to achieve in vivo; out of 1,827 plasma samples from 163 infant patients, only 32% were within the target range, and more than half were below the range[44]. As such, Song et al. developed ML models aimed at prediction of tacrolimus concentration for better clinical outcomes, as summarised in Table 5.
Studies addressing prediction of immunosuppression concentration
| Year | Reference | Clinical use | Dataset & size | AI method | Input | Outcome | Validation | Performance metric | Relevant variables |
| 2023 | Song et al.[45] | Tacrolimus concentration prediction in paediatric recipients | Single-centre 163 patients | Logistic regression Linear regression Support vector regression Ridge regression Lasso regression K-nearest neighbour Multi-layer perceptron Decision tree Extra tree regression Random forest Adaptive boosting Gradient boosting regressor Bagging regressor | Patient demographics CYP3A5 genotype | Tacrolimus concentration | Internal | Absolute prediction error | Donor and recipient CYP3A5 genotypes, GRWR, urea, haemoglobin, albumin, BMI |
Multivariate regression analysis was used to identify CYP3A5 genotypes in both recipient and donor, GRWR, BMI, urea, haemoglobin, and albumin as statistically significant variables. Different ML models were subsequently developed with covariate combinations selected to give the lowest MAE. Their best performing model was the Ridge model with an ideal rate (predicted value with relative error ≤ 30%) of 55.05% (P < 0.05). While further development may be required to improve predictive accuracy, especially with other potential factors such as drug-drug and drug-food interactions, their work could be an important foundational step in optimising post-transplant immunosuppressant titrations.
Immunosuppression tailoring and operational tolerance
Immunosuppression is key to graft and patient survival, and calls for a delicate balance between prevention of rejection and avoidance of side effects and de novo or recurrent malignancy.
Calcineurin inhibitors, such as tacrolimus, have been the mainstay of immunosuppression, but previous studies have shown that higher levels of exposure are associated with a greater risk of HCC recurrence[45-47], while mammalian target of rapamycin (mTOR) inhibitors, such as everolimus and sirolimus, may reduce recurrence rates[48]. Common practice is to minimise tacrolimus exposure through combination regimens with mTOR inhibitors, but studies have yet to demonstrate clear benefits from this combination[49]. This is an important area of post-transplant management where AI could be used for further analysis to tailor and optimise regimen choices, thereby maximising graft and patient survival outcomes.
Taken a step further, operational tolerance - complete and safe withdrawal of immunosuppressants after transplantation without graft loss - is a potential area of study that could lead to further improvement in survival outcomes. This holds particular potential in liver transplantation due to its immunologically privileged nature compared to other solid organ transplantations[50]. Prior studies have investigated clinical parameters and biomarkers that may predict candidates for tolerance, such as allograft gene expression patterns, serum immune exhaustion markers, and histological allograft scores; however, much of the evidence remains conflicting[51]. Indeed, regulatory T cells and complex individualised immunomodulatory expression have already been elucidated as integral in the role of tolerance. However, the complex relationships with inter-recipient variation bring the area into the realm of personalised medicine, where AI analysis might prove beneficial with larger pooled datasets.
DISCUSSION
This study showed that a number of studies have investigated the potential utilisation of AI in LDLT; however, further development is required to develop robust models for clinical application. A large proportion of studies have been dedicated to pre-transplantation assessment, particularly graft steatosis and graft weight estimation, with some studies demonstrating superior AI performance in estimation of steatosis compared with expert pathologist assessment.
While the results were promising, further development and validation would benefit the accuracy and applicability of the models. Most of the studies used internal validation and AUC performance metrics; future studies would benefit from further external validation and decision-curve analyses to determine clinical utility. Restrictions due to limiting the search to English-language studies and databases may also limit the scope of studies incorporated into analysis.
Other challenges remain where the use of AI has yet to be developed; studies applied to transplant oncology are still limited. LDLT leverages expanded criteria to allow a greater number of patients to maximise survival. In this regard, AI has potential for providing guidance for patient stratification and prediction of survival outcomes, especially when considering expanded criteria. Tsilimigras et al.[52] used a ML Classification and Regression Tree (CART) model to analyse preoperative and postoperative variables that may predict overall survival in different HCC stages, and hence implications for resection especially for Barcelona Clinic Liver Cancer (BCLC)-B patients. Radiologic tumour burden score was identified as the best predictor of overall survival for BCLC-B patients, and as an extension of this study, further AI analyses could be applied to investigate similar predictors in transplantation.
Besides clinical prediction models, genomics is increasingly relevant in the era of personalised medicine. ML algorithms could be designed to utilise the large pool of genomic data to identify relevant genes and molecular targets for biomarkers of disease risk, actionable targets for therapy, and survival outcome predictors. For instance, Chaudhary et al.[53] developed a deep-learning model to analyse multiomic data and identified gene expression signatures KIRT19, EPCAM, and BIRC5 and Wnt signaling pathways as being associated with poor survival in HCC. While the model was built from only a small amount of training data, their findings demonstrated the possible application of AI in developing tools for genomics, which may also have applications in guiding transplant survival outcome prediction and patient stratification.
Another important area for development is the use of AI in intraoperative decision support. Most of the AI studies have focused on static preoperative or postoperative variables; however, the dynamic, time-dependent nature of liver transplantation could benefit from procedural guidance with AI. Quantitative metrics from computer-vision-based techniques may provide insight in terms of real-time anatomical support, early allograft dysfunction, biliary complications, thrombotic events, and long-term graft survival. These applications make use of intra-operative data to provide procedural-level outcome modelling beyond static preoperative data.
Despite its potential, there are ethical and applicability issues, which are particularly important in high-risk procedures such as LDLT. The “black box” nature of ML and DL remains an ethical concern, particularly in the context of LDLT and the allocation of scarce resources such as liver grafts. Neural networks use algorithms that even developers are unable to fully understand, leading to issues with reliability, responsibility attribution, and system failure management. Donors, recipients, and their families may also find it difficult to accept life-altering decisions made by neural networks that lack full transparency.
Moving forward, a number of strategies may be developed to improve the reliability and transparency of AI. As AI software is a function of the data on which they are trained[54], ensuring that large-scale, high-quality prospective data is used would improve the quality of AI model training, with measures to ensure the safeguarding of data privacy and protection as these registries expand. Ongoing vigilance to identify and correct inherent biases in registry and AI model training is also particularly crucial in the setting of LDLT with diverse donor and recipient characteristics, and ethical considerations.
LDLT is a dynamic area at both the donor/recipient and institutional level, with different protocols and pathways employed at different transplant centres. No single AI model is currently developed for universal adoption; different localities with varying graft availability may also affect the balance of AI model recommendations. Continued inter-institutional and global cooperation would benefit the development of AI that reflects and enhances the dynamic needs in a real-world context. Ultimately, AI should be a decision-assisting, rather than decision-making, tool, with clinicians working closely with donors and recipients to work towards the best clinical and ethical outcomes.
CONCLUSION
While a number of studies have investigated the use of ML or DL in graft segmentation and weight analysis, steatosis, and post-transplantation survival outcomes, its potential for application in other areas remains largely untapped. With the large amount of existing data from the many years of LDLT, application of AI to this contextual information could elucidate new advances in pre- and post-transplant management.
DECLARATIONS
Authors’ contributions
Conceptualisation, investigation, analysis, writing - original draft: Ho KKY
Conceptualisation, project administration, supervision: Chan ACY
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Peloso A, Moeckli B, Delaune V, Oldani G, Andres A, Compagnon P. Artificial intelligence: present and future potential for solid organ transplantation. Transpl Int. 2022;35:10640.
2. Tricco AC, Lillie E, Zarin W, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169:467-73.
3. Herbst LR, Herrick-Reynolds K, Bowles Zeiser L, et al. The landscape of nondirected living liver donation in the United States. Transplantation. 2022;106:1600-8.
4. Bambha K, Kim NJ, Sturdevant M, et al. Maximizing utility of nondirected living liver donor grafts using machine learning. Front Immunol. 2023;14:1194338.
5. Linares I, Hamar M, Selzner N, Selzner M. Steatosis in liver transplantation: current limitations and future strategies. Transplantation. 2019;103:78-90.
6. Spitzer AL, Lao OB, Dick AA, et al. The biopsied donor liver: incorporating macrosteatosis into high-risk donor assessment. Liver Transpl. 2010;16:874-84.
7. Dutkowski P, Schlegel A, Slankamenac K, et al. The use of fatty liver grafts in modern allocation systems: risk assessment by the balance of risk (BAR) score. Ann Surg. 2012;256:861-8; discussion 868.
8. Lim J, Han S, Lee D, et al. Identification of hepatic steatosis in living liver donors by machine learning models. Hepatol Commun. 2022;6:1689-98.
9. Neil DAH, Minervini M, Smith ML, Hubscher SG, Brunt EM, Demetris AJ. Banff consensus recommendations for steatosis assessment in donor livers. Hepatology. 2022;75:1014-25.
10. Ho S, Kuo E, Allende D, et al. Heterogeneity of hepatic steatosis definitions and reporting of donor liver frozen sections among pathologists: a multicenter survey. Liver Transpl. 2022;28:1540-2.
11. Sun L, Marsh JN, Matlock MK, et al. Deep learning quantification of percent steatosis in donor liver biopsy frozen sections. EBioMedicine. 2020;60:103029.
12. Narayan RR, Abadilla N, Yang L, et al. Artificial intelligence for prediction of donor liver allograft steatosis and early post-transplantation graft failure. HPB. 2022;24:764-71.
13. Gambella A, Salvi M, Molinaro L, et al. Improved assessment of donor liver steatosis using Banff consensus recommendations and deep learning algorithms. J Hepatol. 2024;80:495-504.
14. Jiao J, Tang H, Sun N, Zhang X. Artificial intelligence-aided steatosis assessment in donor livers according to the Banff consensus recommendations. Am J Clin Pathol. 2024;162:401-7.
15. Law JH, Kow AW. Prediction and management of small-for-size syndrome in living donor liver transplantation. Clin Mol Hepatol. 2025;31:S301-26.
16. Kow AWC, Liu J, Patel MS, et al.; ILTS-iLDLT-LTSI SFSS Working Group. Post living donor liver transplantation small-for-size syndrome: definitions, timelines, biochemical, and clinical factors for diagnosis: guidelines from the ILTS-iLDLT-LTSI consensus conference. Transplantation. 2023;107:2226-37.
17. Giglio MC, Zanfardino M, Franzese M, et al. Machine learning improves the accuracy of graft weight prediction in living donor liver transplantation. Liver Transpl. 2023;29:172-83.
18. Chan SC, Liu CL, Lo CM, et al. Estimating liver weight of adults by body weight and gender. World J Gastroenterol. 2006;12:2217-22.
19. Yoneyama T, Asonuma K, Okajima H, et al. Coefficient factor for graft weight estimation from preoperative computed tomography volumetry in living donor liver transplantation. Liver Transpl. 2011;17:369-72.
20. Lemke AJ, Brinkmann MJ, Schott T, et al. Living donor right liver lobes: preoperative CT volumetric measurement for calculation of intraoperative weight and volume. Radiology. 2006;240:736-42.
21. Fan R, Wang J, Du J. Association between body mass index and fatty liver risk: a dose-response analysis. Sci Rep. 2018;8:15273.
22. Park R, Lee S, Sung Y, et al. Accuracy and efficiency of right-lobe graft weight estimation using deep-learning-assisted CT volumetry for living-donor liver transplantation. Diagnostics. 2022;12:590.
23. Ahn Y, Yoon JS, Lee SS, et al. Deep learning algorithm for automated segmentation and volume measurement of the liver and spleen using portal venous phase computed tomography images. Korean J Radiol. 2020;21:987-97.
24. Yang X, Park S, Lee S, et al. Estimation of right lobe graft weight for living donor liver transplantation using deep learning-based fully automatic computed tomographic volumetry. Sci Rep. 2023;13:17746.
25. Hatamizadeh A, Nath V, Tang Y, Yang D, Roth H, Xu D. Swin UNETR: swin transformers for semantic segmentation of brain tumors in MRI images 2022. arXiv 2022;arXiv:2201.01266. Available from https://doi.org/10.48550/arXiv.2201.01266 [accessed 17 March 2026].
26. Oh N, Kim JH, Rhu J, et al. Comprehensive deep learning-based assessment of living liver donor CT angiography: from vascular segmentation to volumetric analysis. Int J Surg. 2024;110:6551-7.
27. Kavur AE, Gezer NS, Barış M, et al. Comparison of semi-automatic and deep learning-based automatic methods for liver segmentation in living liver transplant donors. Diagn Interv Radiol. 2020;26:11-21.
28. Altaf A, Mustafa A, Dar A, et al. Artificial intelligence-based model for the recurrence of hepatocellular carcinoma after liver transplantation. Surgery. 2024;176:1500-6.
29. Chiang CL, Chiu KWH, Chan KSK, et al. Sequential transarterial chemoembolisation and stereotactic body radiotherapy followed by immunotherapy as conversion therapy for patients with locally advanced, unresectable hepatocellular carcinoma (START-FIT): a single-arm, phase 2 trial. Lancet Gastroenterol Hepatol. 2023;8:169-78.
30. Chiang CL, Chiu KW, Lee FA, Kong FS, Chan AC. Combined stereotactic body radiotherapy and immunotherapy versus transarterial chemoembolization in locally advanced hepatocellular carcinoma: a propensity score matching analysis. Front Oncol. 2021;11:798832.
31. Wong TC, Chiang CL, Lee AS, et al. Better survival after stereotactic body radiation therapy following transarterial chemoembolization in nonresectable hepatocellular carcinoma: a propensity score matched analysis. Surg Oncol. 2019;28:228-35.
32. Wong TC, Lee VH, Law AL, et al. Prospective study of stereotactic body radiation therapy for hepatocellular carcinoma on waitlist for liver transplant. Hepatology. 2021;74:2580-94.
33. Mehta N, Frenette C, Tabrizian P, et al. Downstaging outcomes for hepatocellular carcinoma: results from the Multicenter Evaluation of Reduction in Tumor Size before Liver Transplantation (MERITS-LT) Consortium. Gastroenterology. 2021;161:1502-12.
34. Tabrizian P, Holzner ML, Mehta N, et al. Ten-year outcomes of liver transplant and downstaging for hepatocellular carcinoma. JAMA Surg. 2022;157:779-88.
35. Ho KKY, Chiang CL, Wong T, et al. Survival impact of conversion therapy for Barcelona Clinic Liver Cancer (BCLC) stage B and C hepatocellular carcinoma - a propensity score matching analysis. Hepatobiliary Surg Nutr. 2025;14:1063-77.
36. Gotthardt D, Weiss KH, Baumgärtner M, et al. Limitations of the MELD score in predicting mortality or need for removal from waiting list in patients awaiting liver transplantation. BMC Gastroenterol. 2009;9:72.
37. Vasconcellos M, Zamith LM. Impact of the MELD score on the survival of hepatocellular carcinoma transplantation patients in Brazil: a systematic review. Rev Col Bras Cir. 2020;46:e20192392.
38. VanDerwerken DN, Wood NL, Segev DL, Gentry SE. The precise relationship between model for end-stage liver disease and survival without a liver transplant. Hepatology. 2021;74:950-60.
39. Raji CG, Chandra SSV, Gracious N, Pillai YR, Sasidharan A. Advanced prognostic modeling with deep learning: assessing long-term outcomes in liver transplant recipients from deceased and living donors. J Transl Med. 2025;23:188.
40. Gangadhar A, Hasjim BJ, Zhao X, et al. Personalized survival benefit estimation from living donor liver transplantation with a novel machine learning method for confounding adjustment. J Hepatol. 2025;83:1116-27.
41. Giglio MC, Dolce P, Yilmaz S, et al.; European Liver and Intestine Transplant Association (ELITA). Development of a model to predict the risk of early graft failure after adult-to-adult living donor liver transplantation: an ELTR study. Liver Transpl. 2024;30:835-47.
42. Barriga-Rodríguez P, Falcón-Cubillo M, Mejías-Trueba M, et al. Pharmacokinetics of different tacrolimus formulations in the early post-liver transplant period: a scoping review. Pharmaceutics. 2025:17.
43. Brunet M, van Gelder T, Åsberg A, et al. Therapeutic drug monitoring of tacrolimus-personalized therapy: second consensus report. Ther Drug Monit. 2019;41:261-307.
44. Song X, Liu F, Gao H, et al. Compare the performance of multiple machine learning models in predicting tacrolimus concentration for infant patients with living donor liver transplantation. Pediatr Transplant. 2023;27:e14379.
45. Rodríguez-Perálvarez M, Colmenero J, González A, et al.; Chronic Immunosuppression. Cumulative exposure to tacrolimus and incidence of cancer after liver transplantation. Am J Transplant. 2022;22:1671-82.
46. Rodríguez-Perálvarez M, Tsochatzis E, Naveas MC, et al. Reduced exposure to calcineurin inhibitors early after liver transplantation prevents recurrence of hepatocellular carcinoma. J Hepatol. 2013;59:1193-9.
47. Abrahamsson J, Sternby Eilard M, Rizell M, Bennett W, Åberg F. Reduced calcineurin inhibitor exposure with antibody induction and recurrent hepatocellular carcinoma after liver transplantation. Scand J Gastroenterol. 2022;57:325-32.
48. Grigg SE, Sarri GL, Gow PJ, Yeomans ND. Systematic review with meta-analysis: sirolimus- or everolimus-based immunosuppression following liver transplantation for hepatocellular carcinoma. Aliment Pharmacol Ther. 2019;49:1260-73.
49. Mauro E, Sanduzzi-Zamparelli M, Jutras G, et al. Challenges in Liver Transplantation for Hepatocellular Carcinoma: A Review of Current Controversies. Cancers. 2024:16.
50. Ni X, Wang Q, Gu J, Lu L. Clinical and basic research progress on treg-induced immune tolerance in liver transplantation. Front Immunol. 2021;12:535012.
51. Appenzeller-Herzog C, Hartleif S, Vionnet J. Clinical parameters and biomarkers predicting spontaneous operational tolerance after liver transplantation: a scoping review. Am J Transplant. 2021;21:3312-23.
52. Tsilimigras DI, Mehta R, Moris D, et al. Utilizing machine learning for pre- and postoperative assessment of patients undergoing resection for BCLC-0, A and B Hepatocellular carcinoma: implications for resection beyond the BCLC guidelines. Ann Surg Oncol. 2020;27:866-74.
53. Chaudhary K, Poirion OB, Lu L, Garmire LX. Deep learning-based multi-omics integration robustly predicts survival in liver cancer. Clin Cancer Res. 2018;24:1248-59.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].