Bioresorbable scaffolds in lower limb arterial disease: a narrative review of advancements and future directions
Abstract
Endovascular recanalization has revolutionized the treatment of peripheral artery disease (PAD), offering minimally invasive alternatives to open surgery. However, permanent metallic stents, while effective in restoring blood flow, are associated with long-term complications such as chronic inflammation, restenosis, and stent thrombosis. Bioresorbable scaffolds (BRS) have emerged as an innovative solution, providing temporary mechanical support while delivering antiproliferative drugs before fully degrading, thus eliminating the risks associated with permanent implants. Significant advancements in bioresorbable materials have led to the development of new-generation scaffolds with improved radial strength, controlled degradation rates, and enhanced drug-eluting properties. Several BRS platforms, including polymer- and magnesium-based designs, are undergoing clinical and preclinical evaluation for peripheral applications. Early trials suggest that BRS may offer comparable short-term patency rates to conventional stents while potentially reducing late adverse events. However, challenges such as scaffold thrombosis, mechanical integrity in large and calcified vessels, and long-term efficacy remain under investigation. This narrative review explores the evolution of BRS technology, the mechanisms of scaffold resorption, current clinical evidence, and future prospects for their use in peripheral endovascular interventions. As research progresses, BRS may represent a paradigm shift in the treatment of PAD, offering a balance between structural support and vascular restoration.
Keywords
INTRODUCTION
Peripheral artery disease (PAD) is a prevalent manifestation of systemic atherosclerosis and a major cause of morbidity and mortality worldwide, with a prevalence of 5.6% and affecting over 236 million individuals[1]. While awareness of arterial disease is gradually increasing among patients and healthcare professionals, underdiagnosis and undertreatment remain concerns[2,3]. As a result, delays in diagnosis and incomplete implementation of evidence-based therapies still occur, particularly in resource-limited settings[4].
PAD is defined by atherosclerosis-driven narrowing or obstruction of arteries, with the lower extremities being the most commonly affected sites. The resulting ischemia may manifest variably, from intermittent claudication to critical limb-threatening ischemia (CLTI) with ischemic rest pain, tissue loss, or gangrene[5].
Beyond its local vascular manifestations, PAD carries significant systemic cardiovascular implications. The presence of PAD is a powerful predictor of future cardiovascular events, with affected individuals showing a two- to sixfold increased risk of myocardial infarction (MI), stroke, and cardiovascular-linked death[6]. This heightened risk underscores the fact that PAD is not merely a localized disorder affecting the peripheral vasculature, but rather a clinical manifestation of systemic endothelial dysfunction and generalized atherosclerosis[7].
In addition to its systemic impact, PAD is associated with substantial limb-related morbidity and functional decline. Clinically, it leads to decreased mobility, loss of independence, and reduced quality of life, as claudication limits physical activity. Moreover, PAD significantly increases the risk of lower extremity amputation and associated mortality. Among patients undergoing major amputation, one-year mortality rates can exceed 20%, underscoring the critical need for early intervention[8].
From ankle-brachial index to chemical exchange saturation transfer: a multimodal approach to diagnosing and characterizing PAD
The diagnosis and characterization of PAD requires a structured, multimodal approach, combining clinical evaluation, functional testing, and vascular imaging. The ankle-brachial index (ABI) provides a simple, non-invasive initial assessment, while duplex ultrasound (DUS) allows simultaneous evaluation of arterial morphology and hemodynamics, guiding diagnosis, treatment planning, and follow-up. DUS is widely available and free of radiation, though its accuracy depends on operator expertise and patient-specific factors[9,10]. For more comprehensive anatomical assessment and procedural planning, computed tomography angiography (CTA) offers high-resolution, three-dimensional imaging, enabling visualization of stenoses, occlusions, aneurysms, and collateral vessels. While CTA provides detailed macrovessel information, it involves iodinated contrast and radiation exposure[11]. Traditional imaging methods primarily capture large-vessel anatomy, leaving microvascular and tissue-level abnormalities largely unassessed[12].
Advanced magnetic resonance imaging (MRI) techniques address this gap by providing detailed tissue-level information that complements conventional imaging. Arterial spin labeling (ASL) enables non-contrast quantification of tissue perfusion, Blood Oxygen Level Dependent (BOLD) imaging evaluates muscle oxygenation[13], and Chemical Exchange Saturation Transfer (CEST) imaging detects metabolites such as creatine, reflecting muscle metabolic status[14]. These methods can identify early microvascular impairment, monitor therapeutic response, and provide mechanistic insights into PAD pathophysiology. Although primarily applied in research settings, they have the potential to enhance clinical decision-making, optimize patient stratification, and improve outcomes through earlier detection and more precise characterization of disease[15].
Conventional treatment options and their challenges
Over the past several decades, the management of advanced PAD has undergone a significant transition from traditional open surgical revascularization to an endovascular-first approach. This shift has been driven by technological advancements, improved perioperative outcomes, and accumulating evidence supporting the efficacy of minimally invasive techniques. Despite this trend, surgical bypass remains the gold standard for patients with symptomatic complex multilevel or infrapopliteal disease, particularly in those with a longer life expectancy and suitable autologous conduit[16,17].
However, endovascular-first strategies are preferred in patients with high surgical risk or limited conduit availability, and the endovascular approach is increasingly employed for aortoiliac, femoropopliteal, and below-the-knee (BTK) lesions due to lower procedural morbidity, shorter hospital stay, and faster recovery[18].
The therapeutic armamentarium includes percutaneous transluminal angioplasty (PTA), bare-metal stents (BMS), drug-eluting stents (DES), drug-coated balloons (DCBs), and atherectomy devices.
PTA is frequently the first-line option but can be complicated by restenosis, dissection, and recoil[19]. Despite its simplicity, PTA offers limited durability, with reported 12-month primary patency rates of 40%-60%, which decline substantially over time, especially in long or calcified lesions[20]. To improve early lumen gain and patency, BMS can be deployed, achieving approximately 60%-75% patency at one year[21]. However, permanent metallic implants can provoke chronic inflammation, increasing the risk of in-stent restenosis (ISR)[22,23] and, rarely, thrombosis, particularly when antiplatelet therapy is interrupted[24]. DES were developed to mitigate neointimal hyperplasia through localized drug delivery, though concerns regarding delayed endothelial healing and long-term safety persist.
DCBs offer an alternative by delivering antiproliferative agents, such as paclitaxel, during balloon inflation without leaving a permanent scaffold[25]. They have demonstrated favorable early outcomes, particularly in femoropopliteal arteries, with 12-month primary patency typically ranging from 65%-85%[26]. However, their efficacy is reduced in BTK and heavily calcified lesions due to limited drug uptake and mechanical constraints[27].
Despite these limitations, DCBs remain a valuable revascularization option for patients who are unsuitable for stent implantation[28]. However, potential drawbacks include inflammatory reactions, delayed endothelial recovery, and heterogeneous drug distribution, particularly in complex or calcified lesions[27]. Continuous technological innovations have sought to address these challenges by optimizing drug delivery and balloon performance. Enhanced drug formulations and excipient carriers - such as urea, iopromide, and shellac - have demonstrated improved drug transfer and retention even in fibrotic, calcified, or long-segment lesions, while advances in coating uniformity enhance homogeneous drug distribution along the treated vessel[29]. Moreover, semi-compliant and ultra-compliant balloon designs facilitate improved vessel-wall apposition and better adaptation to irregular luminal geometry, thereby promoting localized drug absorption[30]. Refinement of inflation protocols, in combination with lesion-preparation techniques such as scoring or cutting balloons and the introduction of microstructured balloon surfaces, further augments drug penetration and acute luminal gain[31-33].
Definition and concept of bioresorbable scaffolds as an alternative to permanent stents
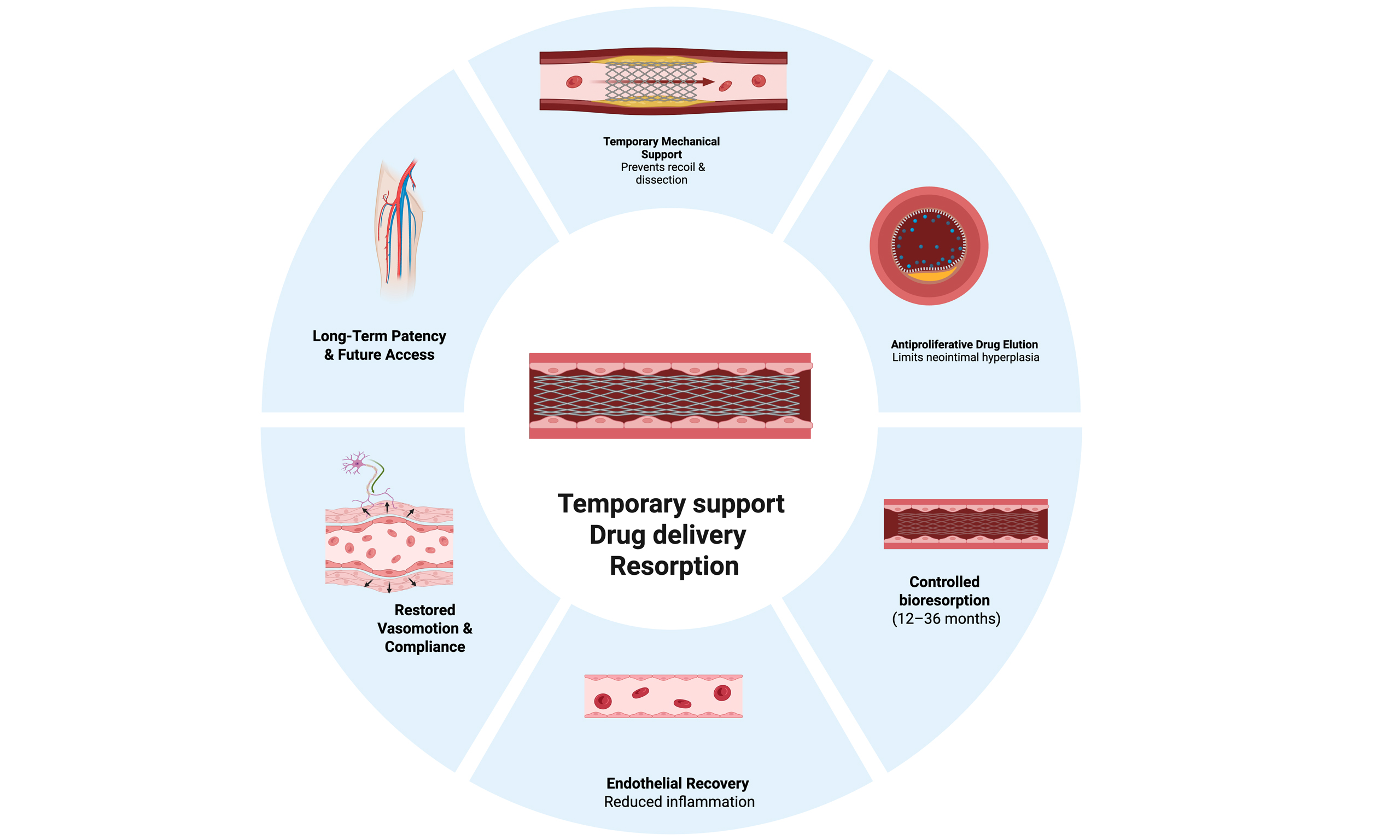
Bioresorbable scaffolds (BRS) represent an innovative class of vascular devices developed to address the long-standing limitations of permanent metallic stents. Conceptually, BRS provide temporary mechanical support to the vessel during the initial healing phase post-intervention while delivering antiproliferative drugs to mitigate neointimal hyperplasia. Unlike metallic stents, which remain permanently in situ, BRS gradually degrade and are fully resorbed by the body, ideally within a few years of implantation[34].
The foundational principle behind BRS is to combine the anti-restenotic benefits of DES with the biological advantages of restoring vascular integrity and function after scaffold resorption. This attribute renders BRS particularly appealing for patients with longer life expectancy or complex lesions, where permanent vessel caging may carry long-term disadvantages, specifically chronic inflammation, impaired vessel compliance, late thrombosis, and limitations in future reinterventions[35].
As the scaffold dissolves, the vessel can gradually return to a more physiological state, promoting positive vessel remodeling characterized by adaptive restoration of vessel diameter, compliance, and wall architecture. This remodeling preserves luminal patency, enhances endothelial function, facilitates vasomotion, and reduces the risk of late restenosis[36]. Collectively, these effects support long-term vascular healing, functional recovery, and the re-establishment of normal hemodynamic responsiveness, positioning BRS as a promising alternative to permanent implants in appropriately selected patients.
Evolution of bioresorbable scaffolds technology
Early development of BRS
The concept of bioresorbable vascular scaffolds (BVS) emerged in the early 2000s with the primary objective of providing temporary vascular support while minimizing long-term complications associated with metallic implants. Initial attempts focused on polymeric materials due to their established safety profile and biodegradability. The Igaki-Tamai stent, developed in Japan and implanted in humans as early as 2000, is composed of poly-L-lactic acid (PLLA) and demonstrated that a fully bioresorbable structure can maintain vessel patency while degrading safely within the human body[37]. Despite its promise, the Igaki-Tamai stent lacked drug-eluting properties and sufficient radial strength, leading to high rates of recoil and restenosis[38]. These limitations spurred the development of next-generation BRS with drug-eluting capabilities to control neointimal hyperplasia and enhanced mechanical integrity to resist collapse in high-pressure coronary environments.
Advancements in BRS design and materials
The first commercially available BRS with drug-eluting properties was the ABSORBTM BVS (Abbott Vascular, Santa Clara, California), which gained widespread clinical use and became the prototype for future scaffolds. It consisted of a PLLA backbone with everolimus coating, designed to elute the drug over a few months
This prompted a shift toward refinement in scaffold architecture and material science. While first-generation devices had relatively thick struts (~150 µm) and slow degradation profiles, newer designs focused on thinner struts (< 100 µm) and faster, more controlled bioresorption.
To address the mechanical shortcomings of polymer-based scaffolds, researchers have focused on metallic bioresorbable materials, particularly magnesium alloys. Magnesium offers enhanced radial strength and is designed for accelerated degradation compared with PLLA-based scaffolds, though the full resorption timeline in clinical use remains under investigation.
Early clinical data from the Magmaris scaffold in the BIOSOLVE trials indicated favorable safety and low target-lesion failure rates at 12 months[41], highlighting the potential of metallic BRS platforms for future peripheral applications.
The ideal duration of BRS resorption remains a central design consideration. Complete degradation should occur after the vessel has achieved adequate remodeling and healing, generally between 12 and 36 months, to maintain luminal integrity while minimizing the risk of late ScT or chronic inflammation[42]. A resorption period shorter than 12 months may compromise structural support during neointimal maturation, whereas persistence beyond three years can perpetuate inflammatory responses and mechanical stress. Consequently, optimal resorption balances transient mechanical support with timely restoration of native vessel physiology.
More recently, composite scaffolds incorporating hybrid materials such as iron and zinc alloys or novel polymers have entered preclinical evaluation[43]. These designs aim to combine the biocompatibility and tunable degradation of polymers with the mechanical strength of metals.
The BRS currently available are summarized in Table 1, along with their main characteristics.
Bioresorbable scaffolds for lower-limb PAD - device characteristics
| Device | Material | Deployment | Strut thickness, mm | Drug | Drug dose | Device diameters, mm | Degradation time, months | Availability | |
| Metallic | |||||||||
| AMS biotronik[44] | Magnesium alloy | Balloon expandable | 165 | None | N/A | 3.00 | 4 | Evaluated mainly in preclinical and early clinical studies; not commercially available | |
| Magmaris biotronik[45] | Magnesium alloy | Balloon expandable | 166 | BIOlute resorbable PLLA eluting sirolimus | 1.4 mg/mm2 | 3.00; 3.50 | 24 | CE-marked and commercially available in Europe; not approved or marketed in the United States | |
| IBS titan, Biotyx medical[46] | Iron | Balloon expandable | 53 | PDLLA + sirolimus | 6.1 mg/mm | 3.00 | > 24 | Undergoing clinical evaluation in Europe; not yet commercially available | |
| Polymeric | |||||||||
| MeRes meril[47] | PLLA | Balloon expandable | 100 | PDLLA + sirolimus | 1.25 mg/mm2 | 2.50; 3.00; 3.50; 4.00; 4.50 | 24-36 | CE-marked and available in several European markets | |
| Absorb abbott[48] | PLLA | Balloon expandable | 157 | Everolimus | 100 mg/cm2 | 2.50; 3.00; 3.50 | 36 | Previously CE-marked but withdrawn from commercial sale; currently limited to research and registry use | |
| Esprit-BTK abbott[49] | PLLA | Balloon expandable | 99-120 | Everolimus | 100 mg/cm2 | 2.50; 3.00; 3.50; 3.75 | 36 | Recently CE-marked and FDA-approved; entering early commercial rollout in Europe and the United States | |
| Motiv Reva Medical[50] | Tyrocore | Balloon expandable | 95-115 | Sirolimus | 1.97 mg/mm | 2.50; 3.00; 3.50; 4.00 | 48 | CE-marked for below-the-knee use; available in select European centers, investigational in the U.S. | |
| MAGNITUDE R3 Vascular[51] | PLLA | Balloon expandable | 98 | PDLLA, sirolimus | 96 mg/cm2 | 3.00; 3.50 | N/A | Investigational device currently in pivotal clinical trials; not yet commercially available | |
Key material characteristics for BRS success
The evolution of BRS has revolved around fine-tuning three critical material characteristics:
1. Radial Strength: Radial strength is vital for maintaining vessel patency post-implantation, particularly in heavily calcified lesions. Magnesium-based scaffolds provide superior radial support compared with first-generation polymer scaffolds and approach the performance of some BMS, although balancing strength with flexibility and resorption kinetics remains a core challenge[52].
2. Controlled Degradation Kinetics: Ideal BRS materials should degrade in concert with vessel healing, generally over a period ranging from several months to a few years, rather than a fixed 12 months. Fast-degrading scaffolds risk mechanical collapse before neointimal maturation, while slow-degrading scaffolds may provoke prolonged inflammatory responses. Controlled hydrolysis in polymers and corrosion control in metals (via alloying or coating) are strategies used to optimize degradation kinetics[52,53].
3. Drug-Eluting Capabilities: Drug-eluting features are now standard in BRS to prevent early restenosis. Drugs such as everolimus and sirolimus are loaded onto the scaffold surface using biodegradable polymers. Advanced designs are exploring multi-layer drug release systems and directional drug loading to enhance local efficacy while minimizing systemic exposure.
As the field advances, a growing body of literature highlights the importance of combining material science with procedural best practices, such as lesion pre-dilation, proper vessel sizing, and high-pressure post-dilatation, to optimize outcomes. These insights have led to the so-called “4P” strategy: Patient selection, Proper sizing, Pre-dilatation, and Post-dilatation, now considered essential for the safe and effective deployment of BRS[54].
Potential advantages of BRS in addressing the complications of metallic stents
Metallic stents, while transformative in the treatment of lower limb artery disease, are not without significant limitations. Their permanent presence within the vessel wall has been associated with a number of long-term complications. Among the most clinically significant are late and very late stent thrombosis, chronic vessel inflammation, impaired vasomotion, neoatherosclerosis, and ISR[55]. These complications arise partly from the continuous interaction between the metallic implant and the vascular tissue, which can trigger pro-inflammatory and pro-thrombotic responses over time[56]. BRS were developed to directly address these limitations by offering a temporary scaffold that supports the vessel only during the vulnerable healing period before being completely resorbed. This “leave nothing behind” strategy has several advantages:
1. Reduction in Late Stent Thrombosis: BRS showed the potential to reduce the risk of late and very late ScT[54]. In permanent stents, thrombosis can occur months or even years after implantation due to persistent metal struts interfering with endothelial healing[57]. In contrast, as BRS degrade, the pro-thrombotic surface area decreases, potentially lowering the risk of thrombotic events over the long term. Clinical studies such as ABSORB III demonstrated a decline in cardiac events after complete scaffold resorption, supporting this hypothesis, although early ScT remains a concern with first-generation devices[40]. Permanent metallic stents can induce long-term impairment of vascular physiology by constraining the arterial wall and eliminating its natural vasomotor activity[58]. This persistent mechanical fixation may result in endothelial dysfunction, disturbed shear stress patterns, and adverse vascular remodeling, ultimately predisposing to late neoatherosclerosis and very late thrombotic events[59]. In contrast, BRS progressively degrades, enabling the restoration of normal vessel compliance and vasomotion[60,61].
2. Reduction of Chronic Inflammatory Response: Permanent metallic implants are associated with a sustained inflammatory response from the vessel wall. The chronic presence of metal may lead to late neoatherosclerosis, contributing to ISR and thrombotic events. Because BRS are eventually absorbed, they may mitigate chronic inflammation and reduce the risk of neoatherosclerotic plaque formation, especially in long-term follow-up[62].
3. Improved Imaging and Surgical Access Post-Resorption: Another significant advantage of BRS is their non-interference with future interventions. After complete resorption, BRS no longer obscure imaging modalities such as computed tomography (CT) or MRI, nor do they interfere with future surgical procedures, including surgical revision, on the treated limb. This feature is particularly relevant for younger patients or those with progressive vascular disease who may require future interventions.
4. Potential for Positive Remodeling: As the scaffold resorbs, BRS may promote positive vessel remodeling by supporting natural healing processes and avoiding permanent mechanical injury to the vessel wall[63]. Animal models and intravascular imaging studies have demonstrated favorable vessel remodeling with increased lumen area and plaque stabilization following BRS implantation[64-67].
Preclinical studies and early evaluations
Before large-scale human trials, extensive preclinical investigations were conducted to characterize scaffold-vessel interactions, degradation kinetics, and biological responses.
Polymer scaffolds in animal models
In rabbit iliac artery models, PLLA scaffolds demonstrated 100% procedural success with preservation of vessel diameter (+1.2% acute gain), controlled degradation over 24 months, and minimal inflammatory infiltrate at 6 and 12 months post-implantation[68]. Histologic evaluation revealed near-complete endothelial coverage by 3 months and no neointimal hyperplasia exceeding 0.5 mm at 1 year.
Magnesium scaffolds in porcine models
Findings from smaller animal studies were corroborated in larger porcine models. In coronary and peripheral arteries, magnesium-based scaffolds degraded predictably, with approximately 70% mass loss at 6 months and near-complete resorption by 12 months[69]. Vessel histology at 6 months demonstrated a thin neointimal layer (mean thickness 0.35 ± 0.08 mm) and absence of chronic inflammation. In a porcine femoropopliteal model, magnesium scaffolds maintained full patency (100% at 6 months) without aneurysm formation or late recoil[70].
Emerging composite designs
More recently, research has focused on hybrid scaffolds that combine biodegradable polymers with magnesium or iron alloys to achieve tailored degradation (6-18 months) and improved radial strength
Encouraged by these preclinical outcomes, early-phase clinical trials have been initiated to assess the safety, vascular healing, and patency of BRS in both coronary and peripheral territories.
CLINICAL EVIDENCE AND CURRENT APPLICATIONS
Clinical evaluation of BRS for infrapopliteal and BTK arterial disease has significantly evolved over the past decade, advancing from initial feasibility investigations to multicenter randomized clinical trials, and yielding an increasingly comprehensive understanding of their safety, efficacy, and overall performance in patients with CLT. The first human data were derived from early feasibility studies: the DISAPEAR trial (NCT02043795), as reported in preliminary publications, demonstrated that an everolimus-eluting PLLA scaffold (Esprit BTK could be safely deployed in small tibial vessels, achieving high procedural success
More definitive evidence emerged through the pivotaL Investigation of saFety and Efficacy of BRS Treatment-Below The Knee (LIFE-BTK) randomized controlled trial (RCT), the first large-scale RCT dedicated to BTK BRS evaluation. In this 261-patient trial (2:1 randomization to Esprit BTK vs. PTA), the scaffold arm achieved superior 2-year primary efficacy (~61.5% vs. 32.8%), significantly lower binary restenosis (≈28%-35% vs. 48%-58%), and improved freedom from clinically driven target-lesion revascularization (TLR), with benefits consistent across high-risk subgroups including diabetics and patients with advanced tissue loss[74]. Importantly, ScT-previously a concern in first-generation coronary BRS - was low (< 1%-2%), reflecting the thinner struts, enhanced radial strength, and improved implantation protocols of the Esprit BTK platform.
Parallel to PLLA-based devices, next-generation BTK-specific BRS technologies have emerged. The Randomized Evaluation of Sirolimus-Eluting Bioresorbable Vascular Scaffold in Below-The-Knee Arteries Study I (RESOLV I), as reported in preliminary publications, evaluated the MAGNITUDE® sirolimus-eluting composite polymer scaffold, engineered specifically for infrapopliteal biomechanics with thin struts and optimized radial strength. In ~30 patients, 6-month primary patency reached 75%-80%, with favorable wound-healing trajectories, providing justification for the ongoing pivotal randomized trial started in 2025[51] Evaluation of a Limus-Eluting bioresorbable scaffold in Below-The-Knee arteries (ELITE-BTK). Similarly, the Multi-center Ongoing Trial Investigating the sirolimus-eluting bioresorbable Vascular scaffold in Below-The-Knee arteries, the Multi-center Ongoing Trial Investigating the sirolimus-eluting bioresorbable Vascular scaffold in Below-The-Knee arteries (MOTIV BTK) program, evaluating a Tyrocore® sirolimus-eluting scaffold, has produced some of the most compelling long-term registry data in this space: in 58 patients (60 limbs), technical success approached 99%, with primary patency of 88.3% at 12 months, 81.7% at 24 months, and ~80% at 36 months. Limb-salvage rates (~95%) and freedom from clinically driven reintervention (~93% at 3 years) further emphasize the potential durability of drug-eluting BRS in BTK arteries[50].
Despite the expanding evidence base, several limitations require cautious interpretation. Most clinical experience remains derived from single-arm feasibility cohorts with small sample sizes, heterogeneity in lesion morphology, and variability in procedural technique, vessel preparation and follow-up imaging. Patency estimates in early trials often rely on Kaplan-Meier projections. Device heterogeneity also complicates comparisons: PLLA scaffolds (Esprit BTK, ABSORB) feature slower resorption and thicker struts, whereas newer composite or magnesium-based devices (MAGNITUDE, MOTIV) integrate higher radial strength and more predictable degradation, raising the possibility of heterogeneous performance among different scaffold platforms. Additionally, BTK outcomes are highly influenced by distal runoff, wound infection, and wound management - overlooked variables in early studies. To date, LIFE-BTK remains the only completed large RCT in this field, highlighting the need for long-term evaluation and additional randomized trials such as ELITE-BTK to define the class-wide safety, efficacy, and optimal role of BRS in CLTI[75] [Figure 1].
Figure 1. Bioresorbable Scaffolds vs. Metallic Stents. BRS: Bioresorbable scaffold; DAPT: dual antiplatelet therapy; ISR: in-stent restenosis.
Together, these findings highlight the potential of BRS as viable alternatives to permanent metallic stents in selected patients with BTK disease. Their ability to maintain vessel patency, avoid long-term complications, and support natural vascular healing offers a compelling therapeutic strategy in the evolving landscape of peripheral endovascular treatment [Table 2].
Overview of major clinical trials investigating bioresorbable scaffold technologies in below-the-knee peripheral artery disease
| Trial name | Vascular territory & population | Device (scaffold type) | Trial phase/status | Key result or milestone | Clinical trial reference |
| DISAPEAR (Below-the-Knee; CLTI)[72,76] | Infrapopliteal arteries; CLTI | Everolimus-eluting PLLA scaffold (Esprit BTK) | First-in-human feasibility (single-arm) | Technical success; ~12-mo patency ~85% in small cohort | NCT02043795 https://clinicaltrials.gov/study/NCT02043795 |
| LIFE-BTK[74,77] | Infrapopliteal arteries (BTK); CLTI | Everolimus-eluting resorbable scaffold (Esprit BTK) | Randomized pivotal RCT vs. PTA (n≈261) | At 2 yrs: restenosis 28.5% vs. 48.2% PTA; improved limb salvage/patency | NCT04227899 |
| RESOLV I/First-in-Human (R3 vascular)[70,78] | BTK/infrapopliteal arteries | MAGNITUDE sirolimus-eluting bioresorbable scaffold | Feasibility FIH (~30 pts) | 6-mo patency ~75%-80% early cohort | NCT04912323 |
| ELITE-BTK (R3 vascular)[75] | Below-the-knee PAD | Next-gen MAGNITUDE sirolimus-eluting scaffold | Pivotal RCT (~276 pts planned) | Enrollment initiated; results pending | NCT06071429 |
| MOTIV BTK[50,79] | Below-the-knee arteries; CLTI (~292 pts) | Sirolimus-eluting bioresorbable scaffold (Tyrocore) | Pilot/registry + RCT ongoing | Early registry: acceptable safety/patency; RCT pending | NCT05406622 |
| ABSORB BTK-pilot[73,80] | Below-the-knee arteries | Everolimus-eluting PLLA scaffold (Absorb BVS) | Single-arm pilot/registry | 12-mo patency ~96%; 24-mo ~84.6% (n≈38 limbs) | NCT02793349 |
Patient selection and decision-making considerations
Careful patient selection is essential to achieving optimal clinical outcomes and reducing procedural risks with evolving BRS technology. Ideal PAD candidates are typically affected by short- to intermediate-length lesions, favorable vessel diameters (≥ 2.5 mm), and low-to-moderate calcification - anatomic profiles that align with the current mechanical constraints and radial strength of contemporary BRS platforms[81]. Younger patients or those with extended life expectancy may achieve the greatest long-term benefit, as resorbable systems eliminate permanent foreign-body burden and may reduce late complications, including stent fracture and ISR. On the other hand, patients with advanced renal insufficiency, poorly controlled diabetes, or diffuse, heavily calcified disease remain at higher risk for scaffold failure, thrombosis, and incomplete resorption. Multimodal imaging, including DUS, CTA and intravascular ultrasound (IVUS), remains critical for precise lesion characterization, patient stratification, and procedural planning[82].
Scaffold design has undergone transformative improvements to overcome the limitations of first-generation devices. PLLA BRS initially required strut thicknesses of 150-160 µm to ensure radial support, but this bulk disrupted laminar flow and delayed endothelialization. Second-generation platforms such as MeRes 100, DESolve 100, and Firesorb have reduced strut thickness to ≤ 100 µm without compromising mechanical integrity[83]. The MeRes 100 scaffold, with 100 µm struts and a hybrid closed-open cell design, demonstrated zero ScT and a 1.61% Major Adverse Cardiovascular Events (MACE) rate at two years in the MeRes-1 Extend study (n = 240), with reduced luminal protrusion enhancing safety[84]. DESolve 100 exhibits rapid self-correction of malapposition, while the Firesorb scaffold in the FUTURE-II trial (n = 433) delivered late loss and strut coverage metrics comparable to metallic DES, confirming the potential of thin-strut platforms to balance efficacy with vascular healing[83-85].
Computational fluid dynamics further highlight the advantages of ultra-thin struts, predicting up to a 25% reduction in low-shear, high-recirculation zones - key loci for platelet activation and ScT - when strut thickness is reduced by one-third[86]. Modern BRS integrate radiopaque markers (platinum or tantalum micro-rings), enhancing deployment precision to sub-millimeter accuracy. Next-generation devices, such as Abbott’s Esprit BTK (< 100 µm struts with dual radiopaque bands), are under investigation in PAD (NCT04227899), with preliminary results reporting 91% acute success in complex peripheral lesions[77].
Matching scaffold degradation to vessel healing remains crucial. First-generation PLLA scaffolds degraded slowly over 3-4 years, prolonging thrombogenic exposure. Newer materials, including magnesium alloys (DREAMS 3G) and advanced polymers (Tyrocore in FANTOM II), achieve complete resorption within 12-18 months[52]. DREAMS 3G, composed of Mg-Zr-Mn alloy coated with poly-D,L-lactic acid (PDLLA) (BIOlute), maintains > 90% radial strength for six months and degrades fully by 12-15 months in preclinical models, inducing minimal inflammation and a neointimal thickness of 0.35 ± 0.08 mm. FANTOM II degrades fully within one year and restores vasomotor function by nine months, key indicators of vascular restoration without late lumen loss[87].
Optimal implantation technique is equally critical, particularly in the mechanically demanding peripheral vasculature. The PSP (Pre-dilatation, Sizing, Post-dilatation) protocol [Figure 1] reduces acute malapposition from 12% to 3% and ScT from 2.8% to 1.1% at 12 months[60]. Optical coherence tomography (OCT)-guided studies show post-dilatation pressures > 16 atm safely increase scaffold expansion by 15% without inducing strut fractures, critical in peripheral arteries with large diameters (4-6 mm) and heavy calcification. Early PLLA BRS, constrained by a low elastic modulus (66-fold lower than cobalt-chromium), required thick struts and exhibited recoil > 20%, driving reintervention rates up to 15%-20% at one year[81]. Magnesium-based scaffolds, while offering higher radial force, may lose support within 3-6 months due to rapid in vivo degradation[52].
Integration of refined scaffold engineering, patient-specific imaging, and procedural optimization has expanded the clinical role of BRS in PAD. Though long-term peripheral data remain limited, early results for second-generation platforms are promising. Personalized, anatomy-driven selection and precise implantation remain cornerstones of safe, effective BRS use[88,89].
Technological innovation has addressed early limitations such as recoil, structural compromise, and inconsistent resorption by focusing on material composition, scaffold architecture, and controlled degradation kinetics[61]. Alloying and polymer overcoating have been pivotal in engineering magnesium-based scaffolds that maintain mechanical integrity during the healing window while promoting rapid endothelialization[90,91]. Pure magnesium, though biocompatible, degrades too rapidly, often losing radial strength within 3-6 months[52]. Mg-Zr-Mn alloys enhance strength via zirconium-induced grain refinement and stabilize corrosion through manganese incorporation. The DREAMS 3G scaffold’s BIOmag alloy exceeds 180 kPa radial strength - ~20% higher than first-generation Magmaris - while the BIOlute coating preserves > 90% diameter for six months in porcine coronary and peripheral models[92].
Synchrotron microtomography shows that these coatings form uniform barriers guiding residual magnesium conversion into hydrated magnesium oxide and amorphous calcium phosphate, achieving full resorption by 12 months. Polymer coatings such as poly(lactic-co-glycolic acid) (PLGA) or PDLLA (5-10 µm) optimize drug elution; in porcine arteriovenous (AV) shunt models, coated Mg-Zr-Mn scaffolds released 80% of sirolimus or paclitaxel in 90 days while maintaining 95% radial strength, compared with 65% in uncoated devices[93]. Histology confirms minimal inflammation at 6 and 12 months.
Scaffold geometry advancements also improve BRS performance in calcified, tortuous peripheral vessels[92]. Second-generation scaffolds employ ultra-thin struts (< 100 µm) in hexagonal or zig-zag frameworks, distributing radial load evenly. Bench fatigue simulations (10 million cycles) show a 30% reduction in stress concentration points versus older ring-and-link geometries, correlating with a 40% lower strut fracture rate[92]. Clinical trials-including MeRes 100, DESolve 100, and Firesorb - demonstrate preserved radial support, enhanced endothelial recovery, and reduced malapposition (MeRes-1 Extend and FUTURE-II)[83-85]. Computational fluid dynamics (CFD) predicts that thinner struts reduce low-shear zones by up to 25%, lowering thrombosis risk[86].
Hybrid polymer-metal scaffolds are emerging, combining high-strength metallic cores with degradable polymer sheaths for early radial support and controlled resorption. Iron-PLLA composites in preclinical femoropopliteal studies show < 4% acute recoil at six months versus > 20% for PLLA-only devices, with late lumen loss of 0.35 ± 0.08 mm at 12 months, comparable to modern DES[94].
Collectively, advances in alloys, polymer coatings, scaffold architecture, and composites bring BRS closer to clinical viability in PAD. Mg-Zr-Mn alloys coated with PDLLA/PLGA provide tunable degradation, preserving patency during healing and minimizing late thrombogenicity. Ultra-thin struts and radiopaque markers enhance procedural precision, and hybrid platforms bridge early radial support with long-term bioresorption. Trials such as BIOSOLVE-IV (Clinical trial reference: NCT02817802) and DREAMS 3G (Clinical trial reference: NCT05540223) suggest sustained patency, reduced thrombosis, and restored vascular function even in challenging peripheral lesions.
Despite these improvements, ScT remains a concern, highlighting biological determinants of thrombus formation from coronary to peripheral circulation[81]. Peripheral trials report low absolute event rates, yet thrombosis during scaffold degradation is an important safety consideration[95]. First-generation polymeric scaffolds in ABSORB BTK-pilot and DISAPEAR feasibility studies showed low ScT through 12 months, though small sample sizes limit assessment of late events[72]. Peripheral arteries, especially infrapopliteal vessels, experience mechanical stress, torsion, compression, and elongation during ambulation[96], which can induce transient malapposition or localized flow separation, promoting platelet activation and thrombus formation[97].
Factors contributing to peripheral BRS thrombogenicity include delayed endothelialization and prolonged exposure of polymer/composite struts during degradation[41]. Strut thickness > 100-120 µm correlates with delayed endothelial coverage beyond three months[98,99]. Degradation by-products (lactic acid, magnesium salts) can modulate local pH and endothelial function, influencing thrombotic risk[100]. Peripheral hemodynamics - variable shear, slow flow, calcification - may amplify these effects[101], though peripheral-specific preclinical data remain sparse[102].
Pharmacologically, dual antiplatelet therapy (DAPT) remains the cornerstone post-BRS implantation. Six- to twelve-month regimens cover incomplete endothelial healing and polymer resorption[103]. Coronary BRS experience supports this duration, with first-year DAPT strongly linked to reduced ScT and MI; extending DAPT beyond year one shows no clear benefit[104]. Shorter DAPT may be considered for high bleeding risk if imaging confirms endothelial coverage[105]. P2Y12 inhibitor selection should consider metabolic variability and thrombotic risk, with potent agents (ticagrelor, prasugrel) preferred in select patients[105].
Economic considerations
Beyond clinical and technological considerations, the economic implications of BRS therapy remain a pivotal factor influencing its adoption in routine vascular practice. Current-generation BRS frequently incur a substantial premium over standard metallic DES, reflecting complex manufacturing, higher material costs, and the need for advanced imaging and precise lesion preparation. In several European centers, BVS pricing has been reported to be multiple times higher than that of DES[106].
Economic subanalyses from coronary programs provide additional insight into procedural cost dynamics. In the ABSORB III economic study, mean procedural expenditure was USD 6,316 ± 1,892 for Absorb BVS versus USD 6,103 ± 1,895 for everolimus-eluting stents (EES), representing a modest yet significant increase of approximately USD 213 per case, largely attributable to scaffold price and device multiplicity[107].
Long-term follow-up from ABSORB Japan reported no significant differences in patient- or device-oriented adverse events between Absorb BVS and XIENCE EES over five years; however, robust peer-reviewed cost-effectiveness data for this population remain limited[108]. In the European context, analyses suggest that BRS implantation may incur additional procedural costs relative to DES, owing to more complex deployment and longer procedure duration, although multicenter, peer-reviewed cost-effectiveness data are scarce[109].
In peripheral revascularization, early modeling from the LIFE-BTK trial estimated two-year total patient costs of approximately USD 31,763 for scaffold-based therapy versus USD 30,995 for standard balloon angioplasty, corresponding to an incremental increase of ~USD 768 per patient[110].
When clinical efficacy outcomes were considered, the incremental cost-effectiveness ratio (ICER) was USD 3,206 per avoided primary endpoint and USD 6,068 per clinically driven TLR, well below conventional cost-effectiveness thresholds {< USD 50,000 per quality-adjusted life-year [QALY]}[110].
Magnesium-based BRS, such as Magmaris, have shown promising clinical outcomes with high patency and low reintervention rates; however, formal cost-effectiveness data are limited, and ICER estimates in the range of USD 4,000-8,000 per major adverse limb event avoided remain unverified in peer-reviewed studies[111].
Additional procedural costs arise from increased use of intravascular imaging (IVUS or OCT), more extensive lesion preparation, and the frequent need for multiple overlapping scaffolds in complex or multilevel lesions[112]. These complexities are not always fully reimbursed in many healthcare systems, and declining reimbursement for imaging and stenting in lower-extremity interventions may further limit adoption[113].
Although preliminary findings are encouraging, definitive cost-effectiveness validation will require long-term follow-up and real-world health-economic registries covering diverse CLTI populations, reimbursement environments, and procedural practices. Broader policy analyses suggest that, without demonstrated downstream cost savings, such as reduced reinterventions, amputations, or hospitalizations, the adoption of BRS in peripheral PAD will likely remain confined to specialized centers with dedicated funding or research support[114].
FUTURE PROSPECTS FOR BRS IN PAD TREATMENT
Technological innovations on the horizon
Advances in polymer chemistry have yielded next-generation materials, including bioresorbable polycarbonate urethanes and poly(trimethylene carbonate), that offer finely tunable hydrolysis windows (9-24 months), tensile strengths exceeding 60 MPa, and elongation at break greater than 300%. These properties enable thinner struts without sacrificing support[60]. Layered nanocoatings capable of releasing nitric oxide or vascular endothelial growth factor (VEGF) are under development to accelerate endothelialization and inhibit thrombosis, while “smart” scaffolds incorporating pH- or thermoresponsive linkers promise to adapt degradation rates to local inflammatory states[115].
Integration with other therapeutic modalities
Multimodal strategies are being explored to further enhance vessel healing. Scaffold surfaces tethered with endothelial nitric oxide synthase (eNOS) or VEGF plasmids aim to promote angiogenesis directly. Porous scaffold architectures enable seeding of endothelial progenitor cells, which in diabetic PAD animal models have significantly increased microvascular density and reduced restenosis[116]. Finally, dual-drug elution platforms that simultaneously release sirolimus and paclitaxel are in preclinical evaluation to target both smooth muscle proliferation and inflammation within the scaffold milieu[116].
Personalized approaches in PAD treatment
Maximizing the benefit-risk profile of BRS in PAD hinges on patient- and lesion-specific customization. First, patient selection should use algorithmic risk stratification that incorporates trans-Atlantic inter-society consensus (TASC) II lesion classification (A-D), reference vessel diameter, lesion length, and patient comorbidities such as diabetes mellitus, chronic kidney disease, and smoking status. For example, small-vessel chronic total occlusions (CTO) in diabetic patients with poor runoff may warrant cautious consideration of slower-degrading magnesium scaffolds to prolong luminal support[117].
Second, imaging-guided deployment using IVUS or optical coherence OCT has increasingly been employed. These high-resolution modalities enable more accurate vessel sizing and assessment of calcified nodules that may predispose to malapposition[118,119]. Although data on their impact in BRS deployment remain modest, their use may help optimize scaffold-to-vessel matching and reduce deployment-related complications.
Finally, computational modelling, specifically finite element analysis (FEA), may allow pre-procedural simulation of scaffold deployment in patient-specific arterial geometries[120].
By predicting stress distributions and areas of potential strut deformation under limb flexion, FEA could inform optimal scaffold design selection and positioning to minimize recoil and fracture risk[121].
Limitations
Despite encouraging early and mid-term results, current evidence supporting BRS use in PAD remains limited. Most data originate from feasibility studies, single-arm cohorts, or moderately sized randomized trials with selective inclusion criteria and relatively short follow-up. Long-term durability, late bioresorption behavior, and real-world outcomes, particularly in complex tibial lesions, require further investigation. Differences in lesion morphology, vessel biomechanics, and procedural practices across studies also limit comparability and generalization of results.
The unique mechanical forces exerted on infrapopliteal vessels, including torsion, compression, flexion, and elongation, pose challenges for scaffold integrity and may influence degradation kinetics, neointimal healing, and long-term patency. Additionally, the impact of comorbid conditions such as diabetes, infection, and impaired microcirculation on scaffold performance remains insufficiently characterized. Ongoing and upcoming pivotal trials evaluating next-generation sirolimus- and everolimus-eluting BRS will be essential to establish their long-term safety, efficacy, and optimal role within the broader framework of limb-saving revascularization strategies.
CONCLUSION
BRS represent a meaningful conceptual advance in PAD management by providing temporary mechanical support while avoiding the long-term drawbacks of permanent metallic implants. Contemporary evidence, particularly in infrapopliteal and BTK disease, indicates that newer-generation polymeric and magnesium-based platforms can achieve acceptable patency and safety when combined with appropriate patient selection and optimized implantation techniques.
Ongoing progress in material science, scaffold architecture, drug delivery, and imaging-guided deployment continues to address early limitations related to radial strength, thrombosis, and degradation control. Future research should focus on long-term durability, comparative effectiveness against established endovascular strategies, and validation in broader, real-world PAD populations. Well-designed randomized trials and longitudinal registries will be essential to define the precise clinical role of BRS within limb-preserving revascularization strategies.
DECLARATIONS
Acknowledgment
The Graphical Abstract was created with BioRender. Galassi L (2026) https://BioRender.com/o7r54p5.
Authors’ contributions
Conceptualization, methodology: Galassi L, Bartolozzi H
Validation: Verri MG, Verrengia A
Writing - original draft preparation: Galassi L, Verri MG, Bartolozzi H
Writing - review & editing: Galassi L, Bartolozzi H, Verri MG, Verrengia A, Facchinetti F, Ravini ML
All authors have read and approved the final version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Song P, Fang Z, Wang H, et al. Global and regional prevalence, burden, and risk factors for carotid atherosclerosis: a systematic review, meta-analysis, and modelling study. Lancet Glob Health. 2020;8:e721-9.
2. Lovell M, Harris K, Forbes T, et al.; Peripheral Arterial Disease Coalition. Peripheral arterial disease: lack of awareness in Canada. Can J Cardiol. 2009;25:39-45.
3. Criqui MH, Matsushita K, Aboyans V, et al.; American Heart Association Council on Epidemiology and Prevention; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; Council on Lifestyle and Cardiometabolic Health; Council on Peripheral Vascular Disease; and Stroke Council. Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the American Heart Association. Circulation. 2021;144:e171-91.
4. Johnston LE, Stewart BT, Yangni-Angate H, et al. Peripheral arterial disease in Sub-Saharan Africa: a review. JAMA Surg. 2016;151:564-72.
6. Salameh MJ, Rundek T, Boden-Albala B, et al. Self-reported peripheral arterial disease predicts future vascular events in a community-based cohort. J Gen Intern Med. 2008;23:1423-8.
7. Pérez Mejias EL, Faxas SM, Taveras NT, et al. Peripheral artery disease as a risk factor for myocardial infarction. Cureus. 2021;13:e15655.
8. McDermott MM. Functional impairment in peripheral artery disease and how to improve it in 2013. Curr Cardiol Rep. 2013;15:347.
9. Normahani P, Mustafa C, Shalhoub J, et al. A systematic review and meta-analysis of the diagnostic accuracy of point-of-care tests used to establish the presence of peripheral arterial disease in people with diabetes. J Vasc Surg. 2021;73:1811-20.
11. Hwang JH, Hwang JH, Lee SY, Lee J. Prostatic abscess caused by klebsiella pneumoniae: a 6-year single-center study. J Clin Med. 2022;11.
12. Erzinger FL, Polimanti AC, Pinto DM, et al. Brazilian Society of Angiology and Vascular Surgery guidelines on peripheral artery disease. J Vasc Bras. 2024;23:e20230059.
13. Törngren K, Eriksson S, Arvidsson J, et al. A reperfusion BOLD-MRI Tissue perfusion protocol reliably differentiate patients with peripheral arterial occlusive disease from healthy controls. J Clin Med. 2021;10:3643.
14. Xu J, Chung JJ, Jin T. Chemical exchange saturation transfer imaging of creatine, phosphocreatine, and protein arginine residue in tissues. NMR Biomed. 2023;36:e4671.
15. Sporkin HL, Patel TR, Betz Y, et al. Chemical exchange saturation transfer magnetic resonance imaging identifies abnormal calf muscle-specific energetics in peripheral artery disease. Circ Cardiovasc Imaging. 2022;15:e013869.
16. Conte MS, Pomposelli FB, Clair DG, et al.; Society for Vascular Surgery Lower Extremity Guidelines Writing Group, Society for Vascular Surgery. Society for Vascular Surgery practice guidelines for atherosclerotic occlusive disease of the lower extremities: management of asymptomatic disease and claudication. J Vasc Surg. 2015;61 Suppl:2S-41S.
17. Kim TI, Zhang Y, Cardella JA, Guzman RJ, Ochoa Chaar CI. Outcomes of bypass and endovascular interventions for advanced femoropopliteal disease in patients with premature peripheral artery disease. J Vasc Surg. 2021;74:1968-1977.e3.
18. Zhou Y, Zhang Z, Lin S, et al. Comparative efficacy and safety of endovascular treatment modalities for femoropopliteal artery lesions: a network meta-analysis of randomized controlled trials. Cardiovasc Intervent Radiol. 2020;43:204-14.
19. Brodmann M, Wissgott C, Holden A, et al. Treatment of infrapopliteal post-PTA dissection with tack implants: 12-month results from the TOBA-BTK study. Catheter Cardiovasc Interv. 2018;92:96-105.
20. Laird JR. Limitations of percutaneous transluminal angioplasty and stenting for the treatment of disease of the superficial femoral and popliteal arteries. J Endovasc Ther. 2006;13 Suppl 2:II30-40.
21. Meraj PM, Jauhar R, Singh A. Bare metal stents versus drug eluting stents: where do we stand in 2015? Curr Treat Options Cardiovasc Med. 2015;17:393.
22. Clare J, Ganly J, Bursill CA, Sumer H, Kingshott P, de Haan JB. The mechanisms of restenosis and relevance to next generation stent design. Biomolecules. 2022;12:430.
23. Dinc R. A review of the current state in neointimal hyperplasia development following endovascular intervention and minor emphasis on new horizons in immunotherapy. Transl Clin Pharmacol. 2023;31:191-201.
24. Lepor NE. Primary prevention, treatment, and secondary prevention of late and very late stent thrombosis. Rev Cardiovasc Med. 2007;8 Suppl 1:S27-33.
25. Liu J, Li T, Huang W, et al. Drug-coated balloons used in peripheral artery disease: experience from a single center. J Int Med Res. 2020;48:300060520940157.
26. Cui HJ, Wu YF. The efficacy of drug-coated balloons and drug-eluting stents in infrapopliteal revascularization: a meta-analysis. J Endovasc Ther. 2025;32:1799-820.
27. Tataru DA, Lazar FL, Onea HL, et al. Benefits and challenges of drug-coated balloons in peripheral artery disease: from molecular mechanisms to clinical practice. Int J Mol Sci. 2024;25:8749.
28. Ogata K, Nishihira K, Asano Y, et al. Clinical comparison of drug-coated balloon and drug-eluting stent for femoropopliteal lesions in chronic limb-threatening ischemia with wounds. Circ J. 2024;88:1647-55.
29. Chang GH, Azar DA, Lyle C, Chitalia VC, Shazly T, Kolachalama VB. Intrinsic coating morphology modulates acute drug transfer in drug-coated balloon therapy. Sci Rep. 2019;9:6839.
30. Stratakos E, Antonini L, Poletti G, et al. Investigating balloon-vessel contact pressure patterns in angioplasty: in silico insights for drug-coated balloons. Ann Biomed Eng. 2023;51:2908-22.
31. Bonaventura K, Schwefer M, Yusof AKM, et al. Systematic scoring balloon lesion preparation for drug-coated balloon angioplasty in clinical routine: results of the PASSWORD observational study. Adv Ther. 2020;37:2210-23.
32. Giannopoulos S, Ghanian S, Parikh SA, Secemsky EA, Schneider PA, Armstrong EJ. Safety and efficacy of drug-coated balloon angioplasty for the treatment of chronic limb-threatening ischemia: a systematic review and meta-analysis. J Endovasc Ther. 2020;27:647-57.
33. Zilinyi RS, Alsaloum M, Snyder DJ, et al. Surgical and endovascular therapies for below-the-knee peripheral arterial disease: a contemporary review. J Soc Cardiovasc Angiogr Interv. 2024;3:101268.
34. Iqbal J, Onuma Y, Ormiston J, Abizaid A, Waksman R, Serruys P. Bioresorbable scaffolds: rationale, current status, challenges, and future. Eur Heart J. 2014;35:765-76.
35. Kazuki Y, Kobayashi K, Hirabayashi M, et al. Humanized UGT2 and CYP3A transchromosomic rats for improved prediction of human drug metabolism. Proc Natl Acad Sci U S A. 2019;116:3072-81.
36. Jinnouchi H, Virmani R, Finn AV. Long-term vasomotion after absorb: fact or fiction? JACC Cardiovasc Interv. 2018;11:1572-5.
37. Shen Y, Yu X, Cui J, et al. Development of biodegradable polymeric stents for the treatment of cardiovascular diseases. Biomolecules. 2022;12:1245.
38. Hu T, Yang C, Lin S, Yu Q, Wang G. Biodegradable stents for coronary artery disease treatment: Recent advances and future perspectives. Mater Sci Eng C Mater Biol Appl. 2018;91:163-78.
39. Diletti R, Serruys PW, Farooq V, et al. ABSORB II randomized controlled trial: a clinical evaluation to compare the safety, efficacy, and performance of the Absorb everolimus-eluting bioresorbable vascular scaffold system against the XIENCE everolimus-eluting coronary stent system in the treatment of subjects with ischemic heart disease caused by de novo native coronary artery lesions: Rationale and study design. Am Heart J. 2012;164:654-63.
40. Kereiakes DJ, Ellis SG, Metzger DC, et al.; ABSORB III Investigators. Clinical outcomes before and after complete everolimus-eluting bioresorbable scaffold resorption: five-year follow-up from the ABSORB III trial. Circulation. 2019;140:1895-903.
41. Tesfamariam B. Bioresorbable vascular scaffolds: biodegradation, drug delivery and vascular remodeling. Pharmacol Res. 2016;107:163-71.
42. Mostaed E, Sikora-Jasinska M, Drelich JW, Vedani M. Zinc-based alloys for degradable vascular stent applications. Acta Biomater. 2018;71:1-23.
43. Stone GW, Kimura T, Gao R, et al. Time-varying outcomes with the absorb bioresorbable vascular scaffold during 5-year follow-up: a systematic meta-analysis and individual patient data pooled study. JAMA Cardiol. 2019;4:1261-9.
44. Waksman R. Current state of the absorbable metallic (magnesium) stent. EuroIntervention. 2009;5 Suppl F:F94-7.
45. Barkholt TØ, Webber B, Holm NR, Ormiston JA. Mechanical properties of the drug-eluting bioresorbable magnesium scaffold compared with polymeric scaffolds and a permanent metallic drug-eluting stent. Catheter Cardiovasc Interv. 2020;96:E674-82.
46. IBS TitanTM peripheral scaffold. Available from https://www.biotyxmed.com/en_ggjt.html [accessed 3 March 2026].
47. MeRes100TM Brochure. Available from https://storage.e.jimdo.com/file/d4a3bcc8-af71-49d6-bf6f-90cb65f9681c/meres100-bioresorbable-scaffolds-brs-1617184336pdf.pdf [accessed 3 March 2026].
48. Lee JM, Joh HS, Choi KH, et al.; SMART-REWARD Investigators. Safety and efficacy of everolimus-eluting bioresorbable vascular scaffold versus second-generation drug-eluting stents in real-world practice. J Korean Med Sci. 2023;38:e34.
49. Gao Z, Jiang W, Ran F. Peripheral vascular bioresorbable scaffolds: past, present, and future. Medicine Plus. 2024;1:100031.
50. Bosiers MJ, Rand T, Uberoi R, et al. MOTIV bioresorbable scaffold in below-the-knee artery disease: European post-market pilot BTK trial: 36-month results. Cardiovasc Intervent Radiol. 2025;48:1576-86.
51. Brodmann M, Falah B, Palena LM, et al. Clinical and angiographic outcomes of a novel thin strut Poly(L-lactide) based bioresorbable vascular scaffold in below knee arterial disease: the RESOLV I first in human study. Eur J Vasc Endovasc Surg. 2025.
52. Seguchi M, Aytekin A, Xhepa E, et al. Vascular response following implantation of the third-generation drug-eluting resorbable coronary magnesium scaffold: an intravascular imaging analysis of the BIOMAG-I first-in-human study. EuroIntervention. 2024;20:e1173-83.
53. Charpentier E, Barna A, Guillevin L, Juliard JM. Fully bioresorbable drug-eluting coronary scaffolds: a review. Arch Cardiovasc Dis. 2015;108:385-97.
54. Green MR, Couchman JR. Differences in human skin between the epidermal growth factor receptor distribution detected by EGF binding and monoclonal antibody recognition. J Invest Dermatol. 1985;85:239-45.
55. Pradhan A, Vishwakarma P, Vankar S, Sethi R. “The unpredictable ABSORB” - very late stent thrombosis of bioresorbable vascular scaffold. Heart Views. 2019;20:65-9.
56. Serruys PW, Katagiri Y, Sotomi Y, et al. Arterial remodeling after bioresorbable scaffolds and metallic stents. J Am Coll Cardiol. 2017;70:60-74.
57. Koppula AV, Raja MS, Ahmad M. Can HbA1c levels be used as a prognostic tool for patients undergoing coronary interventions? JACC Cardiovasc Interv. 2021;14:1159.
58. Giacchi G, Ortega-Paz L, Brugaletta S, Ishida K, Sabaté M. Bioresorbable vascular scaffolds technology: current use and future developments. Med Devices. 2016;9:185-98.
59. Jiang M, Zhang Y, Han Y, Yuan X, Gao L. Neoatherosclerosis: a distinctive pathological mechanism of stent failure. Rev Cardiovasc Med. 2024;25:95.
60. Brugaletta S, Heo JH, Garcia-Garcia HM, et al. Endothelial-dependent vasomotion in a coronary segment treated by ABSORB everolimus-eluting bioresorbable vascular scaffold system is related to plaque composition at the time of bioresorption of the polymer: indirect finding of vascular reparative therapy? Eur Heart J. 2012;33:1325-33.
61. Gomez-Lara J, Brugaletta S, Ortega-Paz L, et al. Long-term coronary functional assessment of the infarct-related artery treated with everolimus-eluting bioresorbable scaffolds or everolimus-eluting metallic stents: insights of the TROFI II trial. JACC Cardiovasc Interv. 2018;11:1559-71.
62. Ortega-Paz L, Capodanno D, Gori T, et al. Predilation, sizing and post-dilation scoring in patients undergoing everolimus-eluting bioresorbable scaffold implantation for prediction of cardiac adverse events: development and internal validation of the PSP score. EuroIntervention. 2017;12:2110-7.
63. Farhan M, Hasan G, Sobhi A, et al. Bioresorbable scaffolds advances, challenges, and future directions. Ann Med Surg. 2025;87:4173-83.
64. Cheng Y, Ferrone M, Wang Q, et al. Impact of coronary atherosclerosis on bioresorbable vascular scaffold resorption and vessel wall integration. JACC Basic Transl Sci. 2020;5:619-29.
65. El Khoury R, Tzvetanov I, Estrada EA, et al. Drug-eluting, balloon-expandable, bioresorbable vascular scaffolds reduce neointimal thickness and stenosis in an animal model of percutaneous peripheral intervention. JVS Vasc Sci. 2023;4:100114.
66. Vahl TP, Gasior P, Gongora CA, et al. Four-year polymer biocompatibility and vascular healing profile of a novel ultrahigh molecular weight amorphous PLLA bioresorbable vascular scaffold: an OCT study in healthy porcine coronary arteries. EuroIntervention. 2016;12:1510-8.
67. Torii S, Yamamoto A, Yoshikawa A, et al. Degradation of a novel magnesium alloy-based bioresorbable coronary scaffold in a swine coronary artery model. Cardiovasc Interv Ther. 2024;39:428-37.
68. Kumar S, Malviya R, Sridhar SB. Bioresorbable polymeric scaffold: advancing minimally invasive surgical procedure for cardiovascular and its applications. In: Malviya R, Sundram S, Editors. Sustainable nanocomposites with green biomaterials. Springer Nature Switzerland: Cham; 2025; pp. 61-82.
69. Lipinski MJ, Acampado E, Cheng Q, et al. Comparison of acute thrombogenicity for magnesium versus stainless steel stents in a porcine arteriovenous shunt model. EuroIntervention. 2019;14:1420-7.
70. Waksman R, Lipinski MJ, Acampado E, et al. Comparison of acute thrombogenicity for metallic and polymeric bioabsorbable scaffolds: magmaris versus absorb in a porcine arteriovenous shunt model. Circ Cardiovasc Interv. 2017;10.
71. Warren BE, Tan KT, Rajan DK, et al. Moving away from metal: step toward the future with bioresorbable vascular scaffolds and novel antiproliferative agents. JVS Vasc Sci. 2025;6:100277.
72. Kum S, Ipema J, Chun-Yin DH, et al. Early and midterm experience with the absorb everolimus-eluting bioresorbable vascular scaffold in Asian patients with chronic limb-threatening ischemia: one-year clinical and imaging outcomes from the DISAPEAR registry. J Endovasc Ther. 2020;27:616-22.
73. Varcoe RL, Schouten O, Thomas SD, Lennox AF. Experience with the absorb everolimus-eluting bioresorbable vascular scaffold in arteries below the knee: 12-month clinical and imaging outcomes. JACC Cardiovasc Interv. 2016;9:1721-8.
74. Varcoe RL, Parikh SA, DeRubertis BG, et al. Evaluation of an infrapopliteal drug-eluting resorbable scaffold: design methodology for the LIFE-BTK randomized controlled trial. J Soc Cardiovasc Angiogr Interv. 2023;2:100964.
75. R3 Vascular Inc. Bioresorbable Sirolimus-eluting scaffold treatment for below the knee disease (ELITE-BTK). Available from https://clinicaltrials.gov/study/NCT06071429 [accessed 3 March 2026].
76. Drug impregnated bioabsorbable stent in asian population extremity arterial revascularization (DISAPEAR Study) (DISAPEAR). Available from https://clinicaltrials.gov/study/NCT02043795 [accessed 3 March 2026].
77. LIFE-BTK Randomized Controlled Trial (LIFE-BTK). Available from https://clinicaltrials.gov/study/NCT04227899 [accessed 3 March 2026].
78. R3 Vascular Inc. Study of the R3 Vascular Drug-Eluting Bioresorbable Scaffold in Treating Below the Knee Arterial Disease (RESOLV I). Available from https://clinicaltrials.gov/study/NCT04912323 [accessed 3 March 2026].
79. MOTIV BTK randomized controlled trial. Available from https://clinicaltrials.gov/study/NCT05406622 [accessed 3 March 2026].
80. Abbott Vascular. The ABSORB bioresorbable scaffold below the knee (BTK) study. Available from https://clinicaltrials.gov/study/NCT02793349 [accessed 3 March 2026].
81. Peng X, Qu W, Jia Y, Wang Y, Yu B, Tian J. Bioresorbable scaffolds: contemporary status and future directions. Front Cardiovasc Med. 2020;7:589571.
82. Martinelli O, Alunno A, Drudi FM, Malaj A, Irace L. Duplex ultrasound versus CT angiography for the treatment planning of lower-limb arterial disease. J Ultrasound. 2021;24:471-9.
83. Demola P, Meucci F, Stolcova M, Mario CD, Mattesini A. The DESolve® novolimus bioresorbable scaffold. Future Cardiol. 2021;17:945-51.
84. Abizaid A, Kedev S, Ali RBM, et al. Imaging and 2-year clinical outcomes of thin strut sirolimus-eluting bioresorbable vascular scaffold: the MeRes-1 extend trial. Catheter Cardiovasc Interv. 2021;98:1102-10.
85. Song L, Xu B, Chen Y, et al.; FUTURE-II Trial Investigators. Thinner strut sirolimus-eluting BRS versus EES in patients with coronary artery disease: FUTURE-II trial. JACC Cardiovasc Interv. 2021;14:1450-62.
86. Jiménez JM, Davies PF. Hemodynamically driven stent strut design. Ann Biomed Eng. 2009;37:1483-94.
87. van Zandvoort LJC, Dudek D, Weber-Albers J, et al. Intravascular ultrasound findings of the Fantom sirolimus-eluting bioresorbable scaffold at six- and nine-month follow-up: the FANTOM II study. EuroIntervention. 2018;14:e1215-23.
88. Fan W, Tan J, Li L, et al. Efficacy and safety of absorb everolimus-eluting bioresorbable vascular scaffold in peripheral artery disease: a single-arm meta-analysis. J Endovasc Ther. 2023;30:651-63.
89. Beckman JA, Schneider PA, Conte MS. Advances in revascularization for peripheral artery disease: revascularization in PAD. Circ Res. 2021;128:1885-912.
90. Zhao N, Zhu D. Endothelial responses of magnesium and other alloying elements in magnesium-based stent materials. Metallomics. 2015;7:118-28.
91. Saberi A, Bakhsheshi-rad HR, Abazari S, et al. A comprehensive review on surface modifications of biodegradable magnesium-based implant alloy: polymer coatings opportunities and challenges. Coatings. 2021;11:747.
92. Leone A, Simonetti F, Avvedimento M, et al. Ultrathin struts drug-eluting stents: a state-of-the-art review. J Pers Med. 2022;12:1378.
93. Haude M, Wlodarczak A, van der Schaaf RJ, et al. A new resorbable magnesium scaffold for de novo coronary lesions (DREAMS 3): one-year results of the BIOMAG-I first-in-human study. EuroIntervention. 2023;19:e414-22.
94. Menze R, Hesse B, Kusmierczuk M, et al. Synchrotron microtomography reveals insights into the degradation kinetics of bio-degradable coronary magnesium scaffolds. Bioact Mater. 2024;32:1-11.
95. Kang SH, Gogas BD, Jeon KH, et al. Long-term safety of bioresorbable scaffolds: insights from a network meta-analysis including 91 trials. EuroIntervention. 2018;13:1904-13.
96. Early M, Kelly DJ. The consequences of the mechanical environment of peripheral arteries for nitinol stenting. Med Biol Eng Comput. 2011;49:1279-88.
97. Struczewska P, Razian SA, Townsend K, et al. Mechanical, structural, and physiologic differences between above and below-knee human arteries. Acta Biomater. 2024;177:278-99.
98. Sakamoto A, Jinnouchi H, Torii S, Virmani R, Finn AV. Understanding the impact of stent and scaffold material and strut design on coronary artery thrombosis from the basic and clinical points of view. Bioengineering. 2018;5:71.
99. Nguyen DT, Smith AF, Jiménez JM. Stent strut streamlining and thickness reduction promote endothelialization. J R Soc Interface. 2021;18:20210023.
100. Marsden Back L, Gentry-Maharaj A, Ladwiniec A. The safety and efficacy profile of magnesium-based bioresorbable coronary stents as compared to poly-L-lactic acid-based bioresorbable and contemporary drug-eluting coronary stents-a systematic review. Cardiol Res Pract. 2025;2025:7481956.
101. Cheng CK, Wang N, Wang L, Huang Y. Biophysical and biochemical roles of shear stress on endothelium: a revisit and new insights. Circ Res. 2025;136:752-72.
102. Kawashima H, Ono M, Kogame N, et al. Drug-eluting bioresorbable scaffolds in cardiovascular disease, peripheral artery and gastrointestinal fields: a clinical update. Expert Opin Drug Deliv. 2020;17:931-45.
103. Azzalini L, Ellis SG, Kereiakes DJ, et al. Optimal dual antiplatelet therapy duration for bioresorbable scaffolds: an individual patient data pooled analysis of the ABSORB trials. EuroIntervention. 2021;17:e981-8.
104. Wu Y, Yin J, Li C, et al. The standard versus prolonged dual antiplatelet therapy after the XINSORB bioresorbable scaffold implantation (SPARTA) trial: study protocol for a randomized controlled trial. Trials. 2023;24:49.
105. Angiolillo DJ, Galli M, Alexopoulos D, et al. International consensus statement on platelet function and genetic testing in percutaneous coronary intervention: 2024 update. JACC Cardiovasc Interv. 2024;17:2639-63.
106. O’Riordan M. The bioresorbable stent story so far: what promise? What price? Available from https://www.tctmd.com/news/bioresorbable-stent-story-so-far-what-promise-what-price [accessed 3 March 2026].
107. Baron SJ, Lei Y, Chinnakondepalli K, et al.; ABSORB III Investigators. Economic outcomes of bioresorbable vascular scaffolds versus everolimus-eluting stents in patients undergoing percutaneous coronary intervention: 1-year results from the ABSORB III trial. JACC Cardiovasc Interv. 2017;10:774-82.
108. Kozuma K, Tanabe K, Hamazaki Y, et al.; ABSORB Japan Investigators. Long-term outcomes of absorb bioresorbable vascular scaffold vs. everolimus-eluting metallic stent- a randomized comparison through 5 years in Japan. Circ J. 2020;84:733-41.
109. Europe Bioresorbable Vascular Scaffold Market Worth $103.45 Million By 2030. Available from https://www.grandviewresearch.com/press-release/europe-bioresorbable-vascular-scaffold-market-analysis [accessed 3 March 2026].
110. McKeown LA. Bioresorbable scaffold for CLTI appears to be an economical option: LIFE-BTK. Available from https://www.tctmd.com/news/bioresorbable-scaffold-clti-appears-be-economical-option-life-btk [accessed 3 March 2026].
111. Wlodarczak A, Montorsi P, Torzewski J, et al. One- and two-year clinical outcomes of treatment with resorbable magnesium scaffolds for coronary artery disease: the prospective, international, multicentre BIOSOLVE-IV registry. EuroIntervention. 2023;19:232-9.
112. Felix C. Absorb bioresorbable vascular scaffold (BVS) for the treatment of coronary artery disease in clinical practice. 2019. https://repub.eur.nl/pub/116450/ [accessed 3 March 2026].
113. Koh DJ, Tao BS, Alonso A, et al. Regional and specialty-based medicare reimbursement trends in arterial endovascular procedures. J Vasc Surg. 2025;82:1503-1510.e1.
114. Schmid T. Costs of treating cardiovascular events in Germany: a systematic literature review. Health Econ Rev. 2015;5:27.
115. Wang Y, Zhang X. Vascular restoration therapy and bioresorbable vascular scaffold. Regen Biomater. 2014;1:49-55.
116. Gaudiello E, Melly L, Cerino G, et al. Scaffold composition determines the angiogenic outcome of cell-based vascular endothelial growth factor expression by modulating its microenvironmental distribution. Adv Healthc Mater. 2017;6:1700600.
117. Chu TM, Chan YC, Cheng SW. Evidence for treating peripheral arterial diseases with biodegradable scaffolds. J Cardiovasc Surg. 2017;58:87-94.
118. Tomilova OG, Kryukov VY, Kryukova NA, et al. Effects of passages through an insect or a plant on virulence and physiological properties of the fungus Metarhizium robertsii. PeerJ. 2023;11:e15726.
119. Gordon LG, Rodriguez-Acevedo AJ, Køster B, et al. Association of indoor tanning regulations with health and economic outcomes in North America and Europe. JAMA Dermatol. 2020;156:401-10.
120. Helou B, Bel-Brunon A, Dupont C, et al. Patient-specific finite element simulation of peripheral artery percutaneous transluminal angioplasty to evaluate the procedure outcome without stent implantation. Int J Numer Method Biomed Eng. 2023;39:e3685.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].