Artificial intelligence in breast reduction surgery: a review of preoperative planning, intraoperative assessment and postoperative outcome prediction
Abstract
According to the German Association of Aesthetic Plastic Surgeons (VDÄPC, 2024), breast reduction surgery is among the most frequently performed plastic surgical procedures on the female breast. The primary goal of this operation is to reduce the volume of glandular and adipose tissue and to reposition the nipple-areola complex, for example, in cases of hypertrophic breasts. Indications include both functional impairments - such as chronic pain in the upper spine or recurrent intertriginous dermatitis in the inframammary fold - and aesthetic concerns. The first documented breast reduction procedures date back to the mid-19th century and have since undergone continuous evolution. To this day, there is no standardized protocol for preoperative marking, resulting in significant variability among clinics and surgeons. In recent years, artificial intelligence (AI) has gained increasing relevance in clinical practice, especially in enhancing diagnostic processes, surgical planning, and outcome prediction. This narrative review examines the current literature on the integration of digital solutions and AI into breast reduction surgery. The objective is to identify current applications, explore the potential of AI-assisted technologies in preoperative planning and assessment, and provide a perspective on future developments. By doing so, this article aims to contribute to the structured evaluation of the interdisciplinary interface between plastic surgery and digital innovation. It highlights the potential of AI to enable standardized, personalized, and precise surgical planning in breast reduction procedures, ultimately improving clinical outcomes and patient satisfaction.
Keywords
INTRODUCTION
To understand the current role of artificial intelligence (AI) in breast surgery, it is crucial to consider the historical evolution of reduction mammaplasty. In 1921, Lexer performed the first documented breast reduction using a centrally pedicled technique in which the nipple-areola complex (NAC) was transposed on a central glandular pedicle, combined with periareolar resection of glandular tissue. Over the decades, numerous modifications have been introduced, particularly regarding pedicle orientation and flap designs, including the lateral intercostal artery perforator (LICAP) flap[1,2]. Today, reduction mammaplasty is primarily performed to reduce glandular and adipose tissue volume, reposition the NAC, and reestablish a functional and aesthetic breast shape. Patients seek the procedure for both medical and aesthetic reasons, including relief from musculoskeletal pain, chronic intertriginous rashes, and dissatisfaction with breast proportions or asymmetry. Surgical techniques such as the Wise pattern (inverted-T incision), vertical scar methods (e.g., Lejour), and superior or inferior NAC pedicle transpositions are commonly employed[3]. Preoperative marking plays a critical role in achieving symmetry and determining resection volumes. Anatomical landmarks such as the sternal notch, current and intended NAC location, inframammary fold (IMF), and breast meridians guide this process. However, significant inter-surgeon variability exists, and no standardized international protocol for preoperative marking is currently established[4].
AI has already been successfully integrated into preoperative planning in various surgical fields. In oral and maxillofacial surgery (OMFS), AI-based deep learning algorithms enable the automatic segmentation of anatomical structures such as bones and nerves in computed tomography (CT) and magnetic resonance imaging (MRI) images. First, high-resolution CT or MRI images of the area to be operated on are acquired. Based on this data, a three-dimensional (3D) patient dataset is created[5]. In the virtual reality (VR) environment, the operation can be rehearsed in detail on, for example, a Styrofoam model to simulate surgical scenarios in advance, identify risks, and adjust incision surfaces or contours. Augmented reality (AR) is used to overlay the planned procedure onto the real body. This enables planning for operations such as osteotomies for jaw or midface repositioning, sagittal split osteotomies of the mandible, or the insertion of patient-specific implants or bone plates based on 3D models for complex reconstructions[5]. Additionally, outcome prediction models assist in preoperative consultations by estimating aesthetic results and identifying potential complications. AR and VR systems further enhance intraoperative navigation[5]. In orthopedic and trauma surgery, AI has demonstrated significant advantages in total joint arthroplasty. It improves prosthesis selection and alignment precision, standardizes planning workflows, and ultimately results in better postoperative functional outcomes for patients[6].
In plastic surgery, AI has begun to reshape elements of preoperative planning. According to Leypold et al., previous studies have shown that ChatGPT-4 can analyze complex clinical scenarios and suggest appropriate treatment options, while also considering relevant comorbidities. These capabilities may support surgeons in preoperative planning and decision-making. Using prompting techniques such as expertise emulation and chain-of-thought reasoning, GPT-4 can simulate realistic surgical decisions. Although current limitations remain, the authors highlight the significant potential of GPT-4 in plastic surgery, particularly for image analysis, outcome prediction, and virtual consultation[7]. Another prominent area of application is in flap-based reconstruction[8]. AI-assisted segmentation of computed tomography angiography (CTA) scans allows for rapid and accurate identification of suitable perforator vessels, reducing manual segmentation times from 2 to 3 h to approximately 30 min. Furthermore, AI systems can now provide surgical recommendations, such as flap selection based on individual anatomy[9,10]. AI has also enabled the generation of 3D models of the torso and breasts using depth sensors, enhancing the precision of surgical markings and spatial planning[11,12]. Additionally, generative AI tools offer realistic pre- and post-operative simulations to support patient education and informed consent, creating a more personalized and transparent surgical experience[13].
Despite these advances, AI has not yet been fully applied to reduction mammaplasty, particularly in the area of preoperative marking, which remains largely manual, subjective, and operator-dependent. Given that preoperative drawings play a pivotal role in determining symmetry, aesthetic outcome, and patient satisfaction, integrating AI-based tools could significantly enhance standardization, reproducibility, and planning efficiency[4,14]. Thus, the application of AI in this context represents a highly relevant and promising field for future research and clinical innovation.
STATE OF THE ART
Clinical need and current practice
Reduction mammaplasty is a well-established procedure in plastic surgery, primarily indicated for patients suffering from macromastia. A disproportionately large breast volume relative to body size can lead to various physical complaints, including chronic back and neck pain, recurrent intertriginous infections in the IMF and reduced mobility[15]. In addition to these physical symptoms, many patients report significant psychological distress related to their breast size and body image[16]. The primary surgical goal of reduction mammaplasty is to reduce breast volume to alleviate the psychological and physical symptoms. However, from an aesthetic perspective, achieving a symmetrical and harmonious postoperative breast shape is equally crucial[17]. According to Xiong et al. and Yang et al., postoperative breast symmetry is a central quality metric and strongly influences patient satisfaction following reduction mammaplasty[18,19]. Preoperative planning in breast reduction surgery currently relies heavily on manual markings, which are dependent on the surgeon’s experience and inherently subjective[20]. Xiong et al. emphasize that precise preoperative planning is essential for achieving postoperative symmetry. Using CT-based 3D-printed vascular models, they visualized the patient’s breast anatomy and arterial supply, enabling individualized incision planning and improved preservation of the NAC to enhance postoperative symmetry[18]. Classical measurement techniques involve determining the sternal notch-to-nipple distance, nipple size, and the nipple-to-IMF length using measuring tapes or calipers. These methods are prone to inter-individual variability and depend strongly on visual approximation[16]. According to Monton et al., such visual estimations are susceptible to distortion and inaccuracies. These inaccuracies arise mainly from the subjective assessment of breast symmetry using tools (e.g., measuring tapes and calipers), which are highly dependent on the surgeon’s experience and can be affected by patient positioning, perspective, and lighting conditions[20]. Li et al. further confirm that postoperative deviations in nipple position and breast volume are frequent, largely due to the limited predictive accuracy of currently used methods[17]. As Xiong et al. note, conventional tools such as rulers or tape measures are highly inconsistent and error-prone[18].
Digital solutions and evidence
3D surface scanning and morphometry
Precise planning of a breast reduction requires not only a thorough understanding of breast anatomy but also detailed knowledge of key measurements and planning parameters. An overview of the currently reported AI-supported technologies and their clinical applications in breast reduction surgery is summarized in Table 1. According to Li et al., 3D scanning is already being used pre- and postoperatively to objectively quantify breast volume, the position of the NAC, breast base width, and IMF[17]. Despite the high relevance of breast symmetry for patient satisfaction, there is no established method to objectively assess symmetry in clinical plastic surgery practice. Among the most relevant landmarks are the sternal notch-to-nipple distance, the NAC-to-IMF distance, and breast projection. For digital volumetric and morphological analysis, tools such as the Breast Analysis Technology (BAT) and the Gradient Boosting Artificial Intelligence (GBAI) allow for an objective evaluation of breast shape before and after surgery. BAT is a computer-assisted image analysis software that quantifies parameters such as breast contour, nipple position, and breast projection using standardized two-dimensional (2D) photographs, enabling reproducible aesthetic assessments. GBAI, on the other hand, is a composite index that integrates various anthropometric and aesthetic variables to generate a numerical score of breast symmetry and proportion, offering a standardized measure of surgical outcomes[20]. Koban
AI applications in breast reduction planning
| Study (first author, year) | AI/digital modality | Phase of application | Clinical application in breast Reduction | Key contribution |
| Seu, 2024[16] | Machine learning | Preoperative | Prediction of total resection weight | Individualized estimation of resection volume |
| Shoham, 2025[23] | Gradient boosting (XGBoost) | Preoperative | Complication risk prediction | Personalized risk stratification |
| Li, 2024[17] | Deep learning + 3D analysis | Pre- and postoperative | Landmark tracking and volumetric analysis | Quantitative evaluation of morphological changes |
| Yang, 2021[19] | Intraoperative 3D scanning | Intraoperative | Real-time symmetry assessment | Dynamic intraoperative correction |
| Koban, 2018[21] | Structured-light 3D scanning | Preoperative | Objective volumetry and nipple position analysis | Feasibility of mobile 3D imaging |
| Chen, 2012[24] | Laser projection grid | Intraoperative | Nipple positioning and symmetry alignment | Improved marking accuracy |
| Chae, 2014[11] | 3D volumetric analysis | Preoperative | Breast volume measurement | Objective volumetric planning |
| Xiong, 2022[18] | 3D printing | Preoperative | Patient-specific breast models | Enhanced spatial planning |
| Abi-Rafeh, 2024[14] | Large language models | Preoperative | AI-based patient education | Improved shared decision-making |
| Boyd, 2024[25] | AI triage tool | Perioperative | Risk triage and monitoring | Workflow optimization |
Devices and systems used for AI-supported applications in breast reduction surgery
| Figure no. | Study (first author, year) | Technology/device | Clinical application | Example image |
| 1 | Koban, 2018[21] | Portable structured-light 3D scanner | Preoperative breast surface scanning and volumetric analysis |  |
| 2 | Chen, 2012[24] | Laser projection grid system | Intraoperative nipple alignment and symmetry control | 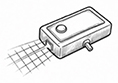 |
| 3 | Xiong, 2022[18] | 3D-printed patient-specific breast model | Preoperative planning and surgical simulation | 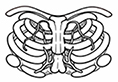 |
Machine-learning prediction
Shoham et al. developed an AI–based prediction model using Gradient Boosting (eXtreme gradient boosting, XGBoost) to identify patients at increased risk of postoperative complications - such as nipple–areola necrosis, infection, or wound dehiscence - within 30 days after breast reduction surgery. Analyzing retrospective data from 322 procedures, the authors evaluated a range of clinical and operative variables, including specimen weight, sternal notch–to–nipple (SN-N) distance, liposuction volume, body mass index (BMI), and smoking or diabetic status. These predictors were selected based on prior clinical evidence and their statistical association with complications. By capturing complex, nonlinear relationships between anatomical and clinical parameters, the Gradient Boosting model enabled individualized complication–risk profiling and supported more nuanced, patient-specific surgical planning. This AI-driven approach thus allows for the preoperative identification of high-risk patients, facilitating personalized decision-making and targeted counseling to improve surgical safety and outcomes[23]. Seu et al. implemented a supervised machine-learning (ML) approach to predict breast resection weight based on preoperative anthropometric measurements, including BMI, body surface area, SN-N distance, and nipple-to-IMF distance. They trained and compared four regression models - linear, ridge, support vector, and random forest - and demonstrated that the random forest regression model achieved the highest accuracy, outperforming traditional methods such as the Schnur scale. The model can be refined through continuous postoperative training with new data, and future implementations could incorporate dynamic updates and more comprehensive multicenter datasets, potentially enabling the tool to support not only volume planning but also real-time surgical decision-making and complication prediction[16]. Shoham et al. and Seu et al. thus demonstrated the clinical applicability of AI in perioperative prediction of complications and estimation of resection volume. These AI-based prediction models and their clinical applications are summarized in Table 1. AI models such as XGBoost and random forest provide personalized, data-driven prognostic outputs, offering a framework for individualized and evidence-based decision-making[16,23].
AI may support decision-making regarding pedicle type and positioning; however, direct evidence specifically addressing pedicle selection in reduction mammaplasty remains limited. Current AI applications primarily focus on volumetric assessment, symmetry analysis, and complication risk prediction[16,17,23]. ML models incorporating parameters such as SN-N distance, resection weight, BMI, and comorbidities may indirectly inform pedicle planning by identifying patients at increased risk of nipple–areola complex compromise. Additionally, 3D surface imaging systems[19,21] provide objective morphological data that can support individualized surgical planning. Thus, while AI does not currently determine pedicle type autonomously, it may function as a decision-support tool to assist technique selection in the future.
AR and laser projection
Chen et al. introduced and successfully implemented intraoperative laser projection systems that use real-time horizontal alignment tools to precisely transfer preoperative markings and delineate nipple positions as well as axes of symmetry during surgery [Table 1]. These systems enhance the accuracy of intraoperative execution, enable real-time assessment of breast symmetry and thereby help prevent postoperative asymmetries[24]. A laser projection grid used for intraoperative nipple alignment is illustrated in Figure 2 and listed among the devices and systems in Table 2. Kiwan et al. were the first to propose and test the application of AR in the context of breast surgery. Their study examined the superimposition of digital breast imaging data onto the patient’s body to support preoperative visualization of incision lines and anatomical contours. While still in the experimental stage, this approach holds significant promise for reducing symmetry-related errors in manual preoperative markings[8].
Vascular and anatomical modeling
Xiong et al. used preoperative CT-based 3D models to simulate vascular anatomy and volume distribution, especially in complex breast anatomies. They reconstructed vascular structures such as the internal thoracic artery and its branches, generating 3D-printed vascular models to guide the surgeon before surgery. This approach supports safer, anatomically accurate resections through visualization of dominant arteries supplying the nipple complex[18]. An example of a patient-specific 3D printed breast model and the associated important vessels used for surgical planning is shown in Figure 3 and listed among the devices and systems in Table 2. These CT-based models can also be used intraoperatively, particularly in cases of complex breast anatomy. Real-time visualization of important vessels, such as the internal mammary artery and its branches, could help minimize the risk of vascular injury and enable more informed surgical decisions[18].
Implementation considerations
While AI, 3D scanning, and AR technologies have demonstrated significant potential in improving preoperative planning and symmetry assessment, several challenges still limit their broad implementation in clinical practice. At present, most AI and 3D-scanning studies in plastic surgery remain single-center, exploratory investigations with small sample sizes, which restrict generalizability and hinder the establishment of standardized workflows. As Boyd et al. emphasize, there remains a critical need to ensure that the preoperatively planned resection volume and breast symmetry are reassessed and dynamically adjusted intraoperatively[25]. The absence of validated multicenter trials and heterogeneous reporting metrics makes it difficult to compare outcomes across studies and to derive robust clinical recommendations[22]. Another key barrier to implementation is the dependence on specialized hardware and software systems. 3D scanners, laser projection grids, and AR headsets require technical expertise, calibration, and training that may not be readily available in every surgical department. Integration into existing hospital information technology (IT) infrastructures can be challenging, particularly in institutions with limited digital resources or data security constraints. Furthermore, the acquisition costs of such technologies - including imaging devices, computational resources, and proprietary analysis software - can represent a significant financial burden for smaller centers[8,20]. In addition to technical and logistical considerations, surgeon acceptance and user training play a pivotal role. Even the most advanced AI-based systems depend on consistent, high-quality input data to achieve reliable results. Variability in patient positioning, lighting, or measurement techniques can adversely affect 3D scans and automated analyses, limiting reproducibility[17]. Moreover, as highlighted by Seu et al., ML algorithms require continuous retraining and validation on diverse populations to avoid overfitting and ensure applicability to different body morphologies and ethnic groups[16]. From a regulatory perspective, there is currently no harmonized approval framework for AI-driven surgical planning tools. Clinical-grade AI applications must comply with data privacy laws [e.g., General Data Protection Regulation (GDPR)] and medical device regulations, yet many prototypes have been tested only in research settings. The transition from experimental tools to certified clinical devices will require rigorous validation, risk assessment, and quality management processes. Lastly, ethical and legal concerns remain an ongoing challenge. The increasing use of AI-based prediction models raises questions regarding transparency, algorithmic bias, and accountability. If an AI-generated recommendation contributes to an unfavorable outcome, the legal attribution of responsibility between surgeon and software developer remains ambiguous. Ensuring explainability and traceability of algorithmic decisions is therefore crucial to maintain patient trust and medico-legal safety[8,23].
In summary, successful implementation of digital and AI-assisted systems in reduction mammaplasty will depend on three critical factors: (1) establishing standardized and validated data acquisition and analysis workflows; (2) integrating user-friendly interfaces that enhance, rather than complicate, clinical decision-making; and (3) conducting prospective multicenter studies that evaluate reproducibility, safety, and patient satisfaction using harmonized endpoints.
Evidence gaps and future directions
Although numerous studies have explored the use of AI, 3D scanning, and AR in breast surgery, the current evidence remains largely preliminary and fragmented. Most published investigations are limited to feasibility analyses, case series, or small-sample pilot trials without standardized outcome measures[22]. This lack of large-scale, prospective, multicenter validation restricts the generalizability of existing findings and prevents reliable comparisons between different technological approaches. As highlighted by Monton et al. and Nogueira et al., even advanced digital tools such as the BAT and the GBAI still rely on heterogeneous datasets and non-uniform imaging protocols, which complicates interstudy comparisons and reproducibility[20,22]. To achieve clinical standardization, future research must focus on defining uniform metrics for symmetry, aesthetic outcomes, and patient satisfaction, supported by validated 3D anthropometric measurements. Furthermore, while 3D scanning and AI-based modeling have demonstrated clear potential for preoperative planning, their integration into intraoperative and postoperative workflows remains underdeveloped. As Yang et al. and Li et al. emphasize, intraoperative real-time symmetry verification is still uncommon in clinical practice, despite its potential to reduce revision rates and enhance outcome predictability[17,19]. Future systems should aim to combine intraoperative 3D scanning, automatic landmark recognition, and real-time correction algorithms to enable a closed feedback loop between planning and execution. AR represents another promising but currently experimental frontier. The prototypes developed by Kiwan et al. illustrate the feasibility of projecting incision lines and volumetric contours directly onto the patient’s body, potentially transforming manual preoperative marking into a precise digital overlay[8]. However, no randomized controlled studies have yet quantified whether AR-guided planning leads to measurable improvements in symmetry, operative efficiency, or complication rates compared to traditional approaches. AI-driven prediction models also warrant further refinement. The ML models presented by Shoham et al. and Seu et al. have proven effective in predicting complication risks and resection weights, but they require continuous retraining on diverse patient populations to prevent bias and maintain accuracy[16,23]. The incorporation of multimodal datasets - including clinical variables, imaging data, and patient-reported outcomes - could further enhance predictive robustness. Xiong et al. demonstrated the value of CT-based 3D vascular models in preoperative planning, yet these systems have not been widely adopted due to their resource intensity and technical complexity[18]. Simplified, automated vascular modeling software could make this approach more accessible for routine use, especially when integrated with AI-based vessel recognition algorithms. Moreover, there is an urgent need for translational research that bridges engineering and clinical disciplines. Multidisciplinary collaborations between surgeons, biomedical engineers, and data scientists will be essential for developing interoperable systems capable of communicating seamlessly across imaging, simulation, and operative platforms. In addition, future studies should assess not only technical accuracy but also patient-centered outcomes, such as satisfaction, body image, and quality of life. As highlighted by Li et al. and Yang et al., postoperative breast morphology continues to evolve dynamically over time, underscoring the need for long-term, quantitative follow-up using standardized 3D datasets[17,19]. Finally, establishing ethical, legal, and regulatory frameworks will be critical for the safe implementation of AI-based tools. Transparent algorithm design, data protection, and clear definitions of clinical accountability must accompany technological progress to ensure patient safety and trust[8,23]. In summary, despite remarkable technological advances, significant evidence gaps persist in the application of AI, AR, and 3D scanning to reduction mammaplasty. Addressing these limitations through rigorous multicenter validation, standardized protocols, and interdisciplinary cooperation will determine whether these tools can transition from experimental adjuncts to indispensable instruments of precision surgery. Future developments should prioritize real-time feedback mechanisms, seamless integration into operative workflows, and long-term outcome monitoring through automated morphometric analysis. If these challenges are met, the convergence of AI, AR, and 3D modeling could ultimately redefine the paradigm of breast reduction surgery - shifting it from subjective craftsmanship to a quantifiable, data-driven, and patient-specific discipline.
CONCLUSION
This narrative review demonstrates that a range of technological methods already exist to support preoperative planning and symmetry optimization in breast reduction surgery. Among these, 3D scanning technologies enable objective and reproducible assessment of breast volume, NAC position, breast base diameter and projection[17,21]. In addition, software-supported systems such as the BAT and the GBAI provide standardized tools for aesthetic outcome assessment, enhancing objectivity in postoperative symmetry evaluation[20,22]. Technologies including laser-assisted and AR-based projections allow for high-precision marking of incision lines, both pre- and intraoperatively[8,24]. Moreover, CT-based modeling offers critical insights into vascular anatomy, particularly in complex cases, enhancing surgical planning with respect to vessel preservation[18]. The supportive role of AI is highlighted by models such as XGBoost, which can outperform conventional clinical criteria in predicting postoperative complications such as wound-healing disorders. This allows for early identification of high-risk patients and improved preoperative stratification[23]. Similarly, ML algorithms such as random forest provide accurate volume and resection-weight predictions, which can help guide surgical strategy[16]. The potential of AI and AR in the future is already reflected in the early success of pilot studies. The integration of AI-based planning, intraoperative 3D scanning, AR-guided projections, and postoperative feedback loops could collectively transform breast reduction surgery - enabling highly individualized, complication-minimized and symmetry-optimized outcomes[16,19,22]. In particular, AR systems may enable real-time visualization of incision paths, adapting dynamically to intraoperative patient positioning[8]. Similarly, real-time intraoperative symmetry control through 3D scanning could reduce the rate of revision surgeries[17,18]. Overall, the integration of modern technologies into breast reduction surgery presents a paradigm shift toward quantifiable symmetry, improved aesthetic predictability and data-driven surgical decision-making. While AI and AR currently serve as adjunct tools, their future role is likely to evolve into disruptive technologies in surgical planning and execution. To achieve widespread clinical integration, prospective, multicenter trials and the development of standardized evaluation criteria will be essential for aligning technological advances with surgical outcomes.
DECLARATIONS
Acknowledgments
The authors gratefully acknowledge Professor Beier for providing the opportunity and academic support necessary to bring this work to publication. Special thanks are extended to Dr. Leypold for his dedicated supervision and continuous guidance throughout all stages of the manuscript preparation.
Authors’ contributions
Jointly authored the manuscript: Flender P, Leypold T
Provided consulting services regarding publication: Leypold T
Contributed expertise and proofreading: Beier JP
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT [(GPT-5.4), released 2026-03-05] was used to assist in language editing and the preparation of visual materials. The tool did not influence the study design, data collection, analysis, interpretation or the scientific content of the work. All authors take full responsibility for the accuracy, integrity and final content of the manuscript.
Financial support and sponsorship
None.
Conflicts of interest
All authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Bugariu M, Roushan A, Liebau J. Mastopexie - historie und aktuelle techniken. J Ästhet Chir. 2021;14:11-9.
3. Lejour M, Abboud M, Declety A, Kertesz P. [Reduction of mammaplasty scars: from a short inframammary scar to a vertical scar]. Ann Chir Plast Esthet. 1990;35:369-79.
4. Elia R, Maruccia M, Tedeschi P, Nacchiero E, Giudice G. The “Mosque Tower Pattern” for the preoperative marking of reduction mammaplasty. Plast Reconstr Surg Glob Open. 2024;12:e6230.
5. Miragall MF, Knoedler S, Kauke-Navarro M, et al. Face the future-artificial intelligence in oral and maxillofacial surgery. J Clin Med. 2023;12:6843.
6. Lan Q, Li S, Zhang J, Guo H, Yan L, Tang F. Reliable prediction of implant size and axial alignment in AI-based 3D preoperative planning for total knee arthroplasty. Sci Rep. 2024;14:16971.
7. Leypold T, Schäfer B, Boos A, Beier JP. Can AI think like a plastic surgeon? Evaluating GPT-4’s clinical judgment in reconstructive procedures of the upper extremity. Plast Reconstr Surg Glob Open. 2023;11:e5471.
8. Kiwan O, Al-Kalbani M, Rafie A, Hijazi Y. Artificial intelligence in plastic surgery, where do we stand? JPRAS Open. 2024;42:234-43.
9. Cevik J, Seth I, Rozen WM. Transforming breast reconstruction: the pioneering role of artificial intelligence in preoperative planning. Gland Surg. 2023;12:1271-5.
10. Sayadi LR, Hamdan US, Zhangli Q, Hu J, Vyas RM. Harnessing the power of artificial intelligence to teach cleft lip surgery. Plast Reconstr Surg Glob Open. 2022;10:e4451.
11. Chae MP, Hunter-Smith DJ, Spychal RT, Rozen WM. 3D volumetric analysis for planning breast reconstructive surgery. Breast Cancer Res Treat. 2014;146:457-60.
12. Lynn AQ, Pflibsen LR, Smith AA, Rebecca AM, Teven CM. Three-dimensional printing in plastic surgery: current applications, future directions, and ethical implications. Plast Reconstr Surg Glob Open. 2021;9:e3465.
13. Farid Y, Fernando Botero Gutierrez L, Ortiz S, et al. Artificial intelligence in plastic surgery: insights from plastic surgeons, education integration, ChatGPT’s survey predictions, and the path forward. Plast Reconstr Surg Glob Open. 2024;12:e5515.
14. Abi-Rafeh J, Bassiri-Tehrani B, Kazan R, et al. Preoperative patient guidance and education in aesthetic breast plastic surgery: a novel proposed application of artificial intelligence large language models. Aesthet Surg J Open Forum. 2024;6:ojae062.
15. Lessard L, Papanastasiou C, Fortin M, Ouellet JA. Breast hypertrophy: a real pain in the back. Plast Reconstr Surg. 2023;151:498-508.
16. Seu MY, Rezania N, Murray CE, et al. Predicting reduction mammaplasty total resection weight with machine learning. Ann Plast Surg. 2024;93:246-52.
17. Li H, Lin Y, Zhang X, Li Z, Mu D. 3D analysis of breast morphological changes after vertical-scar reduction mammoplasty: a prospective study. Aesthetic Plast Surg. 2024;48:3904-13.
18. Xiong S, E B, Zhang Z, et al. Innovative application of three-dimensional-printed breast model-aided reduction mammaplasty. Front Surg. 2022;9:890177.
19. Yang Y, Mu D, Xu B, et al. An intraoperative measurement method of breast symmetry using three-dimensional scanning technique in reduction mammaplasty. Aesthetic Plast Surg. 2021;45:2135-45.
20. Monton J, Torres A, Gijon M, et al. Use of symmetry assessment methods in the context of breast surgery. Aesthetic Plast Surg. 2020;44:1440-51.
21. Koban KC, Härtnagl F, Titze V, Schenck TL, Giunta RE. Chances and limitations of a low-cost mobile 3D scanner for breast imaging in comparison to an established 3D photogrammetric system. J Plast Reconstr Aesthet Surg. 2018;71:1417-23.
22. Nogueira R, Eguchi M, Kasmirski J, et al. Machine learning, deep learning, artificial intelligence and aesthetic plastic surgery: a qualitative systematic review. Aesthetic Plast Surg. 2025;49:389-99.
23. Shoham G, Zuckerman T, Fliss E, et al. Utilizing artificial intelligence for predicting postoperative complications in breast reduction surgery: a comprehensive retrospective analysis of predictive features and outcomes. Aesthet Surg J. 2025;45:536-41.
24. Chen YE, Gerstle TL, Liang F, Lee BT. Use of a novel laser projection grid to assess symmetry in breast surgery. Plast Reconstr Surg. 2012;130:231e-3e.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].